Allergic Rhinitis: Epidemiology, Pathophysiology and Immunology
Info: 9865 words (39 pages) Dissertation
Published: 9th Dec 2019
Chapter 1
INTRODUCTION
- Allergy
Allergy is an IgE (Immunoglobulin E) mediated type I hypersensitivity disorder involving an abnormal reaction to an ordinarily harmless substance called an allergen/antigen. These allergens functions similar in their natural state as an enzymes (A.B Kay, 2001, NEJ). Prevalence of allergy i.e. allergic rhinitis, atopic dermatitis and allergic asthma are widely distributed in the developed countries (Piliponsky et al, 2013). The major aeroallergen in the western developed countries are House-Dust Mites (Dermatophagoides pteronyssinus: Der p 1 and Der p 2), Timothy grass pollen allergen (Phleum pratense: Phl p 1 and Phl p5), Cat dander (Felis domesticus: Fel d 1) and several tree allergens including birch tree (Betula verrucosa; Bet v 1) (A.B Kay, 2001, NEJ). In 1906 ‘allergy’ was first coined by von Pirquet’s, by recognizing that antigens induce changes in both protective immunity and hypersensitivity reactions (A.B Kay, 2001, NEJ, Piliponsky et al, 2013). However, incidence and prevalence of allergy have increased from the past 50 years.
Both genetic and environmental factors are determined to lead exposure to allergies. According to the world health organisation (WHO) estimated prevalence of population by countries range from 10-40% (Pawankar et al, 2013). Whereas, UK has the highest prevalence rate of allergic conditions, with over 20% of the population affected by allergic disorder. Sensitisation can occur on exposure of external environment with an immune system, such as respiratory system and nasal mucosa, cracked skin and eyes. Sensitisation of an allergen is ability of allergen to elicit Th2 response in which IL-4, IL-5 and IL-13 can drive IgE production by promoting class switching of plasma B cells (Akdis and Akdis, 2015). Following class switching to IgE producing B cells, leads to allergen-specific IgE binding to FCRI on surface of mast cells and basophils, causing sensitisation in allergic patients (Akdis and Akdis, 2015).
- Allergy Rhinitis
Allergic Rhinitis is a heterogeneous disorder, cataloged by inflammation of the nasal mucosa. The most common symptoms of allergic rhinitis are running nose, sneezing, itching and blocking/congestion which usually last from few hours to two or more days (Bousquet J, 2001). Allergic rhinitis is mediated by interaction of allergen to specific IgE antibodies present on mast cells in nasal mucosa. Seasonal allergic rhinitis (SAR) is easy to detect because of its prompt and reproducible onset and offset of symptoms in association with pollen exposure (David P. Skoner, 2001). Perennial allergic rhinitis is often more difficult to identify as compared to seasonal allergic rhinitis (SAR) because of its overlap with sinusitis, respiratory infections and vasomotor rhinitis (David P. Skoner, 2001). In allergic rhinitis responses occurs in two phases, considered as early and late phase responses (David P. Skoner, 2001). Early phase allergic response is IgE-mediated and occurs within minutes of exposure, leading to sneezing, itching and rhinorrhoea. Whereas, late phase allergic response develops after 2-8 hrs of allergen exposure, causing congestion, fatigue, malaise, irritability and possibly neurocognitive defects (David P. Skoner, 2001).
- Epidemiology of allergic rhinitis
Epidemiology reports have shown that allergic rhinitis occurs very rapidly and frequently, however underlying cause of rhinitis is very difficult to interpret. The prevalence of rhinitis ranges from 3% to 19% in many countries (David P. Skoner, 2001). Allergic rhinitis is one of the most common problem in UK, with reported prevalence of 10.1% and 15.3% in kids from 6-14 years old, respectively (Asher MI, 2006) and 23.6-29.2% in adults (Burney P, 1996; Bauchau and Durham, 2004). Whereas, prevalence in the UK and Western Europe has increased dramatically over the past 4-5 decades (Brabeck L, 2004; Gupta R, 2007).
It was also been documented that 17% of asthmatics in UK had allergic rhinitis and showed a significantly increased severity of asthma in concomitant to allergic rhinitis (Price et al, 2005). Furthermore studies have highlighted that the frequency of allergic rhinitis increases with age and positive allergy skin prick test leading to the development of hay fever (Skoner et al, 2001). Allergic rhinitis has a tremendous impact on society effecting quality of life (Blanc PD, 2001), sleep quality and cognitive functions (Marshall and Colon, 1993). Economically allergic rhinitis has a direct and indirect impact on the cost that is 2.7 billion dollars for a year in 1995, which excludes cost associated with medical problem like sinusitis and asthma (McMenamin et al, 1994; Skoner et al 2001 JACI).
- Pathophysiology
Allergic rhinitis is a basic mechanism, ensuing re-exposure of allergen in a sensitised individuals. Allergic sensitisation is characterised by allergic rhinitis that has a strong genetic constituents. Atopic patients are inherited by tendency to develop IgE, mast cells and Th2 lymphocytes immune responses (Skoner, 2001). In normal state IgE is bound to high affinity tetrameric receptor (FCRI), helps in forming a stable complex on the surface of immune cells. The subunit of FcεRI is responsible for IgE binding whereas 2 complexes are involved in signal transduction. Studies have highlighted, for optimal binding, allergen must have two or more binding sites that enable FcεRI cross-linking and surface clustering. Prolonged continuous allergen exposure, increases IgE-coated mast and basophil cells number traverse the epithelium, recognize the allergen and causes degranulation (Naclerio RM, 1991). Furthermore, studies have also suggested that less than 1% clustering is sufficient to induce degranulation in mast cells and basophils (Wu LC, 2011; Knol EF, 2006).
Products of degranulation include release of per-formed mediators such as histamine, chymase, kininogenase, tryptase and other enzymes. These mediators have ability to stimulate sensory nerves within seconds, which expresses the sensation of nasal itch and congestion causing sneezing (Skoner et al, 1991). In addition, mast cells also secrete other inflammatory mediator’s prostaglandin D2 (PGD2) (vasodilator, contributes in the nasal blockade) and leukotrienes. Leukotrienes LTC4, LTD4 and LTE4 acts as a receptor for cysteinyl leukotrienes, causing leakage of blood vessels and produce mucus plus water rhinorrhoea, characteristic of allergic rhinitis. The mast cells derived cytokines plus IL-4 and IL-13 initiates IgE production and mucus secretion, whereas IL-5 is involved in eosinophils survival and activation. There are many other factors, which are involved in promoting the upregulation of adhesion molecules on endothelial cells such as IL-6, IL-8 and TNF-. Basophils share properties with mast cells playing role in late phase reaction (Naclerio R, 1985). Basophils are involved in traffic to the mucosal cells and induce IL-4 and IL-13 (Pawankar R, 2011). They may also play role as an antigen presenting cells expressing MHC class II molecules (Barnes PJ, 2011). Allergen also comes across by APC in a mucosa on re-exposure. The primary cell involved is the dendritic cells, macrophages, mast cells, eosinophils and basophils (Barnes PJ, 2011). However, on peptide presentation to the T cells further leads to the stimulation of Th2 cell response, helping class switching of B cells and secretion of IgE stimulates eosinophils recruitment and mucous secretion by IL-4, IL-5 and IL-13.
Subsets of T-helper cells are likely the cause of chronic inflammatory response to allergens. T helper type 2 cells are involved to promote the allergic responses by releasing cytokines (IL-3, IL-4 and IL-5) and other mediators, which are involved in the IgE production and mast cell recruitment (Durham SR, 1992). Released cytokines from Th2 cells circulate in hypothalamus, causing irritability, fatigue and neurocognitive defect noted in allergic rhinitis patients (Sim TC, 1995). However, memory Th2 cells resides within the local nasal mucosa and lymph node, contributing IL-4 and IL-13 to the late phase response activated by non-specific and allergen specific T cells receptor engagement in situ (through CD154, CD28 and IL-4R). Despite the description of both early and late phase response to allergens, these responses overlap in vivo, causing sneezing, itching and rhinorrhea with early phase response followed by nasal congestion or blockage following controlled single exposure (Scadding et al, 2012).
- Immunology of AR
Allergic rhinitis is a Th2-dominated response, resulting in the stimulation of allergen specific IgE. IgE binds to the surface of mast cells and basophils by FcRI (high affinity receptor) (Stone et al, 2010, Baraniuk, 2001). Within seconds or minutes of allergen inhalation, these cells become activated and triggers cascade of early phase allergic responses. After interaction mast cells release preformed granules (histamine, tryptase, leukotrienes) which are key mediator of early phase allergic responses interacts with nerves, blood vessels and mucus producing cells causing congestion and bleeding in the nasal mucosa (Parikh et al, 2003). Mast cells after performing immediate response further lead to the production of cytokines that promulgate the late phase and Th2 responses.
Mast cells also produce TNF-, IL-3, IL-4, IL-5, IL-6 and IL-13, which recruit eosinophils, Th2 cells and helps to class switching of IgE in B cells (Parikh et al, 2003; stone et al, 2010). However, basophils share many properties similar to mast cells and have shorter life span but don’t reside in the tissues. Late phase response is a second peak of rhinitis symptoms or maintenance of symptoms after allergen exposure from baseline to 8 hours (Scadding 2014, Scadding et al, 2012). After hours of allergen recognition and exposure, leads to reinforce sensitisation. Sensitisation most likely depends on the both intrinsic and extrinsic factors. Intrinsic factors includes epigenetic modifications and genetic predisposition whereas, extrinsic factors involves timing dose, duration and route of allergen exposure (Custovic A, 2015). These allergens are deposited and taken up by dendritic cells (antigen presenting cells) in nasal mucosa. Following entrance, these cells migrates in the regional lymph nodes before presenting fragments of peptides to T cells via MHC class II molecules (Wheatley and Togias, 2015). In order to get significant response intrinsic allergen protease or ligation of innate immune receptors activates epithelium by viral or bacterial antigen acting as an additional stimulus. Murine studies and human studies have highlighted that epithelium is a source of cytokines and chemokines production in lower airways (Jackson DJ, 2013; Gregory LG, 2013; Saglani S, 2013). These epithelial-derived cytokines have ability to promote Th2 cells polarization with direct effects on antigen presenting cells with a high capacity for secreting IL-4 and IL-13 in particular. Priming of dendritic cells promotes the development of Th2 cells from naïve T helper cells (Th0) involving engagement of allergen peptides on the MHC class II molecules with the required T cells receptors (TCRs). Development of Th2 cells releases cytokines IL-4, IL-5 and IL-13 in particular. Further, these T cells derived IL-4 and IL-13 cytokines promotes class switching of B cells to IgE producing plasma B cells by interaction of T and B cells in a regional lymph node. These antibodies (IgE) subsequently binds to the high-affinity IgE receptors (FCRI) on the surface of mast cells and basophils and low-affinity receptor (FCRII) on the surface of antigen presenting cells i.e. B cells. Resulting in the priming of the local mucosa for an inflammatory, allergic response on subsequent allergen exposure (Sin and Togias et al, 2011).
- Treatment of allergic rhinitis
Allergic rhinitis is one of the most common allergic disorders in world but their symptoms vary from mild to severe allergic reactions. Treatment of allergic rhinitis in UK is typically more advanced and thorough which is normally offered in addition to following first line treatment (Scadding et al, 2008). Following accurate diagnosis, they also offer disease education to the allergic rhinitis patients. Patients are informed about their disease based on clinical study, SPT and sIgE and suggested to avoid allergen wherever possible (Mahesh et al, 2009). However on exposure of allergen, second line of treatment is been provided i.e. anti-histamines that provide cost effective benefits (Demoly et al, 2014). Other most effective treatment available for allergic rhinitis is nasal corticosteroids, which is associated with greater symptom relief than anti-histamine and limited penetration of drugs into the systemic circulation (Weiner et al, 1998).
Another, treatment mode is Omalizumab, an anti-IgE monoclonal antibodies (mAbs). Previously, studies have shown omalizumab is effective in reducing symptom scores and improving quality of life in severely allergic rhinitis patients (Tsabouri et al, 2014). When omalizumab is used in combination of subcutaneous immunotherapy, there was 36% reduction in the symptom severity during the pollen season (Kopp et al, 2013). Furthermore, there are also systemic treatments such as oral leukotriene receptor antagonist, which are effective but predictably less than nasal corticosteroids and anti-histamines.
- Allergen Immunotherapy (AIT)
Allergen-specific immunotherapy (also known as hyposensitization, desensitization and specific immunotherapy) is the administration of slow increasing dose of specific allergen until a maintenance dose is attained or symptoms in allergic patients are reduced or diminished (J. Ring, 2005, Bousquet J, 1998, de WeckAL, 2006). In 1911, Leonard Noon was the first who prepared the extract of various pollens and find the right dilution in skin test before he started injecting in patients suffering from hay fever in increased doses (J. Ring, 2011). Prior to the therapy he firstly did conjunctival provocation test (CPT) where is added drops of allergen in patients eye and assessed the redness and inflammatory reactions to measure the effect (L. Noon, 1911). Further in 1914, Robert Cooke introduced the term ‘atopy’ also known as active immunization, however, later in 1922 he further proposed the term hyposensitisation for this type of treatment (Cooke RA, 1922). In 1950s William Frankland conducted the first clinical trials with allergen-specific immunotherapy stating that hyposensitisation was significantly more effective in higher dose than lower dose of allergen while treating immunotherapy. From that onwards allergen-specific immunotherapy is used more and more over the world using different extracts and modalities.
Allergen immunotherapy (AIT) is one of the most reliable treatments for allergic rhinitis, which has disease-modifying effects and shows efficacy following withdrawal of immunotherapy (Durham et al, 1999, Morogna et al, 2010). AIT is endorsed to patients with severe allergic rhinitis where allergen can’t be avoided or poorly controlled by pharmacotherapy (walker et al, 2011). AIT is an expensive form of treatment, which is time consuming and has poor adherence only 18% people complete full 3 years of trial (Kiel et al, 2013). Furthermore, these patients are screened carefully before initiating treatment to receive either sublingual immunotherapy (SLIT) or subcutaneous immunotherapy (SCIT). In grass pollen study where double blind placebo control trial was conducted has shown that efficacy of both SCIT and SLIT has shown reduction in the symptom score (Calderon et al, 2010, Durham et al, 1999). However, protocol and dosing of these two forms of grass immunotherapy is licensed for use only in UK.
SCIT is 100,000 standard quantity-unit (SQ-U) depends on the ability of patients to tolerate injections and serious local reactions which likely leads to the lower dosing before it continues to increase (Durham et al, 1999). Whereas, SLIT tablets contains 75,000 SQ-U of grass pollen allergen that is taken once in a clinic in order to observe any reactions, which is followed by daily consumption at home. In order to consume them at home tablet it is placed under tongue for 1 min and no water or food is allowed for 5 mins. In terms of treatment SCIT is associated with greater risk but also improves greater mean improvement in the quality of life and reduce medication and symptoms score compared to SLIT (Calderon et al, 2010). Additionally, large population-based analysis done in Denmark, SCIT was shown to be protective against developing autoimmune diseases and also induce tolerance in allergic individuals (Linneberg et al, 2012).
- Mechanisms of allergen immunotherapy
AIT decreases the sensitivity of allergen leading to the relief of allergic symptoms and induces tolerance after treatment is stopped (L Cox et al, 2011). AIT triggers the cascade of clinical events, which elicits rapid desensitization to allergen, suppressing allergic inflammation and allergen-specific immune tolerance (Jutel M, 2013, Burks AW, et al, 2013). Further, it promotes the production of blocking antibodies or antiallergen immunoglobulin G (IgG) in serum sample and increases IgA in nasal secretions. Furthermore, decreases the production of IgE that rises in the pollen season (Kuna P, 1989). AIT results in the reduced mononuclear cells production resulting in the histamine release. Patients undergoing immunotherapy have shown suppression of both early and late phase allergic reactions after nasal allergen challenge, which has also shown reduction in the eosinophil cells number in the nasal secretion (Wilson DR, 2001).
AIT has been associated with the reduction in the CD4+ T cells proliferation and alteration of Th cytokines in the allergen challenge site. Whereas, in the skin the mRNA expression level of T helper cells type 1 cytokines production (IFN- and IL-2) is upregulated and decreases the Th2 cytokine (IL-4 and IL-5) responses after allergen exposure following immunotherapy (Vaney VA, 1993, Maggi E, 2010). Naïve CD4+ T cells alters to regulatory T cells that produce anti-inflammatory cytokines IL-10 and TGF-suppresses the Th2-type immune responses and controls allergic diseases (Ethem Sahin, 2016). In B cells they induces the IgG4 and IgA antibodies and suppress IgE production. In addition, B cells further phenotype to B regulatory cells, producing IL-10, suppresses effector T cells and play role in IgG4 production (van de veen, 2016). IL-10 helps to class switch plasma cells from IgE to IgG4 production. Therefore, lymphoproliferative response to allergen reduces after allergen immunotherapy and thus leads to deviation of allergen-specific Th2 to protective Th1 response (Evans R, 1976, Till SJ, 2004, Durham SR, 1996). In addition AIT induces the shift of Th2 cytokine response to IL-10 secreting inducible T regulatory cells having same allergenic epitope that increases in the number and function (Ethem Sahin, 2016). Generally, Treg cells which controls the allergic inflammation have the expression of positive Forkhead box protein 3 (FOXP3) on there surface and induce IL-10-producing T regulatory type 1 cell response. However, Breg cells regulate the production of IgG4 versus IgE and induce allergen specific antibodies towards nonanaphylactic and noninflammatory types (Akdis M, 2014, Jutel M, 2014).
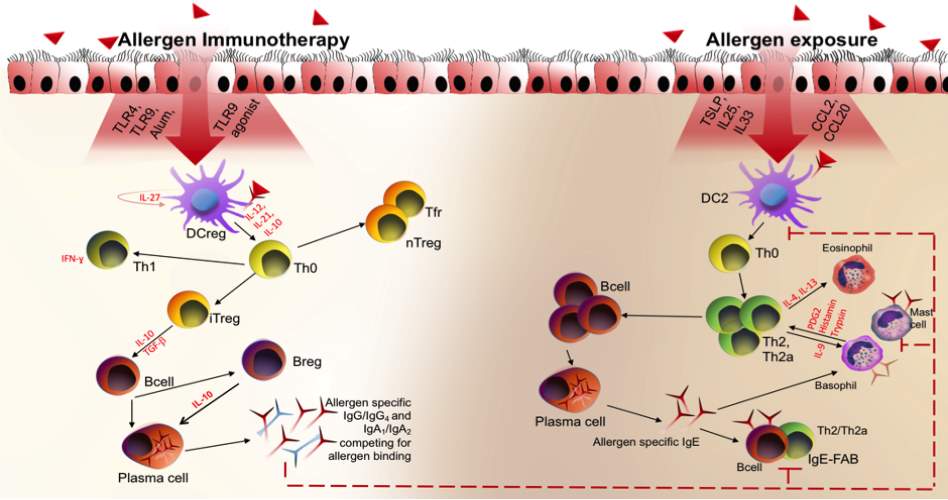
Figure 1. Allergen immunotherapy mechanism. After low dose allergen exposure in rhinitis patients at the nasal mucosal surface epithelial cells activates leading to the activation of DCs. DCs on activation acts as an antigen presenting cells to naïve T cells (Th0) which induce Th2 cytokine response which further activates B cells and class switch them to IgE producing plasma cells. Subsequent allergen exposure leads to the activation and degranulation of effector cells i.e. mast cells and basophils causing early phase allergic reactions. Whereas, after following immunotherapy, high dose of allergen exposure restores DC functions which produce IL-2, IL-27 and IL-10 and promotes immune deviation from Th2 to Th1 response. This further induces Tregs and Bregs responses producing IgG4/IgA antibodies.
- T-lymphocytes
T lymphocytes play a critical role in adaptive and sterile immunity. There are two different types of T cells that exist in the periphery i.e. CD4+ (helper) and CD8+ (cytotoxic) T cells. Main cells of interest in this thesis is CD4+ T cells, plays important role in hypersensitivity and allergic immune mechanism whereas CD8+ T cells are involved in tracking bacteria and anti-viral immunity. Naïve T lymphocytes mature in the thymus and than migrate into the bone marrow without expressing T cell receptors (TCR). Rearrangement of segments (V, D and J) of chains generate enormous majority of TCR diversity in the thymus. Engagement of TCR in order to promote T cells activation requires a threshold of affinity and avidity, addition to co-stimulatory and cytokine signaling (Browlie and Zamoyska et al, 2013). CD4+ and CD8+ T cells recognise protein allergens in context to either major histocompatibility class I (MHC class I) or class II (MHC class II) (Sercarz et al, 2003). Peptides presented by MHC class I are fragmented in the proteasome by cytosolic proteins whereas MHC class II-presented antigens are obtained by phagocytosis of extracellular material, which is antibody mediated. These MHC class II molecules are present on the surface of antigen presenting cells (APCs) such as dendritic cells (DCs), B cells and macrophages. However, naïve CD4+ T cells requires constant interaction with self-MHC class II molecules through either TCR or CD3/CD28 complexes in order to stay alive (Morris and Allen, 2012). T cells require constant binding with APCs in order to perform their function and activation. Naïve CD4+ T cells gets activated and differentiated into T helper cells subsets by cytokine stimuli. Inflammatory cytokines and the engagement of the pattern recognition receptor (PRRs) lead to the expression of co-stimulatory molecules and cytokines production by APCs. Activation of T cells leads to the expression of CD25 along with IL-2 production maintains survival and proliferation.
- T helper cells (TH cells)
T helper cells are involved in the immune regulation, which leads to a re-evaluation of the immunopathogenesis of the enigmatic disease (Markus 1998). Murine and human studies have categorized TH cells into two classes i.e. TH1 (T helper type I) and TH2 (T helper type II) based on their pattern of cytokines secretion. TH1 is characterized by predominant production of IFN- and IL-2, whereas TH2 secretes cytokines i.e. IL-4 (enhance IgE synthesis), IL-5 (increase eosinophil’s growth and differentiation), IL-9 (enhances mast cells differentiation) and IL-13 (increasing mucus production and inducing airway inflammation cause of asthma) (M. Willis-Karp, 1999, D. Umetsu, 2002). This distinct cytokine patterns are associated with specific function (Mosman, 1989). TH1 are involved in cell-mediated immune responses whereas TH2 cells are critical for humoral immunity and are involved in IgE synthesis (E.Maggi, 1988). Studies have shown that following allergen immunotherapy, Th1 cytokine production increases dramatically causing suppression in allergic inflammation and induce tolerance to allergen effect (Shamji and Durham, 2017). Th2 are the most crucial cell orchestrating allergic inflammation, mucus production and severe allergic reactions. Previously studies have suggested that Th2 cells accumulate in the target organs in different allergic reactions. T cells are the dominant source of IL-4, where initiation of Th2 response requires an IL-4 independent polarizing factor. IL-4 also helps Th2 cells development, which are not derived from APCs but absence of IL-2 or IFN-, makes them more dominant (Cook and Miller, 2010, Wan, 2014).
During allergic reactions allergen enters the body through respiratory tracts or skin and are accosted by B cells and macrophages (Singh VK, 1999). These antigens are processed and presented by APCs to naïve T cells, which activates into Th2 cytokines producing cells i.e. IL-4, IL-5 and IL-13. IL-4 and other Th2 cytokines recruit B cells, macrophages and eosinophils all that are involved in IgE synthesis (E.Maggi, 1998). Furthermore, IgE goes and attach to the surface IgE receptors on mast cells and basophils. Subsequent, allergen exposure leads to the activation and activation of cells releasing mediators i.e. histamines, leukotrienes etc that leads to the clinical manifestation of allergic reactions (Singh VK, 1999 & Roitt I, 1998). In addition, patients undergoing immunotherapy injections of purified allergens used to successfully treat allergic reactions have shown reduction in IL-4 responses that is mediated by enhancement of Th1 cytokines. These Th1 cells produce IFN- or IL-2, which suppress IL-4 production. Therefore, leads to suppression of allergic reactions.
- T regulatory cells (Tregs)
The term ‘T regulatory cell’ means cells that actively control and suppress the function of other cells. Regulatory T cells controls development of autoimmune disease and transplant rejection, also playing a analytical role in the suppression of allergic inflammation in allergic disorders (Omid Akbari, 2003). Tregs can also be classified in two categories on bases to there derivation: thymus derived natural Tregs which induce tolerance to self-antigen and peripheral Tregs derived from conventional naïve CD4+ T cells after allergen exposure (Bilate A.M. 2012). There are three major types of Tregs, which are described as: TR (IL-10-producing Tregs), iTR35 (IL-35-producing) and FOXP3 expressing Tregs. Complex functioning of Tregs is not well established but they maintain tolerance by numerous mechanisms. Tregs induce initiation of allergen specific immune response by promoting tolerogenic response by DCs (Dendritic cells). Suppression of DCs is mediated by the expression of CTLA-4, which is expressed in Tregs as negative co-stimulatory molecule. Tregs also acts directly on the dendritic cells by decreasing expression of CD80/CD86 and suppress Th2 immune responses (Wing K, 2008). Furthermore, Tregs also inhibits Th2 cells development by inhibiting the secretion of Th2 cytokines (IL-4, IL-5, IL-9 and IL-13) and also effects Th1 development by inhibiting IFN- production (Martin-Orozco, 2017). Recently, its been validated that peripheral induced Tregs suppress ILC2 response and inflammatory cytokines associated with them i.e. IL-5 and IL-13 (Martin-Orozco, 2017). In addition, Tregs also controls effector cells function by suppressing degranulation of mast cells/basophils by reducing expression of FCRI. Due to this Tregs are involved in the reducing inflammation at the local area and also effects damaged tissue (Kashyap. M 2008, Gri. G, 2008, Nonaka, M, 2008). Humoral immune response is also modulated by Tregs. Further, these cells inhibit the progress of allergen specific B cells by reducing production of IgE and increases level of anti-inflammatory immunoglobulin IgG4 (F.Meiler, 2008). Therefore, Treg cells are the most important in modifying allergic reactions.
- T regulatory cytokines in allergen tolerance
One of the other most important functions of Tregs is inducing allergen tolerance by secretion of soluble mediators also known as suppressive cytokines i.e. IL-10, TGF- and IL-35.
1.5.3.1 Interleukin 10 (IL-10)
IL-10 protein is a homodimer, which is formed of 178 amino acids (P.Vieira 1991, A.Zdanov 1996). Apart from Tregs they are also synthesized and secreted by wide range of cells including macrophages, monocytes, dendritic cells and Bregs. Two chains form IL-10 cytokines receptors: IL-10-R1, expressed on the surface of target cells and IL-10-R2, expressed universally. Signaling is activated by phosphorylation of STAT pathway formed by ligand-receptor binding. Following this activation IL-10 are produced which has an immunosuppressive effects (D Calzada et al, 2018).
IL-10-producing Tregs also plays a vital role in protecting against inflammatory immune response and autoimmune diseases. There insinuation in inducing tolerance and effecting allergic responses is well studied and understood. DCs isolated from allergic patients shows less IL-10 production as compared to the healthy individuals. (O.Palomares, 2010, V.Pacciani, 2010) Cells involved in the allergic diseases modules IL-10 activity by mast cells, Th2 cells and eosinophil’s. Exposure of higher level of allergen induces IL-10 production by Tregs, which are associated with induction of Bregs, production of IgG4 antibodies and improvement of clinical symptoms (Platts-Mills, 2001).
- Tumor growth factor- (TGF-)
TGF-are a pleiotropic cytokines that are member of very complex superfamily, which are involved in maintaining peripheral tolerance. TGF-also regulates the homeostasis of lymphocytes by inhibiting Th cytokine responses (Th2 and Th1), which converts naïve T cells to Tregs by expression of Foxp3, promotes CD4+CD25+ Tregs generation and inhibits allergic airway diseases (L.Gorelik, 2000, 2002, V.L Heath, 2002). TGF-inhibits antibodies secretion by B cells, macrophages proliferation and blocks the expression of high affinity receptor (FCRI) on mast cells. In damaged tissues, TGF-regulates airways inflammatory response, including fibrosis (M. Akdis, 2014).
- Interleukin 35 (IL-35)
IL-35 is a heterodimer, which belongs to IL-12 family, having immunosuppressive effects. IL-35 is composed of two subunits p35 (IL-12/ IL-12) and Ebi3 (Epstein-Barr virus induced gene 3) (D. Hu, 2017). However, other cytokines of IL-12 family (IL-13, IL-23) are considered as pro-inflammatory cytokines, having suppressive effect (Huang et al, 2017). Previously in vitro and in vivo studies have shown that Tregs lacking Ebi3 or IL-12p35 were defective (Min, 2017). Furthermore, many others studies on respiratory diseases have supported the importance of Tregs and its benefits in reducing allergic inflammations. However in asthma IL-35 don’t have a significant role but are involved in the pathogenesis of allergic diseases (W. Niedbala, 2007). Therefore, following sublingual allergen immunotherapy, level of IL-35 in serum increases which is associated with reduction in pathogenesis following improvement in clinical symptoms.
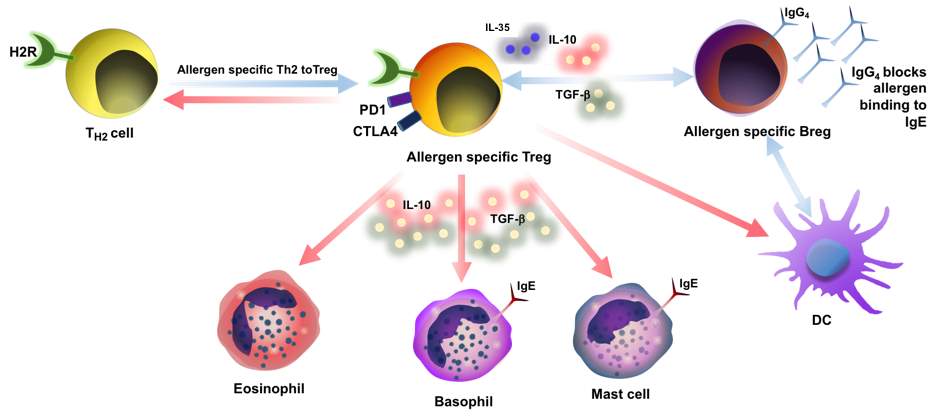
Figure 2: Mechanism of Regulatory T cells. Allergen specific Tregs, switch from Th2 cells to Tregs involved in the progress of tolerance. Effector cells causing allergic inflammation i.e. mast cells, basophils and eosinophil’s are also controlled by suppressive function of Tregs. Tregs secretes IL-10, IL-35 and TGF- suppress these cells. Cytokines IL-10, IL-35 and TGF- suppress production of IgE by B cells, and meanwhile IL-10 triggers IgG4 production. IL-35, IL-10 and IL-10-producing DCs induce regulatory B cells (Bregs), plays an important role in inducing IgG4 production by switching plasma cells.
1.4 B-lymphocytes
B-lymphocytes plays important role in immune system by producing antibodies. In 1965, B cells were first named, when developed in the bursa of Fabricius of chicken (primary site of B-cell development in birds), distinguishing it from T cells, which are derived from thymus (B. David Stollar, 1998). In humoral immunity B cells are the central elements playing a protective part in adaptive immunity, against pathogens and allergens (Kathrin Pieper, 2013, JACI). Defects in B-cell development and functions leads to immune defects causing autoimmunity and allergy (Kathrin Pieper, 2013, JACI). Immature B cells are developed in bone marrow and are selected against autoreactivity by stromal cell expression of self-antigens. B cells are the potent antigen presenting cells (APCs), which have ability to develop and activate T cells directly and are capable of producing variety of inflammatory and pro-inflammatory cytokines (Barr et al., 2012, Mauri and Bosma, 2012). However, complex and diverse roles of B cells in vivo are important, to understand the current ever-increasing use of monoclonal antibodies to deplete B cells in the treatment of autoimmunity.
1.4.1 B-cells Development
Early stage of B cells development occurs in the fetal liver prenatally, followed by maturation in the bone marrow throughout life. In adaptive immune system B cells are responsible for mediating the production of allergen-specific immunoglobulin, which helps to fight against intrusive pathogens (Ali Rohganian, 2014). During early stages of maturation, progenitor B cells progresses from pre- to pro- B cells, rearranging heavy and light chain genes until they express IgM receptor on the cell surface as immature B cells (Joshua A. Boyce, 2013). Through B cells development in bone marrow the rearrangement of heavy chain (H-Chain) genes VH, DH and JH at the pro- B cell stage is a major checkpoint. If this rearrangement is successful, it will combine with the substitute light chain (L-Chain) genes VL-JL to from pre-B cell receptor, causing transition into the pre- B cell stage providing allelic exclusion. Furthermore, receptors presents on pre-B cells expresses which leads to the proliferation of pre-B cells and forms a complete IgM receptor by attempting for light chain rearrangement. However, each light and heavy chain pair comprises a fragment of antigen binding (Fab) region, allowing binding of single antibodies to two antigens.
Immature naïve B cells leaves bone marrow as transitional (T) subsets in blood, expressing both IgM and IgD isotypes as rearranged B cells receptor by alternative splicing also known as CD4hiCD38hi subsets (Marie-Cardine et al, 2008; Sims et al, 2005; Chen and Cerutti, 2011, Vossenkamper et al, 2012). Human CD24hiCD38hi B cells make up a small proportion of CD19+ B cells which are associated with regulatory capacity (Blaire et al, 2010) leading to the production of IL-10 as discussed further below (Garaud et al, 2009; Gary-Gouy et al, 2002; Mageed et al, 2012). In the spleen transitional B cells acts a checkpoint for maturation of marginal or follicular zone B cells. Also they are highly prone to death and require stromal cells or IL-4 and CD40 signaling for cell cycle progression and survival (Sims et al, 2005), B cell receptor (BCR) signaling also acts as a regulator of autoreactivity by promoting cell death. Recently BCR signaling has also received the utmost clinical attention, which might turn out to be beneficial for B-cell driven autoimmune conditions (Chang BY et al, 2011; Hutcheson J, 2012).
1.4.2 Germinal Centre Interaction
Germinal centre are the site of B cells undergoes maturation, proliferation and class switching. However, transitional B cells loose the expression of CD24 and CD38 by becoming follicular or marginal zone B cells (Sims et al, 2005). A follicular B cell circulates in the B cells zone of the spleen and secondary lymphoid organs, whereas marginal zone B cells occupy the margins of red and white pulp of spleen (Weill et al, 2009). Marginal zone B cells can become short-lived IgM-producing plasma cells producing rapid response towards pathogens in absence of T cells. In more than 80% cases these cells express high level of surface IgM and CD27 under somatic hypermutation (Garraud et al, 2012). Follicular B cells circulate through the secondary lymphoid which replies to BCR or antigens allied interactions (Gonzalez et al, 2011; Shlomchik and Weisel, 2012). B cells directly interact with follicular DCs by CXCR5, which are expressed on surface of B cells, responding to a gradient of CXCL13 produced by stromal cells (Cyster, 2010). Follicular dendritic cells are non-proliferative antigen-presenting cells of lymphoid organs, they complement receptor in the absence of MHC, which traps antigen complexes in their confirmation form and present them to B cells. Following successful BCR signaling follicular B cells requires the presentation of CD40L by activated T cells. Activated follicular B cells gets converted either into plasma cells, which secrete high affinity, somatically hypermutated and class-switched antibodies or extra follicular plasmablasts, which secrete low affinity antibodies (IgM) leaving the follicle.
Germinal centre is well established and consist of two zones i.e. light and dark zone. In light zone B cells contact with T cells and follicular Dendritic cells which are subjected to the antigenic selection, while in the dark zone class switch recombination takes place. Class switching recombination involves the pairing of the rearranged and mutated V, (D) and J sections with different constant region, allowing expression of IgG1-4, IgA1, IgA2 or IgE depending on the T cells production. Importantly, in the pathogenesis of allergic rhinitis and allergen immunotherapy tolerance is induced by switching of IgE to IgG1, which is mediated by production of IFN- (Davies et al, 2013). Further, IgG4 is driven by IL-10, which further drives IgG4 and inhibits switching of IgE (Aalberse et al, 2009). B cells in germinal centre differentiate into plasma cells, which are antibody secreting cells that exist in the bone marrow whereas memory B cells recirculate through lymphoid organs. Plasma cells get differentiated by transcription factor BLIMP-1 that has high affinity for BCR signaling along with cytokines signaling (IL-21) (Gatto and Brink, 2010, Eibel et al, 2014; Shlomchik and Weisel, 2012). However, memory B cells are predominantly CD27+IgD– leaves germinal centre reaction earlier, having BCRs with lower affinity than plasma cells (Giesecke et al, 2014).
1.4.3 Cytokine-producing B cells
B cells are known to produce cytokines by TLRs (Toll like receptors) or additional BCR stimulation. Ligation of B cells by TLRs have shown to trigger the generation of class switching B cells under germinal centre conditions and drives the differentiation of plasma cells (Pone et al, 2012; Huggins et al, 2007). Signaling by TLR contributes in switching the decision by B cells. However, in PBMC cultures, TLR4 and TLR9 agonist have been shown to drive IgG, IgA and IgE switching in presence of IL-4 and CD40L (Sackesen et al, 2013). B cells also produce IL-10, IL-6, TNF- and IL-8 following TLR1, 4, 7 and 9 stimulation (Agrawal and Gupta, 2011). Moreover, human B cells following CD40 and TLR9 stimulation induce IL-12 and are capable of inducing IFN- by Th1 cells. The production of immunoregulatory cytokines by B cells will be discussed in next section.
1.5 Regulatory B cells (Bregs)
The perception of suppressor or regulatory B cells was originally orchestrated in the mid 1970s by their ability to maintain suppressive function by producing ‘inhibitory’ antibodies (A. Morris 1968, Claudia Mauri 2008). In 1982, Kennedy and Thomas using murine model was able to described the differentiation of antigen-specific Bregs that were able to remove antihapten immune response (Kennedy and Thomas, 1983). Further, in vivo studies have also highlighted that spleen-cell population depleted antigen presenting cell population was able to induce partial tolerance by MHC alloantigens. B regulatory cells (Bregs) were first termed by Bhan and Mizoguchi in 2002 (Claudia Mauri 2008) while studying the putative pathogenic role of B cells in the expansion of colitis (Mizoguchi et al, 1997). He found that B cells and their autoantibodies can suppress colitis in mice models and later that B regulatory cells (Bregs) upregulates CD1d and enforce regulatory functions through their production of IL-10 (Mizoguchi et al. 2002). The regulatory B cell (Bregs) represents a small population of B cells, which participate in supporting immunological tolerance via its immunosuppressive abilities (Rosser and Mauri. 2015). They achieve this immunosuppressive capacity by secreting the cytokine IL-10 that inhibits the production of IL-2, IL-3, IFN- and TNF- also known as pro-inflammatory cytokines (Rosser and Mauri, 2015). Increased IL-10 secretion is also associated with a range of immune-suppressive properties including increase in the allergen specific IgG4 production and suppression of IgE antibodies (Van de Veen, W. 2017). These IgG4 function as a allergen neutralizing antibodies thus preventing cross-linking of receptor bound IgE thus culminating in the lack of an allergic response (James and Till, 2016).
Overall, these initial studies highlight the existence of Bregs but didn’t clarify the functional difference of Bregs from other B cell subsets. Characterisation of Bregs was made difficult by several mystifying factors. Firstly, IL-10 that has immunosuppressive ability, also acts as a growth factor promoting B cell maturation into antibody producing plasma cells that leads to rheumatoid arthritis (Calame, 2001), systemic lupus erythematous (Edwards 2006; Rahman et al, 2008). Secondly, activation of Bregs requires CD40, which leads to B cell maturation and antibody production (Kehry MR, 1996). Thirdly, no master transcription regulator factor is known till date in Bregs development (Claudia M, 2012). However, many studies have expanded identification of wide variety of B cells associated suppressive functions, including inhibition of Th1, Th17 or Th2 mediated responses, conversion of effector T cells to T regulatory cells and maintains the expression of Foxp3, these studies have supported the existence of distinct Bregs subsets (Gray M, 2007; Carter NA, 2011).
1.5.1 IL-10-producing regulatory B cells (B10)
IL-10 is an anti-inflammatory cytokine causing immune modulation and exerts suppressive effects on most hematopoietic cells (Geunwoong Noh, 2011; Dilillo DJ, 2010). IL-10 is involved not only in the prevention of Th2 response but also inhibits the Th1 polarization. Secretion of pro-inflammatory cytokines by macrophages and monocytes are suppressed by IL-10 (Roncarolo MG, 2006). Release of IL-10 in the local environment leads to more cell-to-cell interactions than systemic IL-10. Furthermore, expression of IL-10 is also critical for the generation of immunological tolerance in the mucosal environment (Berg DJ, 1996). IL-10-producing B10 are rare population originated within spleen. B10 cells are categorized as CD1dhiCD5+, which is a useful novel character of these B cells for analyzing their distribution (Yanada K, 2008), whereas in human it’s categorized as CD27+CD24hi. Regulatory B10 cells are derived from progenitor B10 (B10pro) cells. Prolonged stimulation of B10 cells with anti-CD40 mAb and LPS induces CD1dhiCD5+ B cell to mature converting B10pro cells to B10 (Dilillo DJ, 2010). However, development of B10 cells is influenced by B cell receptor (BCR) (Yanaba K, 2009). Whereas, frequency of B10 cells decreases in absence of CD19, enhancing transmembrane, humoral immunity and BCR signaling (Geunwoong Noh, 2011). BCR (CD40L) dramatically increases CD27+CD24hi and IL-10-producing B cells (Yanaba K, 2008) (Figure 3). Furthermore, diversity of BCR and CD19-generated signals are precarious for normal B cells development, which enhance further B10 cell generation. Other than BCR expression requirement for B cells development, stimulation of B cells with mitogenic anti-IgM antibodies doesn’t induce cytoplasmic IL-10 expression (Yanaba K, 2009). Development of B10 is mostly T-cell independent. In vivo B10 and B10pro cells are identified in blood, bone marrow, spleen and lymph nodes of the mice, however in humans they are found in blood (DiLillo DJ, 2010, Yanaba K, 2008, Matsushita T, 2008).
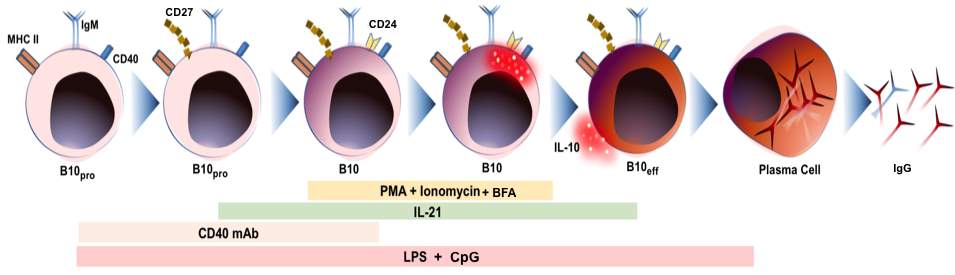
Figure 3: In vivo B10 cells differentiation model in humans. B10 cells originate from progenitor B10 cells (B10pro). CD27 and CD24 are expressed on the surface of adult human blood. IL-10 competent B10 cells are expressed on the surface of B cells following PMA (Phorbol-12-myristate-13-acetate), Ionomycin and Brefeldin A (BFA) stimulation for 5 hours. B10 cells development in human takes place in presence of LPS or CpG. B10 cells in human gets differentiated into B effector cells by IL-10, which further gets differentiated into plasma cells secreting Immunoglobulin (IgG). Furthermore, B10 cells also get differentiated into memory B cells (B10M) needs further confirmation and validation.
1.5.2 Phenotype of Bregs
Bregs cells vary with distinct phenotypic characteristics and mechanism of suppression in mouse and humans (Van de veen, 2016). Multiple subsets of Breg cell have shown similarity in phenotype and effector functions. CD19+ B cells express high level of CD1d in both mice and humans. Both marginal zone and transitional zone comprises of CD1dhi B cells having pathogenic and suppressive effect. For proper analysis of Bregs two markers should be used, as single marker is not adequate to give reliable results. Furthermore, the combined expression of CD1dhi and CD5 helps to identify rare splenic IL-10-producing B cells population (B10). Marker CD5 is expressed on autoreactive B cells which upregulates following surface Ig cross-linking. In vitro studies have shown marginal zone (MZ) B cells produce IL-10 upon stimulation by CpG or CD40L. However in mouse model IL-10 producing B regulatory cells have been identified; these include transitional 2 marginal-zone precursor (T2-MZP) cells (Evans et al, 2007; Rosser et al, 2014), CD5+CD1d+ B cells (B10) (Yanaba et al, 2008; Yoshizaki et al, 2012), marginal zone (MZ) B cells (Gray et al, 2007), CD138+ plasma cells (Neves et al, 2010; Shen et al, 2014) and plasmablasts (Matsumoto et al, 2014). However in human CD27+CD24hiCD48hi B10 (proB10) cells (Evans JG, 2007), CD19+CD24hiCD38hi immature cells (Blair et al, 2010; Das A et al, 2012), Br1 cells (Van de veen et al, 2013) and plasmablasts (Matsumoto et al, 2014)(Table 1).
Table 1: Breg cells subsets
| Name | Phenotype Mouse | Phenotype Human | Suppressor Molecule | Induction | Reference |
| B10/proB10 | CD19+CD1dhi CD5+ | CD27+CD24hi CD148hi CD48hi | IL-10 | LPS, CpG, CD40L | Horikawa et al, 2013, Iwata et al, 2011, Yanaba et al, 2008 |
| Immature /Transtional B cells | CD19+CD21hiCD23hi | CD19+CD24hiCD38hi | IL-10 | Anti-CD40 (Mice),
CD40L |
Blair et al, 2010, Bosma et al, 2012, Floresborja et al, 2013 |
| MZ B cells | CD24hiIgMhiIgDlo CD1dhi | – | IL-10 | Gray et al, 2012, Bankoti et al, 2012 | |
| Plasma cells | IgM+CD138hiCXCR4+ CD1dint | – | IL-10 & IL-35 | Oxaliplatin | Shen et al, 2014, Nerves et al, 2010 |
| Plasmablast | CD138+CD44hi | CD27intCD38+ | IL-10
Immunoglobulin |
CpG-ODN, IL-6 & IL-2 | Matsumot et al, 2014, Claudia Mauri et al, 2015 |
| BR1 cells | – | CD25+CD71+ | IL-10 & IgG4 | CpG-ODN | Van de veen et al, 2013, Claudia Mauri, 2015 |
| B1a cells | CD19+CD5+ | – | IL-10 | TLR ligation, anti CD40, LPS | Lal G, et al, 2016; Shimomur y, 2008 |
1.5.3 Induction of Bregs
Diversity in the phenotype of Bregs has shown their immunosuppressive capacity both in mice and human. Different type of B cells regulates immune responses by multiple mechanisms (Table 2). Though function of IL-10 both in mice and human has been very useful in defining the population of suppressive B cells. Many different surface markers are used to identify population of Breg cells, which can be either up- or down regulated during the immune mechanism. Previously studies have suggested two models of Bregs development, firstly, Bregs, which are similar to thymus-derived Tregs are true B cells lineages controlling specific factors of genes responsible for immune suppression (Claudia Mauri et al, 2015). Secondly, after stimulation with certain factors these B cells converts into B regulatory phenotype in order to suppress local inflammation (Shen et al, 2014; Claudia Mauri et al, 2015). However a lot is known about Bregs but no study has been performed which states the gene array and lineage-specific marker equivalent to Foxp3.
Table 2: Marker and functions of human B regulatory cells
| Marker | Human | Function |
| CD1d | Yes | Antigen presentation of Lipids to T cells |
| CD5 | Yes | Reduction of BCR signaling in B1 cells |
| CD19 | Yes | Activation of BCR |
| CD27 | Yes | Memory B cells marker; induces T cell costimulation and B cells activation |
| CD38 | Yes | Marker for activation; also monitors calcium levels |
| CD138 | Yes | Plasma cells marker; mediates B cells-matrix interaction |
| CD24 | Yes | Promotes allergen driven B cells proliferation and don’t allow differentiation of plasma cells |
| CD71 | Yes | Regulation of cellular Fe+ uptake |
| IgM | Yes | BCR functions through binding of cognate antigen; role in complement activation |
| IL-10 | Yes | Anti-inflammatory cytokines |
1.5.4 Regulatory B cells in autoimmune diseases
Many animal studies have characterized the regulatory function of B cells in various models of inflammation, cancer and autoimmune diseases. B cells are considered to play pathogenic role in autoimmune diseases as they produce autoantibodies causing target tissue damage (Edwards JC, 1999). Furthermore, these autoantibodies can also exert these protective antibodies by triggering the clearance of apoptotic cells (Shimomura Y, 2008). These B cells they acts as an antigen presenting cells, leading to the activation and amplification of native CD4+ T cells responses and can induce tolerogenic effect (Shlomchik MJ, 2001; Rodrigue-Pinto D, 2005; Yan J, 2006). Furthermore, B cells unlike T cells are also called B effector type 1 and 2 cells. B effector 1 cells produce pro-inflammatory cytokines IFN-, TNF- and IL-12, whereas, B effector cells 2 produce Th2 cytokines i.e. IL-4 and IL-13. Remarkably, regulatory B cells induce IL-10 or TGF- production, which possess inhibitory functions in autoimmune diseases (Mizoguchi A, et al, 2006). Previously, murine studies have shown that IL-10-producing Breg cells secrete IL-10 that has a therapeutic effect due to its short half-life.
In rheumatoid arthritis (Ma et al., 2013) patients, B regulatory cells transferred to the local inflammatory sites and reside in the joint tissue. Hence, Bregs can exert regulatory function in the local target site depending on the homing capacity and the survival signal in their local environment (Min Yang et al, 2013). This is also true for other autoimmune diseases i.e. Multiple sclerosis (Correale et al, 2008; Knippenberg et al, 2013), systemic lupus erythematous (Blair et al, 2010). One study has also concluded that Breg cells in autoimmune diseases exert suppressive effects by secreting anti-inflammatory cytokines (IL-10 and TGF-b) and are engaged in cell-to-cell interaction by activating costimulatory marker (Min Yang et al, 2013). Additionally, Bregs can also regulate the balance between T helper cells.
1.5.5 Regulatory B cells in allergies
After initial studies in 1979 on a guinea pigs model for hypersensitivity, very limited studies have validated the existence of the IL-10-suppressive B cells in allergic disease models. However, studies have shown that mice lacking CD19 gene (CD19-/-) was also lacking IL-10 producing B cells which increases hypersensitivity responses that have pointed role of these cells in regulating skin allergic diseases (Watanabe R, 2010). Following this, a specific deficiency in IL-10-producing B cells dramatically increased the allergic skin inflammation in mice caused by accumulation of effector T cells molecules and IFN- production in the skin (Jin G, 2013). However, injection of CpG reduces inflammation in a late-phase allergic conjunctivitis model. Further flow cytometry analysis revealed that CpG treatment significantly increased the proportion of IL-10-producing B cells, which confines inflammation and inhibits eosinophilia (Miyazaki D, 2009).
Other studies have highlighted that B cells isolated from helminth-infected mice shown protective function in allergy by controlling airways inflammation and fatal anaphylaxis via IL-10 production (Amu S, 2010, Mangan NE, 2004, van der Vlugt LE, 2012). Studies on asthmatic allergy had revealed that IL-10-producing regulatory B cells was significantly lower in blood of CD24+CD27+ human subjects as compared to healthy control (van der Vlugt LE, 2014). Additionally, CD24+CD27+ patients demonstrated a nonoptimal induction of IL-10 following LPS and allergen stimulation resulted in the partial induction of regulatory T cells in vitro (van der Vlugt LE, 2014). On the other hand, CD24+CD27+ healthy patients produce more IL-10 and regulatory T cells in same stimulated systems. This suggests that Breg cells from asthmatic patients don’t optimally respond to LPS, contributing in increasing inflammation in asthma (van der Vlugt LE, 2014).
1.5.6 Bregs in allergen Immunotherapy
Allergy is the result of an imbalanced response to the mild environmental antigen leading to the production of Th2 cells and IgE production (van de veen, 2016). However, understanding the mechanism driving allergic responses to allergen is critical for the development of novel strategies to induce allergen tolerance. Allergic patients can be benefited from these therapies that have a disease modifying effect on the allergic immune response towards a healthy response (van de veen, 2016). Allergen Immunotherapy (AIT) is the only curative treatment for the allergic diseases, which is considered clinically beneficial in inducing allergen tolerance (Jutel M, 2016; Jutel M, 2015). Many different regulatory mechanisms are involved in induction of tolerance. As discussed above Breg cells secrets IL-10 and TGF- that can suppress allergic inflammation mediated by allergens. Furthermore, they also promote allergen tolerance through preferential promotion of IgG4 antibodies by differentiating plasma cells (van de veen, 2016).
In 1998, bee venom study highlighted that frequency of IL-10-expressing human B cells were significantly increased in bee venom allergic patients after receiving AIT. However, more evidences on Breg cells in allergen tolerance induction were further obtained when B cells were ligated of CpG (TLR9 agonist) releasing IL-10. This IL-10 was further collected using IL-10 secretion assay, which allowed isolation of live–IL-10+, and IL-10– B cells (van de veen, 2016). IL-10 has also been shown to promote IgG4 production by B cells (C.A Akdis 1998). This was further confirmed when purified naïve IL-10+ Br1 cells produced higher IgG4 than IL-10– B cells (Mubaccel Akdis, 2013). However, during AIT, IgG4 levels increases particularly decreasing IgE/IgG4 ratio (M. Akdis et al, 2014). Taken together, this data we can demonstrate that B regulatory cells do merge a key element in triggering healthy immune response to allergens and induction of IgG4 skewed the humoral immune response.
Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allRelated Content
All TagsContent relating to: "Medicine"
The area of Medicine focuses on the healing of patients, including diagnosing and treating them, as well as the prevention of disease. Medicine is an essential science, looking to combat health issues and improve overall well-being.
Related Articles
DMCA / Removal Request
If you are the original writer of this dissertation and no longer wish to have your work published on the UKDiss.com website then please:




