Global Challenges Facing Antibiotic Resistance
Info: 7289 words (29 pages) Dissertation
Published: 16th Dec 2019
Tagged: Public Health
Antibiotic resistance is a current global crisis that needs to be controlled. Within this article, the issues of overuse and misuse within humans are looked in detail including how they are being prescribed drugs when their doctor is unsure if they have a bacterial or viral infection, and also the excessive use within the agriculture industry. As well as this the economic influences accelerating resistance and preventing the breakthrough of new antibiotics is used to show how low income countries are rapidly increasing their antibiotic use although high income countries are still consuming the most, but increase is much slower. The report then moves on to show how this is a global issue that everyone is trying to fight against, with schemes to prompt pharmaceutical industries to do more research into bacteria and new antibiotics and prevention plans against further resistance issues with world awareness weeks and more education needed.
I. INTRODUCTION
Antibiotics have been a huge discovery in the medicine industry by increasing life expectancies by up to 8 years between 1945- 1972. They are now the most common drug to be given out to patients,1 as they aim to either kill(bactericidal) or inhibit bacterial infections from spreading(bacteriostatic).2,3 They work in several ways and the type of antibiotic depends on the bacteria being gram positive (one cell membrane) or gram negative (two cell membranes).4
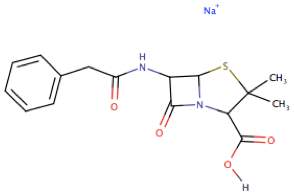
The first antibiotic to be discovered was Penicillin G (figure 1) back in 1928, by Alexander Fleming a Professor of Bacteriology at St Mary’s Hospital, London. He made the discovery after spotting that a petri dish of Staphylococcus (bacteria causing sore throats and abscesses) had an area of mould where directly surrounding it was clear of bacteria, showing that the mould secreted something capable of inhibiting bacterial growth. Although discovery was by Flemming, it was lecturers at the Sir William Dunn School of Pathology, University of Oxford who purified it for human consumption. They began their work on the purification of penicillin in 1939, whereby they grew mould in large amounts, then Norman Heatley, a biochemist, used the filtrate to extract penicillin using amyl acetate then back into water. The purification was continued by Edward Abraham, who used alumina column chromatography to remove any impurities. Penicillin’s use as an antibiotic was first proved by Florey in 1940 by experiments on mice against Streptococci; a bacterium that causes sore throats and fevers.5 The first human use was in 1941 by a policeman who had a cut on his mouth that become infected forming abscesses. He was given the penicillin by injection, and within in a few days his condition had improved, but the supply of the drug run out, and so his condition declined again and he unfortunately died. This showed the need for more penicillin, but the UK could not meet this due to its chemical industry being solely for the war. So Florey went to the Northern Regional Research Laboratory (NRRL), USA , where they produced a method that used- lactose in- stead of sucrose as the culture medium, corn-steep liquor and a penicillin precursor in the fermentation medium and grew the mould in submerged culture rather than just on the surface nutrient medium and used a strain of penicillium found on a mouldy cantaloupe melon exposed to UV rays. This alternative method lead to higher yields of the drug.6
Figure 1. The structure of the first Antibiotic discovered- Penicillin
II. ANTIBIOTICS AND THEIR MODE OF ACTION
Antibiotics can be classed together in several ways, with the most common being due to them having the same; core structure, mode of action or range of activity. This section will focus on the five main modes of action; Inhibition of cell wall synthesis, Breakdown of Cell membrane structure or function, Inhibition of nucleic acids, inhibition of protein synthesis and blockage of key metabolic pathways.
Inhibition of cell wall synthesis is usually undertaken by inhibiting the synthesis of Peptidoglycan (PG), by drugs binding PG units, or blocking Transglycosylase and Transpeptidase (Penicillin-binding proteins (PBPs)). β- lactams (Penicillins, Carbapenems etc), all contain a ring of three carbon’s and a Nitrogen (figure 2), and use this method. They interfere with essential proteins for the
synthesis of the cell wall, which either kills or inhibits the growth, some bind to PBPs (responsible for cross- linking proteins when synthesising PG) which interferes with the synthesis of the PG, leading to cell death.
Figure 2. The structure of the β-Lactam Ring
Protein synthesis is an essential process in all living cells, and it depends on information contained within the Deoxyribonucleic Acid (DNA), so any disruption to the process of will affect a cell majorly. There are two sub- classes; 50S ribosome inhibitors and 30S inhibitors. 50s block the initiation or elongation phase of protein synthesis. Examples are Oxazolidinones, a recent group of synthetic antibiotics, with the first being Linezolid in 2000. Their mode of action is still not fully understood, but it is believed that they bind to site P of the 50S inhibiting protein synthesis. Another example are macrolides, which contain either a 14,15 or 16 membered macrocyclic lactose rings with D-desosamine and L-cladinose (deoxy sugars) attached (figure 3). They bind to ribosomes stop-ping the addition of an amino acid to a polypeptide chain. 30s block aminoacyl-tRNAs access to ribosomes.
tivity affects RNA polymerase which prevents RNA syn- thesis in just the bacteria.
Some antibiotics copy the structure of a substrate needed for metabolism, which results in the bacteria enzyme binding to the drug instead of the normal substrate. An example is sulphonamides (figure 4), the first group of antibiotics to be used for therapeutic medicine. They mimic tetrahydrofolate required for folic acid synthesis, which is vital for nucleic and amino acid metabolism.
Figure 4. The structure of sulphonamides
The breakdown of the cell wall depends on each individual bacterium, due to them containing different cell lipids. One example is Daptomycin which ends synthesis of macromolecules by depolarising calcium-dependent membranes, resulting in disintegration of the cell membrane as the polymyxins bind to the lipid part of the lipopolysaccharide.3
III. HOW AND WHY RESISTANCE HAS COME ABOUT
There are a range of antibiotics, but resistance has become a growing issue. Resistance of a drug is defined as being when a bacterium can grow in the presence of the antibiotic which would usually kill/prevent the growth of it.7 This means that infections that use to be easily treated no longer can, and resistance can be passed on.
Alexander Fleming warned us of this era of antibiotic resistance in 1945 in his Nobel Lecture saying: ”The time may come when penicillin can be bought by anyone in the shops. Then there is the danger that the ignorant man may easily under dose himself and by exposing his microbes to non-lethal quantities of the drug make them resistant.”8
Bacteria become resistant due to mutating or Horizontal Gene Transfer (HGT), with HGT’s being said to be the main cause of this current crisis. Antibiotics will stop the bacteria it is targeting from reproducing, leaving the resistant strains of bacteria to multiply and pass on their resistance. These resistant bacteria are the disease caus- ing, and so it can no longer be treated with the antibiotic. HGT is how resistance can be passed between different types of bacteria, either by conjugation, transformation, transduction or gene transfer agents(GTAs) (Figure 5).
2
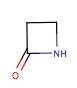
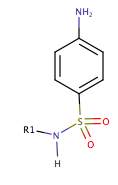
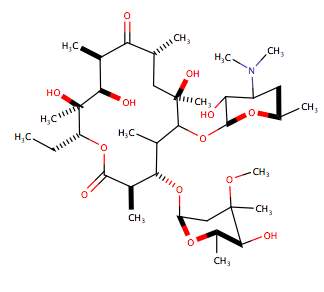
Figure 3. The structure of Macrolides
Inhibition of nucleic acid synthesis is achieved by blocking replication or preventing transcription. Quinolones interfere with the helicase enzymes function, preventing it doing its job of unwinding DNA for repairing and replication. Antibiotics of this type also target Topoisomerase 2 and 4, as disrupting these enzymes ac-
3
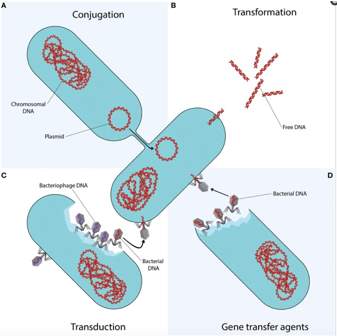
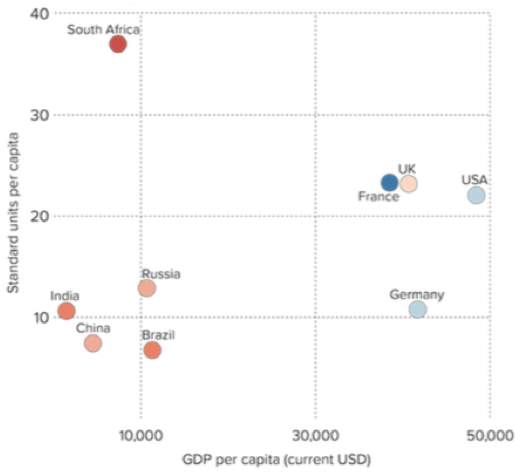
Figure 5. This shows the four different ways that horizontal gene transfer can occur by; a) conjugation, b)transformation, c)transduction, d)Gene Transfer Agents (GTAs) 9
Conjugation is the most likely due to its protection from the surroundings, and as its way of entering the host cell is more efficient. It occurs when DNA is transferred through a multi-step process but requires cell to cell contact such as plasmids, containing genes or elements in the chromosome. Once the resistant genes are on the plasmids, they can then spread very quickly to different strains, species and even genera, which has been shown in resistant quinolones and sulphonamides. Transformation is when bacteria can uptake and integrate naked fragments of extracellular DNA. There are specific requirements that need to be met for transformation to be able to occur, including; stabilised extracellular DNA and competent receiving bacteria, of which competence has seen to be stimulated when the bacteria are exposed to antibiotics. Transduction uses bacteriophages to transfer DNA between bacterium’s. Then finally there is GTA’s, which are particles produced by the host-cell (similar structure to bacteriophage). The GTAs carry small amounts of genome of the producing cell which is not enough to reproduce by its self. It enters the cell by a combination of transduction and transformation and released by cell lysis.9
The resistance of these drugs has been hugely enhanced due to the misuse and overuse of them.10 As well as this, the diseases can be more easily transmitted from one to another due to the increase in travel and globalisation.
The concept of misuse and overuse has been seen glob- ally, with an increase in consumption of around 30 per- cent between 2000-2010. The largest increase was seen in LMICS (Low and middle income countries), although HICs (high income countries) are still consuming the most (figure 6).11
Figure 7, shows how GP’s are majorly to blame for misuse and overuse, as most the time they are prescribing
Figure 6. The percentage increase between 2000-2010 in the use of antibiotics in different countries 11
antibiotics not knowing if they should be or because they cannot handle demanding patients. So, if GP’s were to be able to correctly prescribe them without pressures from their patient, then resistance may not have become such of a big problem within countries.
Figure 7. How surveyed GPs in 2014 reasoned why they pre- scribed antibiotics 12
A. Resistance from the agriculture industry
The agriculture industry uses a massive 130,000 tonnes of antibiotics annually on animals13 of which humans then go on to consume, this figure is higher than the consumption of humans, this shows just how much the industry is enhancing antibiotic resistance.
Antibiotics are used in animals to increase their growth rate on a smaller amount of food and to remove any chance of infection in their overcrowded homes. This causes resistance due to a regular low dose of antibiotics being used daily on healthy animal stocks. The animals can then pass on their resistant bacteria to humans by people directly touching them, eating or drinking pro- duce from them, and by touching surfaces that have had uncooked meat on.14
An example to show that livestock bacteria can be

passed onto humans, was shown between the years of 2005-2006, when the government of Canada made Quebec chicken hatcheries stop using the ceftiofur (antibiotic against respiratory diseases that inhibits the synthesis of cell walls15) injection on their eggs. After this withdrawal, it was seen that salmonella and E-Coli in retail chicken products fell and also resulted in a sharp decline in Salmonella infection in humans.16
IV. ECONOMIC CHALLENGES OF ANTIBIOTIC RESISTANCE
The economy of each country has an influence in the rate at which resistance is happening but economically it can be seen how much of a burden resistance is on the economy of a country, especially in countries that have a free healthcare system, as it is increasing the need for more funds, but more on this shall be explained in this section.
In the last decade, travel has become very affordable for many populations, especially to more exotic countries, which is increasing resistance to more strains of bacteria, as they can be more easily moved from country to coun- try, as well as tourism, there is also the increase of trade between countries.17
People becoming increasingly resistant, will cost them- selves or their national health scheme (NHS) millions to be treated for the infection, as they will require longer time in hospital, more scans/tests, more sick leave from work and more medication to try and treat them. The impact of an outbreak can have on the economics of a hospital is massive, because as part of infection control, they will have to close the affected unit and further to this surgery may have to be cancelled due to the risk of infection after the procedure. An example of costs in the US, where it is anticipated that a patient that has a resistant infection will need US $10,000 to 40,000 extra in healthcare costs,18 but the worrying fact is that it is expected that the cumulative cost of antibiotic Resistance will reach 100 trillion US $ by 2050.19 There was also an example in Manchester, UK where it showed how much of a burden on the NHS antibiotic resistance is, when a single super-bug outbreak cost the hospital 8.4million pounds!17 The EU predicted in 2010 that due to infections resistant to multiple antibiotics, they would require an extra 1.5 billion euros per year for healthcare,20 and the world bank has estimated the cost globally will be around 1 trillion dollars every year after 2030!17
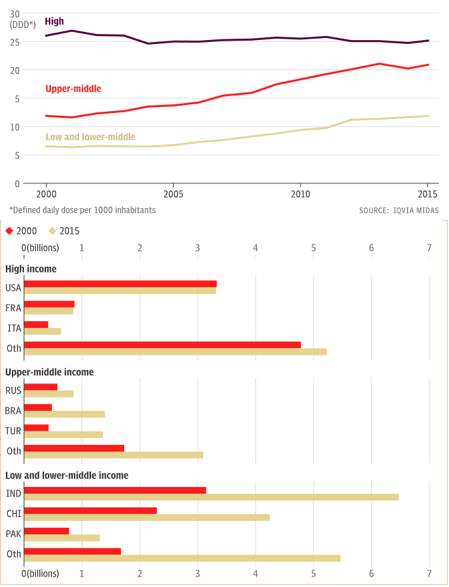
The graph’s in figure 8 show how, HICs are consuming the highest number of antibiotics, but there has not been much variation between the years of 2000 and 2015. But in countries that are still developing consumption has rocketed over the fifteen years, especially between 2007- 2015. There have been huge worries over countries such as India, as they are being targeted by pharmaceutical companies to increase their profits, due to the population of India being able to still buy the drugs over the counter without a prescription, this boosts the sales, even though the drug has not been approved by the Indian National Medicines Regulator.21
Figure 8. The two graphs show how the income of a country is influencing antibiotic consumption 21
In order for new antibiotics to be found, more research is required and this could cost a pharmaceutical company up to $2.5 billion to bring just a single drug into the market,22 this would then lead to the new drugs being more expensive than the old, as they will be under patent meaning only one company can manufacture them for several years. However, most companies are not willing to be involved with looking for new antibiotics, due to there being no profit within this sector as sales will be restricted because of this fear of resistance.17
V. HOW RESISTANCE VARIES IN HUMANS
In the previous section’s, it was shown how antibiotic’s work, how resistance has come about due to four types of HGT and why resistance has spread- mainly looking at how they are misused and overused due to agriculture’s excessive use and due to economics of each country, but now this section will look at how humans are affected differently due to these factors mentioned previously.
Humans are being majorly affected by the resistance of bacteria, as infections are not being able to be treated and after operations, they are more at risk if they do get an infection. In estimates made by the EU (2010) over 25,000 patients died from infections that have five different resistant bacteria a year,20 and it has been said
4
mortality rate will increase by the tenfold by 2050 which is just of deaths caused by resistant bacteria.23 In the UK alone, it was estimated by the government in 2016 that 5,000 people a year die from superbugs, however the bureau said that the true value is at least double this, which was backed up by NHS statistics- with 12,000 people dying from drug-resistant blood poisoning infections in the UK each year.24 USA’s Centre for Disease Control and Prevention (CDC) estimated that there are 2 mil- lion patients suffering from antibiotic-resistant infections annually, and that 23,000 of these patients end up dying from the infection.22
In the LMICs, there is greater harm to be done, as they have poorer health care systems, and more bacterial infectious diseases, and statistics showed that 70% of neonatal infections within hospitals could not be treated with the suggested treatment path by the World Health Organisation (WHO) due to resistance.20 Globally, it is predicted that by 2050, there will be 10 million deaths due to resistant infections.22
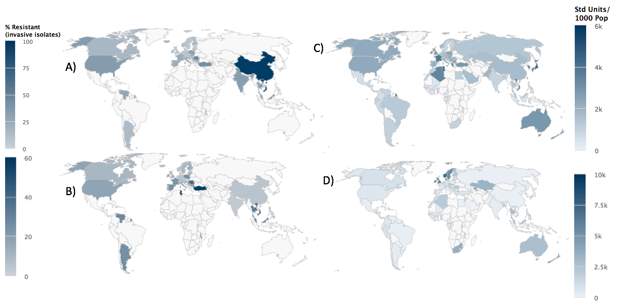
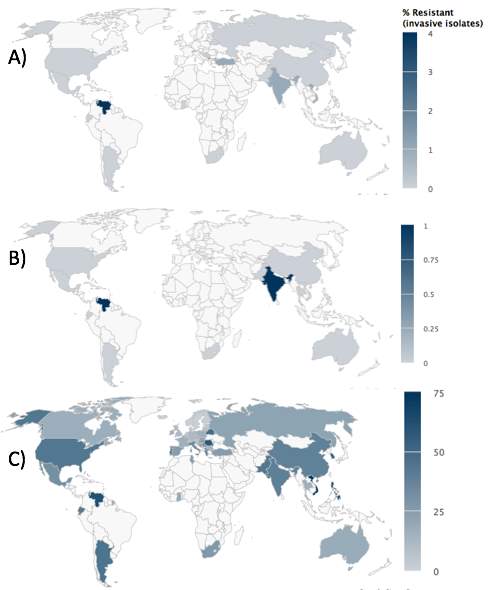
Looking at Streptococcus pneumoniae bacteria, which can cause meningitis and pneumonia, of which infections are highest in under two years and over sixty-five years. In 2011, there were 1.3 million deaths worldwide due to pneumonia, and one third of these were in children aged under five years old. This type of bacteria use to be treated with antibiotics such as penicillin, but since the 1960s, resistance has increased to this antibiotic, so we moved on to use macrolides, fluoroquinolones (FQs) and non-beta-lactams. Although resistance is most common in children, FQs resistance is more common in adults, as they are not as commonly used to treat children.25 Looking at figure 9, you can see that China is the country most resistant to Macrolides and Turkey and Tunisia are most resistant to Penicillin. Comparing this to the maps of use, you can see Penicillin use is much less than Macrolides, this is due to resistance to the drug being earlier than macrolides as they are an older drug. In countries with high resistance of Macrolides, it is due to the bacteria carrying both erm(b) and mef(A and B) genes rather than just erm(b) and mef(A or B) like in USA.25 In HICs which have a vaccine for Streptococcus pneumoniae, there is lower chance of infection, which can be seen in the maps.
Figure 9. The maps on the left show the resistance of A) Macrolides and B)Penicillin for treatment of Streptococcus pneumoniae, and the maps on the right show antibiotic use C) Macrolides and D) Penicillin 26,27
Moving on to Enterococci, gram positive bacteria, with Enterococcus faecalis (63%) and Enterococcus faecium(20%) being the most common, causing urinary tract and bacteraemia infections.
E.faecium are very well known for their resistance to ampicillin and high levels of vancomycin. Vancomycin Resistant Enterococci (VRE) are most common in hospitals, with the risk of colonisation being increased due to surgery and dialysis, of which it is estimated to then take 26 weeks to get rid of the colonisation. There are five different types of Vancomycin resistance; VanA is mostly in the US, Europe and Korea, VanB in Singapore and Australia. VanD has low level vancomycin resistance and has the the same characteristics clinically and microbiologically to VanA, and so can convert to VanA if exposed to glycosides.
Looking at the maps in figure 10, there is more E.Faceium resistance compared to E.Faecalis, this is due to there being more cases in hospitals, so the spread of the infection is more rapid, therefore more treatment of that strain is required.
Figure 10. The top map shows Enterococcus faecium and the bottom shows Enterococcus faecalis resistance to Vancomycin
26,27
5
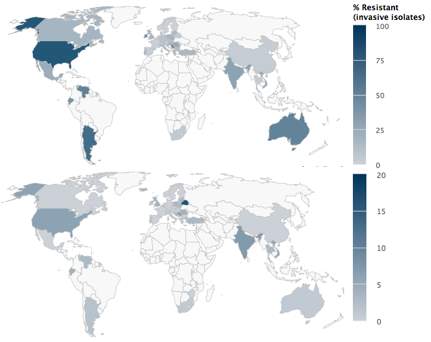
Staphylococci Aureus is one of the most well-known pathogens of which Methicillin (1958) and Vancomycin (1959) were approved for use against it. The first case of Methicillin-resistant S.aureus (MRSA), was in 1961, and as seen in figure 11C, it still has high levels of resistance,especially in Vietnam, The Philippines and Venezuela. Due to MRSA, vancomycin became the drug to use, until in 2002 when the first resistant strain of Vancomycin was obtained, known as Vancomycin-resistant S.Aureus (VRSA), but the resistant levels are very low (figure 11) with the highest levels being 1% in both India and Venezuela. Although it has the highest rate for mortality for infections, there was a 31% decrease in the number of invasive MRSA infections between 2005 to 2008. In 2000, Linezolid, the first oxazolidinone was approved for treatment of MRSA, which inhibits protein synthesis, but has lower cure rates compared to Vancomycin. The first resistance outbreak to Linezolid was reported in Madrid in 2008, where they found that the mechanism of resistance was plasmid-mediated-acquisition of chloram-phenicol/florfenicol resistance gene, although cases of resistance of Linezolid as seen in figure 11 are very low, with resistance highest in Venezuela.25 Venezuela seems to have the highest percentage of resistance for a lot of S.Auerus antibiotics, this is thought to be due to the poor healthcare systems they have.
Figure 11. the three maps show resistance of Staphylococcus aureus to A)Linezolid B) vancomycin C)Oxacillin (MRSA)
26,27
Next a gram- negative bacteria, Neisseria, with the infection causing being N.meningitidis and N.gonorrhoaeae, with others being found in the respiratory tract of pigs, cows etc. N.meningitidis is usually unharmful, unless it bypasses the nasopharyngeal
epithelium, of which then it causes serious infections such as sepsis and meningitidis. The same occurs with N.gonorrhoeae, which causes cervicitis (women) and urethritis (men).25
N.gonorrhoeae is a massive global problem with the WHO estimating there are 106 million new cases every year. It is now resistant to all classes of antibiotic, even to third-generation drugs such as Cephalosporin. Gonococcal infections can be easily transmitted through sexual intercourse due to it infecting mucosal surfaces. There has been evidence to show that gonococcal infections are highly more likely if someone has Human Immunodeficiency Virus (HIV). The number of cases of this infection is extremely high in developing nations such as Africa and Asia, due to their lack of diagnostic skills and less treatment options available. There is said to be a 13.5- fold high incidence rate for 15-49year olds in the Western Pacific compared to the Eastern Mediterranean in 2008. There are many reasons for this big difference, from the number of antibiotics available to them, to the beliefs in sexual practices, with the most influencing being the socioeconomic conditions of the country’s population. Although the UK has a low rate of the infections (63/100,000 people), certain locations such as London the capital city has a rate of 634/100,000 people, where the black ethnicity is four times as likely to be associated with the infection compared to the white ethnicity. This is similar to the USA, where the highest rates are in the African-American population 460/100,000 compared to the overall incidence of the USA being 107.5/100,000. Research has shown that metropolitan areas in HICs and rural areas in LMICs are most at risk due to health re- sources being sparser or overworked.25
The last few case studies, have shown how geographical, economical, and health care are all factors which affect the rate of resistance to antibiotics.
Not only does it affect people who are resistant to antibiotics but it also affects those who are not, be- cause they are then reduced to the prescription of antibiotics due to antibiotics coming off the medicinal market when resistance of the populations reaches a certain high point.18
VI. LOOKING TO THE FUTURE OF NEW ANTIBIOTICS
There has been a serious downfall in the number of new antibiotics being discovered by scientists in recent years, a huge 90% downfall was seen between 1980s and the early 2000’s,22 as there seemed no real demand for new ones, and as mentioned in Section 4 there is still not a market value for pharmaceutical companies to invest into the research for new ones.4 Over the past thirty years only two new classes have appeared (Oxazolidinones and cyclic lipopeptides), of which both are not active against gram-negative bacteria.20 But as the issue of resistance is getting worse, companies are investing into the research
6
of new antibiotic drugs, although they are finding it a lot harder to find antibiotics against gram negative com- pared to gram positive due to the drug needing to get through two cell membranes to be effective.4
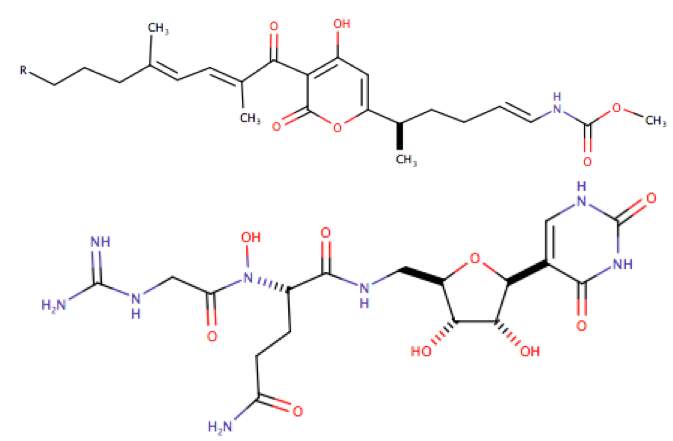
The most recent discovery has been the Malacidins (metagenomics acidic lipopeptide antibiotic-cidins), an antibiotic found in soil microbiomes, they are found to be able to treat MRSA. Malacidins are 10-membered cyclic lipopeptides and each one is only different by a methylene at the terminus branch on their lipid tails and their core is made up of four non-proteinogenic amino acids (figure 12). This class of antibiotics have been seen to be dependent on calcium by experimenting at different calcium concentrations on MRSA, which clearly showed the activity of this antibiotic is dependent on calcium being present, other cations were also used but did not give a good outcome of antibacterial activity.
Figure 12. The structures of Malacidin A (R1 is Me) and B)(R1 is Et)
Malacidin was seen to be a cell wall intermediate binding antibiotic, that was targeting for Lipid – II of undecaprenyl-N-acetylmuramic acid-pentapeptide (UDP-MurNAc-pentapeptide) of the cell wall.23
Pharmaceutical company Entasis Therapeutics, has taken a different approach to drug resistance, and have been working on recreating a common antibiotic called cefpodoxime(figure 13). Cefpodoxime was a β-lactam antibiotic used for severe infections in the urinary and gas- trointestinal (GI) tracts, but resistance came around due to bacteria evolving to produce β-lactamase enzymes, which break down the structure of the antibiotics to stop its antibiotic properties. So Entasis are creating an intravenous complex called ETX1317 that will bind to β- lactamase to inhibit it, this will then allow β-lactam antibiotics to work as usual. They have also produced an oral alternative called ETX0282. Both these drugs have been shown to work against gram negative resistant bacteria such as Klebsielle Pneumonia. They are hoping to start trials with these two drugs next year.4
Figure 13. The structure of Cefpodoxime
Microbiologist Kim Lewis from University of Boston is also approaching the development of new antibiotics in a different way, by producing a Teixobactin (figure 14) which works from the outside of the bacteria rather than the inside, preventing ejection (bacteria get rid of unwanted molecules from inside). Teixobactin attaches to peptidoglycan and teichoic acid (building blocks of the cell wall), inhibiting synthesis of the cell wall. Lewis also says that the drug will not become resistant due to it binding to two proteins produced by reactions by en- zymes rather than by the DNA of the bacteria itself, so the bacteria will be less likely to be able to mutate as a lot will need to be changed, and if mutations did occur that could cause resistance, it would also affect the cell function due to it affecting the cell wall, causing death to the bacteria. Tests have shown that Teixobactin is effective against MRSA and streptococcus pneumoniae. It is now going under tests for The US Food and Drug Ad- ministration (FDA) approval for human clinical trials.4
Figure 14. The structure of Teixobactin
Richard Ebright, has been studying the structure of RNA polymerase(RNAP), an enzyme which converts DNA into RNA (transcription) in bacteria for the last two decades, and has found two new potential antibiotic molecules. One being Myxopyronin(figure 15- top), which targets the hinge region of RNAP, this region enables RNAP to open up to allow DNA in to translate to RNA. But taking Myxopyronin would stop this process from happening so that translation does not happen. Currently, the drug has been showed to be effective in mice. The other is Pseudouridimycin (figure 15- bottom) which looks like uridine-5-triphosphate (UTP), a building block for the synthesis of RNA, therefore UTP cannot bind to RNAP stopping RNA synthesis. The drug has shown to be effective against Streptococcus pyogenes in mice. These two drugs are less likely to become resistant, as if the bacteria were to mutate in these two regions, the activity of RNAP would completely stop. Although Ebright is not very optimistic of this saying that ”bacteria will always find a way”.4,28
Figure 15. The structure’s of Myxopyronin (top) and Pseu- douridimycin(bottom)
Locus Biosciences in North Carolina, have found an- other new way to overcome antibiotic resistance, and their process looks at using the bacteria’s immune system- CRISPR. The researchers are adding DNA that matches the sequences in the bacteria into the phage. This allows the virus to infect the bacteria, and insert its DNA into the nucleus, but when the DNA is transcribed, the new RNA will guide the CRISPR cutting and de- grading enzyme (CAS3) resulting in DNA that cannot be repaired. There is now hope for the start of clinical trials in 2019, after being effective against C.difficile in mice.4
VII. WAYS THAT RESISTANCE CAN BE PREVENTED
There are many ways global organisations are trying to help us get to a point where resistance is no longer a problem.
To reduce animal resistant bacteria being passed on, the EU (2006) and Denmark have both restricted the use of antibiotics as use as a growth enhancer in animal food and water. The FDA have not been as promising in reducing antibiotic use in live feed, but in 2016, they
did put in place documentation that mentioned that antibiotics could not be sold as a growth enhancer, but this means that similar alternatives can still be added to animal feed.14
A way of prevention was shown in French hospitals between 1993 and 2007. They showed how strict hygiene measures would reduce the spread of MRSA. They ensured continuous training of their staff and that the staff were fully engaged in sharing their opinions. It was shown that there was a 35% decrease in MRSA rates between the two-time periods.20 This is an easy way to prevent spread, if we all take this advice and always wash our hands when visiting a hospital, after going to the toilet, and before and after handling/eating food.
In the US, there have been recent public-private partnerships, which they hope will help increase the interest of antibiotic research and innovation. The two main programmes by the government are National Institute of Allergy and Infectious Diseases (NIAID) and Biomedical Advanced Research and Development Authority (BARDA). For 2016, there was 605 million dollars placed for these two programmes (413 million to NI- AID and 192 million to BARDA). NIAID works on the early stages of research, submitting new drug submissions and looking at the new ways of clinical trialling new antibiotics. The NIAID also created a group called Antibacterial Resistance Leadership Group (ARLG) in 2013 which is for existing antibiotics and how to decrease resistance. The goal of NIAID is to produce two new antibiotics for gram-negative infections to then be passed on to BARDA. BARDA puts its funds to clinical trials, manufacturing and regulatory costs, there five-year aim is submission of two or more drugs for FDA approval.
Globally there is a private-public partnership called Carb-X (Combating Antibiotic-resistant Bacteria Xccelerating global antibacterial innovation) which begun in 2016, it includes; BARDA, NIAID, AMR Centre (UK) and Wellcome trust (UK), this partnership aims to pro- vide better funding for drugs for preclinical research and development. The total funding from now until 2021 is currently $350 million.
New Drugs for Bad Bugs (ND4BB) is a collaboration between the EU and European pharmaceutical industries. The total funding is estimated to be e700 million, which is only used for clinical trial expenses.22
The WHO has also dedicated a week in November every year since 2015 to bringing the awareness of antibiotics to the public. They have a campaign motto of ”Antibiotics: Handle with Care”. They have activities taking place during the week to get involved in and aim to inform the public of what is happening and what can happen if action is not undertook.29 In 2017, the theme was ”seek advice from a qualified health care professional before taking antibiotics”, and they promoted this with a variety of posters (figure 16)30
8
9

Figure 16. The posters used by the WHO to get people to see their doctors for advice before taking antibiotics 30
This moves us onto how GP’s as mentioned previously, are prescribing antibiotics the most the time when they should not be, which is making patients more likely to harbour resistant bacteria. It has been suggested to GP’s that they should be educating their patients on why they are prescribing the antibiotic or why they are not, and also ensure they are aware of why they should stop the antibiotic when they are being advised to, so they are more aware of antibiotic resistance. They are also suggesting that GPs should advice their patients on how they can prevent further infection from occurring.30 There is also the idea of Sequential Regimen which uses two or more drugs in an alternating pattern, allowing for maximum sensitivity but reducing cross-resistance, as it is thought to be harder to overcome dual antibiotics than just one. However, the problem with this is finding two drugs that work well together. Another way would be using proper doses, but it has been found hard to find the optimum dose for good treatment but minimise resistance, and the research into this uses in vivo experiments, which are expensive.31 There has also been a challenge set by the longitudinal Prize between 2014 and 2019 to get scientists to create a test which is quick, simple, affordable and will allow GP’s to diagnose if a patient has a bacterial or viral infection, so they can pre- scribe antibiotics appropriately, reducing misuse. With the winner getting £10million funding.32
As a population, preventing infection by having vaccinations, washing food and not cross contaminating foods, being hygiene aware to prevent the spread of infections achieved by simple things such as washing hands before eating, handling food, after using the toilet etc, preventing Sexually Transmitted Diseases by using protection is
also a massive prevention aid. But if a person does have an infection, the spread of it can be reduced by making sure they do not leave the house when they are seriously ill, using tissues for blowing the nose, going to a health professional to get a diagnosis, so the correct medication is prescribed, this will prevent the over-use or misuse of antibiotics, and if taking antibiotics, ensure to follow the instructions provided (e.g. when and how many to take), never take more than indicated as this increases the chance of resistance to that antibiotic, as it may not be needed.33
VIII. CONCLUSION
Having looked at how antibiotics work, how resistance comes around due to the four types of horizontal gene transfer (conjugation, transformation, transduction and GTAs) and how this process is being accelerated due to economics and misuse/overuse of antibiotics in both animals and humans, it can be seen that there is a major global crisis. If humans do not overcome this era of resistance, it could see everyone go back to pre-antibiotic times, where morbidity and mortality rates are increased and every day common medical procedures cannot take place due to the risk of infection with no cure. As a population in a crisis, action needs to be taken, regarding what is bought, and what can be prescribed. Stop buying food from companies that allow antibiotics to be used daily to livestock that are healthy. This will force farmers to stop using the antibiotics, as they will have reduced income, and need to listen to their consumers. The government should make this issue of antibiotics resistance clearer to the public, and should have education systems teaching their students on how medication should be taken, and promoting going to the doctors. As non-medical people, there should not be any self-diagnosing, but instead be taking the advice from doctors. Saying this, doctors and other health professionals should be more educated on the major crisis of antibiotics and be told what precautions they can take, and how to educate their patients. It is not possible to rely on just the discovery of more antibiotics, because it can take up to 30years for a drug to be allowed to be prescribed, and there is not this time, as well as this people do not want to also become resistant to the new drugs that are to be discovered, as this will just cause this problem of resistance to become even worse in the future. Action needs to be taken now before it is too late!
ACKNOWLEDGMENTS
The author acknowledges Sweta Ladwa for the support in the process of the completion of this report.
- [1] https://microbiologysociety.org/education- outreach/antibiotics-unearthed/antibiotics-and- antibiotic-resistance/what-are-antibiotics-and-how- do-they-work.html(date accessed: 3/3/18)
- [2] https://www.nhs.uk/conditions/antibiotics/, last re- viewed: 09/06/2016, (date accessed: 3/3/18)
[18] N.D.Friedman, The negative Impact of Antibiotic Resis-tance, European Society of Clinical Microbiology and Infectious Diseases, Elsevier Ltd, 2015
[19] Y. Ortiz-Martz, The Journal of Hospital Infection, World Antibiotic Awareness Week 2017 and its influence on digital information seeking on antibiotic resistance,
[3] Ebimieowei Etebu, Ibemologi Arikekpar, International 2018
Journal of Applied Microbiology and biotechnology Re- search, Antibiotics: Classification and mechanisms of action with emphasis on molecular perspectives, 2016, pages 90-101
- [4] Natasha Gilbert, Nature555, Four stories of antibacterial breakthroughs, 2018, doi: 10.1038/d41586-018-02475-3
- [5] https://medlineplus.gov/streptococcalinfections.html (date accessed:3/3/18)
- [6] The Discovery and Development of Penicillin, National Historic Chemical Landmarks program of the American Chemical Society, 1999, Pages 2-5
- [7] https://www.niaid.nih.gov/research/what-drug- resistance, last reviewed: 2009
- [8] Fleming A. Penicillin. Nobel Lecture, December 11, 1945
- [9] Petra F.G. Wolffs, Frontiers in Microbiology, Dissemination of Antimicrobial Resistance in Microbial Ecosystems through Horizontal Gene Transfer, 2016, DOI:10.3389/fmicb.2016.00173
- [10] https://www.cdc.gov/antibiotic- use/community/about/antibiotic-resistance-faqs.html (date accessed:5/3/18)
- [11] The state of the worlds antibiotics 2015, Centre for Dis- ease Dynamics, economics and policy,Washington D.C, 2015, pages 8- 10
- [12] Dr Rosemary Leonard, Longitude Prize, IN THE BAL- ANCE: GPS, PATIENT CARE AND ANTIBIOTICS, 2014
- [13] Deborah Mackenzie, New Scientist, 2017, DOI:10.1126/science.aao1495
- [14] Michael J.Martin, Antibiotics Overuse in Animal Agri- culture: A call to Action for Health Care Providers, American Journal of Public Health, 2015
- [15] https://www.drugs.com/vet/ceftiofur-sodium-for- injection-can.html (date accessed:7/3/18)
- [16] Benjamin J Koch, Frontiers in Ecology and the Environ- ment, Food-animal production and the spread of antibi- otic resistance: the role of ecology, The Ecological Society of America, Volume 15, Issue 6, Pages 309-318, 2017
- [17] Madlen Davies, Callum Adams, Claire Newell, Tele- graph, The True cost of antibiotic resistance in Britain and around the world, 2018
[20] Hogberg,L.D, Trends in Pharmacological Sciences, The global need for effective antibiotics: challenges and recent advances, 2010, doi:10.1016/j.tips.2010.08.002
[21] Claire Newell, Callum Adams, Paul Nuki, Telegraph, Global Antibiotic consumption soars feeding spread of UK Superbugs, 2018
[22] H.Luepke, J.Suda, etl, Past, Present and Future of Antibacterial Economics: Increasing Bacterial Resistance, Limited Antibiotic Pipeline and Societal Implications, Pharmacotherapy, 37: 71?84, 2017
[23] Bradley M. Hover, etl, Nature Biology, 2018, DOI: 10.1038/s41564-018-0110-1
[24] Madlen Davies,The Bureau of Invesitgative Journalism, Superbugs killing twice as many people as government says, 2016
[25] Douglas l. Mayers, etl, Antimicrobial Drug Resistance: Clinical and Epidemiological Aspects, Springer, 2017, Volume 2, pages: 777,778, 781, 811, 812, 819,820, 822, 843, 844
[26] https://resistancemap.cddep.org/AntibioticUse.php (Date Accessed: 09/03/18)
[27] https://resistancemap.cddep.org/AntibioticResistance.php (date accessed: 09/03/18)
[28] Mathieu F. Chellat, Rainer Riedl, Angewandte Chemie International Edition, Pseudouridimycin: The first nucleoside analogue that selectively inhibits bacterial RNA polymerase, 2017, pages 13184 ? 13186, DOI: 10.1002/anie.201708133
[29] http://www.who.int/mediacentre/factsheets/antibiotic- resistance/en/ (date accessed:7/3/18)
[30] https://www.health.govt.nz/our-work/diseases-and- conditions/antimicrobial-resistance/world-antibiotic- awareness-week-2017, last updated: 17/11/17
[31] Lauren A. Richardson, PLoS Biol, Understand- ing and Overcoming Antibiotic Resistance, 2017, doi:10.1371/journal.pbio.2003775
[32] https://longitudeprize.org/challenge [33] https://www.canada.ca/en/public-
health/services/antibiotic-antimicrobial- resistance/prevention-antibiotic-resistance.htmla2 date modified : 23/02/2018 (date accessed: 8/03/2018)
10
Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allRelated Content
All TagsContent relating to: "Public Health"
Public health concerns the wellbeing and health of the general public as a whole. Through organised efforts, public health bodies look to inform the choices of society to help protect from threats to their health and prolong and improve the quality of life.
Related Articles
DMCA / Removal Request
If you are the original writer of this dissertation and no longer wish to have your work published on the UKDiss.com website then please:




