Electrospraying for Drug Delivery
Info: 6254 words (25 pages) Dissertation
Published: 20th Feb 2025
Tagged: Sciences
Abstract: Current studies have uncover the expanding significance of nanotechnology in the field of bio-medical application, especially, the drug loading and its delivery. Different conventional techniques including nanoprecipitation, spray drying, solvent evaporation, supercritical fluid extraction, and ionotropic gelation are well investigated and each system advantages and constraints are defined. This review will focus on the electrospraying technique and specific on the parameters of polymeric solution’s concentration, viscosity, distance to the collector and applied voltage. All these parameters are essential for developing desired particles nanoparticles, which will be explained in this review, together with the advantages and challenges of electrospraying technique.
Keywords: Electrospray; particles; process parameters; co-axial
Introduction
Nanotechnology, and particularly the fabrication of nanoparticles, has discovered an uncommon consideration in broad scientific territory over the last few decades. The polymeric nanoparticles (NPs) as a drug carrier is found to be the best alternative method for controlled drug delivery, due to its critical properties such as biocompatibility, nontoxic, biodegradability, and higher drug loading. However, there are many other parameters to be considered while designing Nano carriers, such as size (100-500 nm), shape and morphology, stability to deliver drug to explicit tissues, and the ability for a sustained and controlled release treatment1. NPs have the ability to deliver the therapeutic agents to the targeted site ton explicit tissues/cells and release the drug, and can also encourage the permeation of therapeutic agents through the biological barriers, such as the blood-brain barrier (BBB)2. Polymeric nanoparticles have been formulated by different strategies based on the application and the physiochemical properties of the drugs3. Choosing the best technique, plays an important role in order to develop polymeric nanoparticles with the ideal properties for a specific role or application. These methods are divided in two groups, which depends on the polymerization of monomers (e.g. Emulsion, mini emulsion, micro emulsion) or on the advantage of performed polymers (e.g. emulsification-solvent evaporation, emulsification-solvent diffusion, nanoprecipitation, spray drying, dialysis)4. All these methods are multiple step methods for the production of NPs with an additional step of drying. The utilization of organic solvents, except carefully controlled, is an disadvantage in many of these procedures, especially when protein based drugs are used it may lead to denaturation during process and at the same time increasing the variation in encapsulation efficiency and loading5. Particles developed by using emulsion based techniques are believed to have inhomogeneous and broad size distribution adding to their deficiency of reproducibility, which thusly obstructs their clinical use6. Developing polymeric nano/microparticles by electrospraying can conquer the limitations of emulsion based methods7.
Electrohydrodynamic atomiztion (EHDA) is a procedure that changes ionized liquid jets into fine beads by the influence of voltage8. The procedure uses electromechanical and hydrodynamic forces, which are working together to provide a precise control over size and morphology of the particles. Electrospinning and electrospraying, classified under EHDA and have the distinction in their process parameters and polymer properties, which are essential characteristics to develop particles by the electrospraying technique9. Electrospraying and electrospinning are two highly versatile and scalable electrohydrodynamic methods that have a prospective response to the constraints of NP development, such as reproducibility, scalability and effective encapsulation in pharmaceutical nanotechnology10.
Electrospraying The capacity to reproducibly stack bioactive molecules into polymeric microspheres is a challenge5. Traditional microsphere fabrication strategies commonly will provide a low loading, multiple-step methods and heterogeneous release profiles, ruining their capability for up-scale and introduction to the clinic. To overcome these issues, it was necessary to identify and understand the key parameters in particle formation and learn how these parameters influence the product, i.e. particles morphology and size. Electrospraying, is equipped for conquering these restrictions by the single-step method and simple instrumental setup. Advancements in nanotechnology and nanoengineering have resulted in progress in the fabrication of the particles with optimized properties. Some setups have chambers carrying air/nitrogen stream as a carrier gas to help the movement of particles to the collector. Utilization of these chambers result in controlled evaporation of solvent from the surface of the particles, in order to obtain a smoother surface morphology.
The principles of electrospraying, also known as electrohydrodynamic atomization, are used for the fabrication of the nano/microparticles. High voltage is applied to the polymer solution, when the critical voltage is reached, a charged liquid jet is ejected from the tip of needle in the form of fine droplets and collected11. The needle is connected to a high voltage current act as a cathode and the collector is the bottom plate which acts as a anode. The distance between the needle tip and the collectors can be adjusted during optimisation process. Particles can be collected on the metal plate or aluminum foil or in the solution, depends on application. Also, it’s important to focus on the polarity of the electric potential applied, which can have a wide range of impacts on the electrospraying, with certain materials showing better electrospraying at positive charge than at a negative charge and vice-versa 10. Electrospraying has a simple and one-step setup which can be tailored as per requirement, Figure 1a
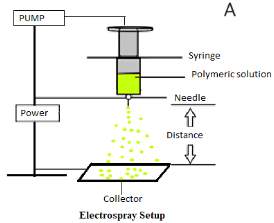
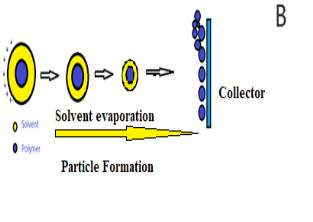
Figure 1: A)Typical electrospraying process and B) Formation of Droplets during electrospraying method.
Evaporation of solvent occurs on the way towards the collector, resulting in the formation of nano/microparticle12. The four main processing parameters, which can influence electrospraying are: applied voltage, flow rate, distance/high between needle and collector and diameter of the nozzle (Figure 1a). Moreover, the technique involves the formation of a Taylor Cone jet on the tip of the needle due to electrostatic force (between cathode and liquid) and columbic forces (between anode and liquid). The fine jet with a charged polymeric solution is then sprayed from the Taylor Cone towards the collector as the electrostatic force overcomes the surface tension of the solution. As the droplet travels from the tip of the needle towards the collector, solvent evaporates rapidly and nano/micro particles or beads are formed13. The concentration of polymeric solution and rate of flow plays an important role in these two techniques. Low concentration polymeric solutions are used for fabricating nano/micro particles by the electrospraying method, whereas high concentration polymeric solution is used in electrospinning to fabricate nano/microfibres14. Using low concentration of polymer solutions (1-2 %) destabilizes the liquid jet due to high voltage applied forming large droplets from the liquid jet which break down into smaller droplets15. Applied electric potential generates charge in the polymeric solution, which then produces coulombic repulsion in the droplets. Higher voltages result in greater repulsion forces between particles, resulting in smaller droplets and therefore smaller particles16. The highest columbic force needed to break a large droplet into the smaller droplets is known as the Rayleigh Limit5. The Rayleigh Limit, LR, determines the breaking of the beads17. This cut off, is the point at which the surface tension pressure of the drop is overwhelmed by the electrostatic forces can be calculated using Equation 1, which is the Rayleigh Limit16; Figure 1b illustrates the droplet formation during the electrospraying process.
LR = q((64π2εγR3)
q = 8π
ε0γR3 .(Eq.1)5
where q = charge on the droplet surface, ε = permittivity of the surrounding medium, γ = surface tension on the liquid, and, R = radius of the droplet.
Electrospraying Modes Understanding the electrospraying modes is essential to manage the process parameters, which consist of applied voltage, flow rate, electrical conductivity, surface tension and the viscosity of the polymeric solution18. The types of electrospraying modes are dripping, micro dripping, spindle, cone jet mode, twin jet and multi-jet modes (Figure 2a). As the applied voltage increases in the electrospraying liquid ,the modes of electrospraying change18. The dripping mode can be explained as the polymeric solution flows directly from the needle and relatively large drops are formed, resulting in elongated spindles being observed in these modes. Dripping mode and micro-dripping mode were observed, when low voltage is applied, the polymeric solution is not viscous and having higher surface tension12. In the Taylor Cone jet modes, the polymeric solution flows into a long and fine jet, which is stable. As the polymeric solution flows into a long continuous jet from the needle, the jet breaks down into droplets due to the electrostatic forces. The droplet size depends on the flow rate and is inversely proportional to the conductivity of the polymeric solution. Hypothetically the droplet size can be calculated by the help of scaling law which affirmed by numerous analysis18 by Eq.2.
dD = α
(Eq.2)
Where ε0 = the vacuum permittivity, voltage = V, flow rate = Q, electrical conductivity = γ, surface tension = σ, density = ρ, (α =2.9)18.
Ideal size droplet can be fabricated if the flow rate and polymeric solution properties are controlled. For instance, the droplet size can be decreased by an increase in the applied voltage or decrease the flow rate of the solution. The initial droplet size (dD) and the final particle size (dp) can be correlated equation 3.
dp = dD
(Eq. 3)
where, c = concentration of solute and
= particle density.
Another more jet mode is observed whereby a couple of fine jets can be seen on the capillary axis. This uncommon mode is known as the multi-jet mode. The unstable cone jet mode is observed most often when the flow rate is not proportionate to the applied voltage, which results in unpredictable mass on the nozzle tip or in the event where the applied voltage is too high. The corona discharge occurs before enough charge is collected in the bead at the nozzle end to form a stable conical shape. The most well-known and utilized electrospray mode is the Taylor Cone jet mode, which can deliver highly monodisperse particles in stable conditions18.
Smeets et al., shows the impact of flow rate on the spraying pattern of the polymeric solution19. Electrospraying of 1% Eudragit® L110 in methanol was carried out at varying voltages of 12-15kV, 15-20kV, and 20-25kV respectively19. By increasing voltage, a pattern of spraying from dripping mode to a spindle mode and finally a stable cone jet mode was obtained. These modes affect the molecule size and morphology, for example, in dripping mode the droplets collected are still wet due to its morphology and didn’t had enough time to dry. Whereas, in the cone jet mode dry and homogenous spheres or particles were obtained (Figure 2).
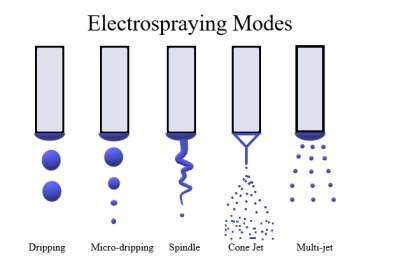
Figure 2. a) Modes of electrospraying method: Dripping, micro dripping, spindle, cone jet to multi-jet mode.
Essential electrospraying process parameters Essential process parameters for developing particles of desired morphology and size incorporated of applied voltage, distance from the tip of the nozzle to the collector, flow rate of the polymeric solution and the type of collector. Polymer related characteristics, which influence the procedure which includes molecular weight (Mw), concentration, viscosity, conductivity, surface tension, and solvent used in the polymeric solution. In addition, temperature, humidity, and air velocity have a direct impact on the speed of solvent evaporation. All these parameters, affect the size, morphology, and performance of the particles developed. Based on these parameters various particles were formulated such as hollow, cup-shaped, porous, cell-shaped, spherical, core-shell, and multi-layered, accordingly (Figure 3)20.

Figure 3: Illustrates different structures of electrosprayed particles
Effect of flow rate and voltage on particle formation The flow rate and applied voltage is considered to be the most important parameters in the formation of ideal particles. Yixiang Xu et. al., formulated BSA-loaded PLA particles with 1,2-DCE as a solvent and 3% of PLA at a flow rate of 0.5ml h-1 by varying voltage (e.g. 10kV, 12.5kV and 15kV) during optimisation process21. At 10kV, the Particles were formed with tails due to the low voltage which is not sufficient to overcome the surface tension of the droplet21 (Figure 3b). Spherical particles were observed when the voltage was increased up to 12.5kV & 15kV. Further increase in voltage creates multi-jet mode, which is not ideal for stable and consistent particles with respect to size and morphology. On increasing the applied voltage from 10kV to 15kV, the average particle size decreases due to the conical meniscus that is formed at the tip of the needle and is forced to disfigure to form smaller size droplets on applying higher voltage. This shows that voltage optimisation is one of the essential processing parameters22. The effect of flow rate on particle size was also studied22, and data shows that at each applied flow rate (0.5 – 3 ml hr-1), the size of particles kept increasing due to the formation of large particles, uncontrolled atomization and uneven distribution of polymeric solution at the tip of the nozzle at higher flow rates21. The authors concluded that at higher flow rate, larger size particles formed due high polymer concentration on nozzle tip resulting wet particles, whereas low flow rates, high electrical conductivity can lead into smaller dry particles23. In addition, it was observed that at each flow rate with high voltage enables to form stable cone jet, which able to produce uniform small particles and acceptable particle distribution
.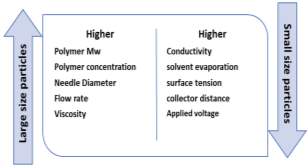
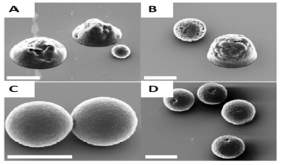
Figure 4: a) Effects of essential electrospraying parameters on the size of particles22; b) Morphology of particles at different concentration of PCL observed under SEM: A) 5%, B) 7.5%, C) 9%, D) 10% (scale bar 5µm)5.
Effects of polymer concentrations
Polymer molecular weight and concentration plays an important role on viscosity and surface tension of the polymeric solution, which will affect the size of the particles. High molecular weight polymers tends to form large particles even at low concentrations. At higher polymer concentrations the density and viscosity of the solution is high, which leads to form a stable Taylor cone jet mode with high flow rate and low applied voltage. During the process, the rate of solvent evaporation is less tend to develop particles with large diameters and high polydispersity index (PDI). It is also explained that at higher polymer concentration, the particle, in general, will aggregate and cannot overcome the surface tension, resulting in larger particles24. In contrast to this, at low concentrations of polymer, the Taylor Cone is unstable due to the high voltage and low flow rates applied, which tends to develop particles of smaller size. A study by José et. al., regards to the effect of polymer concentration on the particle size of gluten nanospheres24. Particles of different sizes, ranging from 0.54-1.42 µm were fabricated using wheat gluten in concentrations ranging from 2-8% (w/v)24, and the data demonstrate that larger size particles prepared when higher polymer concentrations were used..
In a study by Bock et. al., the concentration and Mw of the polymer in the solution show to have a great influence on the morphology of the particle (Figure 4)5. Also, demonstrated that at lower polycaprolactone (PCL) concentration, a particle with flat base was obtained and at higher concentration of PCL more spherical particles where obtained. This can be explained as at high polymer concentration the solvent evaporation is not rapid and incomplete, by which more spherical particles with the smooth outer layer and sphere shape were formed. However, in order to get particles that are completely spherical in shape, it is necessary to evaporate the solvent completely. Polymer chain entanglement is critical to the physical properties of the particles fabricated. At low polymer concentrations, there less potential for entanglement of the polymer chains, where the working system is known as the semi dilute untangled system. In this condition, the concentration is large enough for chains to overlap, but not sufficient to generate a significant degree of entanglement5. This can be possible only when the polymer concentration is equal to or just above the polymer critical entanglement concentration5,22. Figure 4b shows that at lower polymeric concentration particles were observed to have pores on surface, due to insufficient entanglement. The surface morphology of the particles also depends on the intrinsic viscosity of the polymeric solution and type of solvent used in polymeric solution25. At higher polymeric concentration there is high polymer entanglements and at critical entanglement concentration spherical shape particles are obtained with smooth surface morphology (Figure 4b C & D) 26.
Effect of Pressure in electrospraying chamber
Fabrication of particles via the electrospraying process to date has observed the use of ambient conditions of atmospheric pressure. Minimum attention is given to study the effect of pressure on the size and morphology of the particles fabricated. In this respect, Wu et. al., have contemplated the impact of various applied pressure alongside varying the solution concentration on the electrosprayed PCL particles. In this study, experiments were conducted at varying concentrations and pressure. Pressure ranging from 0-380 mmHg was observed different polymer concentrations (e.g. 3, 5 and 7 w/v %) in dichloromethane (DCM). Both these parameters, pressure and concentration) were screened and observed to have a direct impact on the size and morphology of nanoparticles, as the solvent evaporation rate and particle formation rate gets changed27. The distance from the tip to the collector was optimized to 30 cm and voltage at 12 kV. Alteration in pressure demonstrated huge changes in microstructure and morphology. Analysing particles by SEM showed normal molecule size (2, 3.5, 8.5, 26µm; Figure 5) at pressures of 0 mmHg, -150 mmHg, -250 mmHg, and -380 mmHg respectively. By using higher polymer concentrations, fibres with an average size of 0.8 µm were obtained with pressure at 0 mmHg. It was also observed that the particle size expanded continuously when the pressure Decreases. The most likely reason that can be explained for this is fast drying of droplets during jet mode leading to quick evaporation and no repulsion or breakdown of particles into smaller particles. Furthermore, during variations in the chamber pressure and decreasing polymer concentration, they likewise discovered changes in the surface morphology from rough, porous to smooth28.
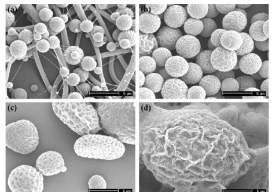
Figure 5. SEM images at higher magnification of PCL micro particles at respective pressure
a) 0 mmHg, b) -150 mmHg, c) -250 mmHg, d) -380 mmHg29.
Effects of solvent properties on electrospray
Solvent properties have a broad effect on the particle size and morphology as it controls the conductivity and viscosity of the polymeric solution25. In general, the surface morphology of a particle is mostly affected by the solvent with high boiling point, which decreases the evaporation rate and also decreases the pore formation in a particle21. The study by Xu et. al., examined the impact of aqueous and organic solvents ratio on the particle morphology21. Results proposed that with an increasing aqueous concentration over the organic solvent, the surface morphology was observed as a more non-spherical shape and rough at the surface of a particle due to the low viscosity of the polymeric solution. The solvent with lower boiling points tends to evaporate at a very early stage, i.e. it starts evaporating at the tip of the needle or just at the Taylor cone jet formation and blocks the needle. To obtain the particles of different morphology, the rate of solvent evaporation and forming dry droplets by the jet mode has a huge role in particle morpholgy27. Another study examined the effect of solvent properties and their electrical conductivity on the particle size and morphology, states that at high electrical conductivity, lower concentration and lower surface tension, particles with small size can be fabricated21.
Coaxial Electrospraying Coaxial electrospray is an electro hydrodynamic method that produces multi-layered nano/microparticles by coaxial electrosprayed jet mode. In correlation with other microencapsulation/nanoencapsulation forms, coaxial electrospraying has a few potential benefits such as effective protection of bioactivity, uniform size distribution., and high encapsulation efficiency30. The major difference in uniaxial and coaxial electrospraying is it utilizes two needles simultaneously in which one solution helps to form outer shell and other helps inner core-shell. In such systems, different polymer solutions were used for controlled formulation31. Hwang et. al., have studied a continuous single-step approach for the preparation of uniform sized particles having polystyrene in core and Poly-ε- caprolactone in the shell (PS/PCL), and polymethylmethacrylate in core and polycaprolactone (PMMA/PCL) in shell31. Using coaxial electrospraying polymeric spherical microcapsules were developed using concentrated polymer solution. This article reveals that the flow rate of the outer shell solution should be higher than the core-shell forming solution to create spherical shape microcapsules or else it could give mixed morphologies of the particles. This process can be further studied by using different polymers for outer and core shell formation31.
Coaxial electrospraying additionally limits introductory burst release as the drug is entrapped in the centre part of particle and surface morphology can be maintained to avoid burst release and resulting in a prolong controlled sustained release32. Utilization of triple coaxial electrospraying for engineering multi-shell particles has in popularity for encapsulation studies. The release profile of drug and size of particles relies upon the relative concentration of polymer solutions and applied voltage. Coaxial electrospraying method with two different capillary feeders can provide a beneficial method to develop core-shell nano-drug delivery systems, without the use of any surfactants or elevated temperatures30. This capability of electrospraying in fabricating core-shell nanoparticles can be used in drug delivery studies, which enhances the drug release kinetics for prolonged period. In addition, multi-drugs can be delivered 32.
Future aspects and challenges in Electrospraying
The future of eletrospraying should head towards the improvement of technical propelled frameworks to fulfil the universal GMP rules for enormous scale transplantation32. This includes controlled frameworks, operational thoroughness and better-quality control. Observing furthermore, irretrievability whether through genetic, biochemical or attractive labelling are key to research to reduce biosafety concerns32. Electrospraying is fit to create fine transporters with controlled shapes/sizes and high encapsulation. It involves sensitive engineering and material control instead of complex biological or chemical modification20. Regardless of its far reaching use in different areas, the major hindrance in commercialization of this system, particularly for large scale manufacturing in industry is its low yield production19. Another restriction is the use of organic solvents during eletrospraying that may harm the bioactivity of genes, enzymes, and cell vitality. “Green eletrospraying,” which utilizes the aqueous solvents, would be the choice to decrease toxic quality33. Bio-eletrospraying (BES) offers us a chance to encapsulate cells legitimately in scaffolds. This system, can be utilized for organs-on-chip, which are applied to impersonate the genuine organ to replace some portion of animal tests20. Another alteration could be utilization of AC (alternate current) supply, which offers better efficiency.
In case of oral drug delivery, tablet manufacturing needs blending properties, flowability, compressibility, etc., for simple handling which is for the most compromised while working with ES. Démuth et. al., have explained tableting process prepared with electrospinning technique and optimized parameters like excipients and fillers,. at various ratios (e.g. …) to obtain the desired properties of a tablet34. Phase separation can be observed in various steps of process like granulation and grinding, which can create phase separation or phase changes and can create gelling of polymer systems and may influence or damage the product35. To overcome this problem, coaxial electrospray is the best alternative.
Regulatory Consideration
…
Conclusion
Electrospraying and electrospinning are novel techniques which have the potential to generate polymeric nano/microparticles of various size and morphology, capable of drug encapsulation and delivery in a sustainable manner. By varying key parameters, electrospraying provides researchers control over the size and morphology of particles, which could be used as novel drug delivery systems. The main advantages are it’s single-step method for particle fabrication and researches are able to get dry particles as the end products, without any additional step required for drying particles. over all, eletrospraying is a highly versatile technique to prepare nano/micro polymeric formulations for genes, drugs, proteins, enzymes, growth factor, and cells20.
Reference
- 1. Rizvi, S. A. A. & Saleh, A. M. Applications of nanoparticle systems in drug delivery technology. Saudi Pharm. J. 26, 64–70 (2018).
- 2. Zhang, J. & Marksaltzman, W. Engineering biodegradable nanoparticles for drug and gene delivery. Chem. Eng. Prog. 109, 25–30 (2013).
- 3. Nasir, A., Kausar, A. & Younus, A. A Review on Preparation, Properties and Applications of Polymeric Nanoparticle-Based Materials. Polym. – Plast. Technol. Eng. 54, 325–341 (2015).
- 4. Crucho, C. I. C. & Barros, M. T. Polymeric nanoparticles: A study on the preparation variables and characterization methods. Mater. Sci. Eng. C 80, 771–784 (2017).
- 5. Bock, N., Woodruff, M. A., Hutmacher, D. W. & Dargaville, T. R. Electrospraying, a reproducible method for production of polymeric microspheres for biomedical applications. Polymers (Basel). 3, 131–149 (2011).
- 6. Yang, G., Sau, C., Lai, W., Cichon, J. & Li, W. 蚊子网状进化HHS Public Access. 344, 1173–1178 (2015).
- 7. Arya, N., Chakraborty, S., Dube, N. & Katti, D. S. Electrospraying: A facile technique for synthesis of chitosan-based micro/nanospheres for drug delivery applications. J. Biomed. Mater. Res. – Part B Appl. Biomater. 88, 17–31 (2009).
- 8. Naderi, P., Shams, M. & Ghassemi, H. Investigation on the onset voltage and stability island of electrospray in the cone-jet mode using curved counter electrode. J. Electrostat. 98, 1–10 (2019).
- 9. Chakraborty, S., Liao, I. C., Adler, A. & Leong, K. W. Electrohydrodynamics: A facile technique to fabricate drug delivery systems. Adv. Drug Deliv. Rev. 61, 1043–1054 (2009).
- 10. Alehosseini, A., Ghorani, B., Sarabi-Jamab, M. & Tucker, N. Principles of electrospraying: A new approach in protection of bioactive compounds in foods. Crit. Rev. Food Sci. Nutr. 58, 2346–2363 (2018).
- 11. Zhang, S., Campagne, C. & Salaün, F. Influence of Solvent Selection in the Electrospraying Process of Polycaprolactone. Appl. Sci. 9, 402 (2019).
- 12. Wilm, M. Principles of Electrospray Ionization. Mol. Cell. Proteomics 10, M111.009407 (2011).
- 13. Boda, S. K., Li, X. & Xie, J. Electrospraying an enabling technology for pharmaceutical and biomedical applications: A review. J. Aerosol Sci. 125, 164–181 (2018).
- 14. Thakkar, S. & Misra, M. Electrospun polymeric nanofibers: New horizons in drug delivery. Eur. J. Pharm. Sci. 107, 148–167 (2017).
- 15. Xie, J., Jiang, J., Davoodi, P., Srinivasan, M. P. & Wang, C. H. Electrohydrodynamic atomization: A two-decade effort to produce and process micro-/nanoparticulate materials. Chem. Eng. Sci. 125, 32–57 (2015).
- 16. Tapia-Hernández, J. A. et al. Micro- and Nanoparticles by Electrospray: Advances and Applications in Foods. J. Agric. Food Chem. 63, 4699–4707 (2015).
- 17. Xie, J., Lim, L. K., Phua, Y., Hua, J. & Wang, C. H. Electrohydrodynamic atomization for biodegradable polymeric particle production. J. Colloid Interface Sci. 302, 103–112 (2006).
- 18. Dai, L. L. Advanced Core-Shell Composite Nanoparticles Through Pickering Emulsion Polymerization. The Delivery of Nanoparticles (2012). doi:10.5772/2647
- 19. Smeets, A., Clasen, C. & Van den Mooter, G. Electrospraying of polymer solutions: Study of formulation and process parameters. Eur. J. Pharm. Biopharm. 119, 114–124 (2017).
- 20. Wang, J., Jansen, J. A. & Yang, F. Electrospraying: Possibilities and Challenges of Engineering Carriers for Biomedical Applications—A Mini Review. Front. Chem. 7, 1–9 (2019).
- 21. Xu, Y., Skotak, M. & Hanna, M. Electrospray encapsulation of water-soluble protein with polylactide. I. Effects of formulations and process on morphology and particle size. J. Microencapsul. 23, 69–78 (2006).
- 22. Pawar, A., Thakkar, S. & Misra, M. A bird’s eye view of nanoparticles prepared by electrospraying: advancements in drug delivery field. J. Control. Release 286, 179–200 (2018).
- 23. Chang, M. W., Stride, E. & Edirisinghe, M. Controlling the thickness of hollow polymeric microspheres prepared by electrohydrodynamic atomization. J. R. Soc. Interface 7, (2010).
- 24. Tapia-Hernández, J. A. et al. Porous wheat gluten microparticles obtained by electrospray: Preparation and characterization. Adv. Polym. Technol. 37, 2314–2324 (2018).
- 25. Bohr, A. et al. Pharmaceutical microparticle engineering with electrospraying: the role of mixed solvent systems in particle formation and characteristics. J. Mater. Sci. Mater. Med. 26, 1–13 (2015).
- 26. Wu, Y. Q. & Clark, R. L. Controllable porous polymer particles generated by electrospraying. J. Colloid Interface Sci. 310, 529–535 (2007).
- 27. Fantini, D., Zanetti, M. & Costa, L. Polystyrene microspheres and nanospheres produced by electrospray. Macromol. Rapid Commun. 27, 2038–2042 (2006).
- 28. Wu, Y., Kennedy, S. J. & Clark, R. L. Polymeric particle formation through electrospraying at low atmospheric pressure. J. Biomed. Mater. Res. – Part B Appl. Biomater. 90 B, 381–387 (2009).
- 29. Chen, C., Liu, W., Jiang, P. & Hong, T. Coaxial electrohydrodynamic atomization for the production of drug-loaded micro/nanoparticles. Micromachines 10, (2019).
- 30. Ho, H. N. et al. Development of electrosprayed artesunate-loaded core–shell nanoparticles. Drug Dev. Ind. Pharm. 43, 1134–1142 (2017).
- 31. Yoon, K. H., Unyong, J. & Eun, C. C. Production of uniform-sized polymer core-shell microcapsules by coaxial electrospraying. Langmuir 24, 2446–2451 (2008).
- 32. Naqvi, S. M. et al. Living Cell Factories – Electrosprayed Microcapsules and Microcarriers for Minimally Invasive Delivery. Adv. Mater. 5662–5671 (2016). doi:10.1002/adma.201503598
- 33. Agarwal, S. & Greiner, A. On the way to clean and safe electrospinning-green electrospinning: Emulsion and suspension electrospinning. Polym. Adv. Technol. 22, 372–378 (2011).
- 34. Démuth, B. et al. Development and tableting of directly compressible powder from electrospun nanofibrous amorphous solid dispersion. Adv. Powder Technol. 28, 1554–1563 (2017).
- 35. Démuth, B. et al. Downstream processing of polymer-based amorphous solid dispersions to generate tablet formulations. Int. J. Pharm. 486, 268–286 (2015).
Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allRelated Content
All TagsContent relating to: "Sciences"
Sciences covers multiple areas of science, including Biology, Chemistry, Physics, and many other disciplines.
Related Articles
DMCA / Removal Request
If you are the original writer of this dissertation and no longer wish to have your work published on the UKDiss.com website then please:




