Effects of High Intensity Interval Training (HIIT) vs. Moderate Intensity Continuous Training
Info: 8207 words (33 pages) Dissertation
Published: 13th Dec 2019
Tagged: Fitness
The acute effects of high intensity interval training (HIIT) vs. moderate intensity continuous training on vascular health.
Abstract:
The aim of this study was to investigate the acute effects of high intensity interval training verses moderate intensity continuous training on vascular health on healthy patients. Six (4 male and 2 female) participants (age: 23 ± 2.13 years; height: 1.73 ± 17.11 cm; body mass: 76.53 ± 0.09 kg), free of injury and illness, volunteered to take participate in the study, which has received Ethics Committee Approval. Participants took part in two exercise conditions: high intensity interval training (HIIT) and moderate continuous exercise (CE), on a cycle ergometer, at similar caloric expenditure calculated using [kcal/minute=
METS*3.5*body weight(kg)200], within a two-week period. Vascular biomarkers (AIx; AIx(75); PWV) were measured and calculated at different intervals before and after both exercise conditions (baseline; 1-minute; 10-minute; 20-minutes). No significant interactions were found between condition and time for AIx (p=.278) AIx(75) (p=.653) and PWV (p=.289). Similarly, there were no significant differences between HIIT and CE conditions for AIx (p=.916) AIx(75) (p=.543) and PWV (p=.249). Additionally, there were no significant differences in time for AIx (p=.368), however, there were significant differences reported for AIx(75) (p=.004) and PWV (p=.020). ………….
Introduction:(944)
Arterial dysfunction has been shown to develop early in life, even in patients with modest risk of cardiovascular diseases (CVD) (Perk et al., 2012), as such it has become a substantial and growing problem within health care (Heidenreich et al., 2011; World Health Organization, 2011). Arterial stiffness (AS), congealing and loss of elasticity of the arterial wall (Fleenor & Berrones, 2015), is considered an important predictor of CVD (Baulmann, Weber & Mortensen, 2010; Baulmann, Herzberg & Störk, 2013; Nitzsche et al., 2016). It generates a larger overall demand on the heart due to an increase of pulse pressure and stress on the walls (Belz, 1995; Kass, Saeki, Tunin & Recchia, 1996; Burr, Beck & Durocher, 2017), and so, is linked with arteriosclerosis and condensed coronary artery perfusion (Kass et al., 1996; Burr et al., 2017). Therefore, physical activity is strongly advocated for treatment and prevention for many chronic conditions and is allied with low risk of CVD and mortality (Tanasescu et al., 2002; Gregg et al., 2003; Lee, Sesso, Oguma & Paffenbarger, 2003; Rognmo, Hetland, Helgerud, Hoff & Slørdahl, 2004; Warburton, Nicol & Bredin, 2006a; Warburton, Nicol & Bredin, 2006b; Burr, Rowan, Jamnik & Riddell, 2010; Soares & de Sousa, 2013; Montero, 2015).
There are many cardiovascular health benefits to physical activity (Blair et al., 1989; Cameron & Dart, 1994; Ketelhut, Franz & Scholze, 2004; Lavie et al., 2015), including, improvements in vascular (Hambrecht et al., 2000; Linke et al., 2001) and cardiac function (Haykowsky et al., 2007), physical fitness (Tol, Huijsmans, Kroon, Schothorst, Kwakkel, 2006), decrease in AS (Hayashi, Sugawara, Komine, Maeda & Yokoi, 2005) and counterbalance of the expected age-related stiffening (Tanaka, DeSouza & Seals, 1998). Continuous exercise (CE) is the mode of physical activity generally endorsed to CVD patients (American College of Sports Medicine, 2004). However, epidemiological and clinical research have demonstrated that the beneficial effects of physical activity are dependent on the quality and volume of effort produced during exercise (Rognmo et al., 2004; Schnohr, O’Keefe, Marott, Lange & Jensen, 2015; Tschentscher et al., 2016). As such, high intensity interval training (HIIT) has been suggested to have a better effect on vascular health in the healthy population (Fox et al., 1975; Wenger & Macnab, 1975; Shiraev & Barclay, 2012) and those with CVD (Rognmo et al., 2004).
For gains in health, patients at risk of CVD are recommended to exercise habitually at intensities oscillating from 40-90% of
V̇O2peak (American College of Sports Medicine, 1994; Fletcher et al., 1996; Fletcher et al., 2013). Conversely, training programmes of CE are frequently completed at low-to-moderate intensities (40-60%
V̇O2peak) (Swain & Franklin, 2002; Rognmo et al., 2004; Ross, Porter & Durstine, 2016). For this reason, HIIT has grown in popularity (Burr et al., 2017), branded by its short bouts of vigorous exercise (10-seconds to 5-minutes) at an intensity higher than the anaerobic threshold (Laursen & Jenkins, 2002), intermingled by low-intensity exercise or rest (Gibala, Little, MacDonald & Hawley, 2012; Cochran et al., 2014). HIIT is proposed to be the superior mode of exercise over submaximal CE (Laursen & Jenkins, 2002) due to its validated capability to evoke considerable adaptations in cardiorespiratory fitness (Park, Rink & Wallace, 2006; Helgerud et al., 2007; Jones, Taylor, Lewis, George & Atkinson, 2009), while remaining time-saving (Rognmo et al., 2004; Ketelhut, Milatz, Heise & Ketelhut, 2016; Burr et al., 2017). However, this is not a unanimous verdict (Murtagh, Boreham, Nevill, Hare, & Murphy, 2005).
The vascular effects of exercise modalities can be enumerated using vascular biomarkers (Hanssen et al., 2015). One valid measure, which forecasts cardiovascular events in CVD patients and the general population (Vlachopoulos et al., 2010; Chirinos et al., 2012), is the augmentation index (AIx), an examination of the augmentation systolic blood pressure by reflection of the peripheral pulse wave (Hanssen et al., 2015). A low AIx shows reduced influence on the reflected wave on the central systolic blood pressure, demonstrating reduced AS (Hanssen et al., 2015). AIx links inversely with heart rate (HR) (Wilkinson et al., 2000). Due to fluctuations in HR between measures, a tweaked AIx, calculated at a HR of 75bmp, known as AIx(75) is used for a better contrast to rest (Hanssen et al., 2015). Another measure, carotid-femoral pulse wave velocity (PWV), is the gold standard marker of AS (Laurent et al., 2001; Boutouyrie et al., 2002; McEniery et al., 2005; Vlachopoulos, Aznaouridis & Stefanadis, 2010; Elias et al., 2011), which is a composite measure that is dependent on the location and degree of wave reflection (Safar & London, 2000; McEniery et al., 2005).
To date, research has demonstrated that HIIT training increases
V̇O2max (Helgerud et al., 2007), reduces total body mass (Perry, Heigenhauser, Bonen & Spriet, 2008; Tjønna et al., 2008) and subcutaneous fat (Boutcher, 2010), as well as insulin sensitivity (Trapp, Chisholm, Freund & Boutcher, 2008) compared to CE. Similarly, past research has explored the effects of ageing on AIx and PWV (Avolio et al., 1985; Smulyan, Asmar, Rudnicki, London & Safar, 2001; Mitchell et al., 2004). However, minimal research has been conducted on the acute effects of different exercise modalities on AS through AIx and PWV (Hanssen et al., 2015). Therefore, this study will endeavour to investigate the acute effects of HIIT versus moderate-intensity CE on vascular health. The hypotheses for this study are:
H0 – There will be no significant differences in AIx or AIx(75) after completing a HIIT versus moderate-intensity CE.
H1 – There will be significant differences in AIx or AIx(75) after completing a HIIT versus moderate-intensity CE.
H0 – There will be no significant differences in PWV after completing a HIIT versus moderate-intensity CE.
H2 – There will be significant differences in PWV after completing a HIIT versus moderate-intensity CE.
Methods:(882)
Participants: Six healthy participants, 4 male and 2 female (age: 23±2.13 years; height: 1.73±17.11 cm; body mass: 76.53±0.09 kg), free of injury and illness volunteered to take part within the study. All participant were actively taking part in vigorous sport and exercise through competing in university and club sports in addition to their own recreational time. Before any testing measures commenced all the participants were required to complete a Physical Activity Readiness Questionnaire (PAR-Q) alongside providing written consent. During this time, the participant’s resting blood pressure, height and weight were all recorded to ensure their suitability and safety for testing. The current study was approved by the University of Winchester Ethics Committee of the Department of Sport and Exercise.
Experimental Design: This study was a non-randomised cross-over design. To start the participants rested in a supine position for 15-minutes (Nitzsche et al., 2016; Milatz, Ketelhut & Ketelhut, 2017), after which, baseline pulse wave analysis (PWA) and carotid-femoral PWV were performed on the left-side using a SphygmoCor (AtCor Medical, SphygmoCor, Australia). Baseline measures were recorded, alongside a resting blood lactate. While supine the participant was fitted with a face mask, where 1-minute of
V̇O2 was collected using a cortex metalyzer (Metalyzer 3B, Germany). Post-rest, the participant was equipped with a HR monitor (Polar, T31c Transmitter, England) and the cycle ergometer (Monark, 874 E, Sweden) was set to the correct position and recorded. The participants took part in a 2-minute warm up at 70RPM before the test commenced.
The HIIT protocol consisted of six, 1-minute low-intensity stages at 70RPM, alternate with five, 10-seconds of strenuous cycling effort (Rakobowchuck et al., 2008; Lauren, 2010; Hazell, MacPherson, Gravelle & Lemon, 2010) performed against a resistance equal to 0.075kg (kg body mass)-1 (Rakobowchuk, Stuckey, Millar, Gurr & MacDonald, 2009). Each stage was timed via stopwatch (Digi Lap, DT100, UK) and the participants
V̇O2 was collected using the metalyzer throughout. The participants HR and RPE was recorded at the end of each high-intensity stint, while blood lactate was obtained after the first and fifth high-intensity exercise bout, plus immediately post-exercise. Post-exercise the participants return to a supine position where PWA and PWV measures were repeated on the left-side at 1-minute, 10-minute and 20-minutes post-exercise (Tordi, Mourot, Colin & Regnard, 2010). At the same time the participants blood lactate samples were analysed using a Biosen (C-Line, EKF Diagnostic, UK) and average peak power was recorded from all five 10-second high-intensity phases from the cycle ergometer test software (Monark, Anaerobic Test Software, Sweden).
The CE took place within two weeks of the HIIT, in between each participant’s CE time and cycling resistance was calculated to the same total energy expenditure (TEE) produced during their HIIT condition (Fletcher, Froelicher, Hartley, Haskell & Pollock, 1990; American College of Sports Medicine, 2013), thus, each participant exercised for different times, at different resistance. Pre-exercise PWA and PWV protocol was the same as the HIIT condition. Each participant’s exercise time was broken down into 5 equal sections, at the end of each section the participants HR and RPE was recorded, while blood lactate was obtained after the first and fifth sections, plus 1-minute post-exercise. Post-exercise PWA and PWV protocol was similary to the HIIT condition.
Data Analysis: To estimate the caloric expenditure from the HIIT exercise condition data, the equation [kcal/minute=
METS*3.5*body weight(kg)200], published by the American Heart Association (Fletcher et al., 1990) and highlighted in succeeding ACMS guidelines (American College of Sports Medicine, 2013), was used (Humphrey, 2006; MacArdle et al., 2010). For example, the primary step is to calculate average energy expenditure (AEE) during HIIT, to achieve this MET cost of exercise must be calculated by dividing overall average
V̇O2 by a MET [
2.7ml/kg/min3.5=7.86ml/kg/min]. This is then added to the original equation to achieve the participants kcal/minute [
7.86ml/kg/min*3.5*102.2kg200=14.6kcal/min], next this was multiplied by total work out time (6-minutes, 50-seconds) to attain total TEE [14.06kcal/min*6.83 = 96.03 kcal].
The next step follows the same format, though substituting overall average
V̇O2 with resting
V̇O2[
5.82ml/kg/min3.5=1.66kcal/min],followed by the equation [
1.66ml/kg/min*3.5*102.2kg200=2.97kcal/min], multiplied by total test time [2.97*6.83 = 20.29 kcal], to calculate energy expenditure at rest. AEE is then subtracted by expenditure at rest to calculate TEE during HIIT [96.03 kcal – 20.29 kcal = 75.74 kcal]. Thereafter, an equation on average high-intensity
V̇O2 [
41.8ml/kg/min3.5=11.94kcal/min]is calculated, followed by [
11.94ml/kg/min*3.5*102.2kg200=21.35kcal/min]. This value is then subtracted by expenditure at rest [21.35 kcal/min–2.97 kcal/min =18.38 kcal/min] and 15% (Fisher et al., 2015) of this value [
18.38kcal/min100*15=2.67kcal/min] is then divided by TEE [
75.74kal2.76kcal=27.44] to achieve total CE time of 27-minutes and 26-seconds. Finally, 15% of average peak power [
1140.49W100*15=171.07W] during HIIT (Fisher et al., 2015) was calculate to accomplish the intensity to be cycled at during CE.
Statistical Analysis: All data has been articulated as mean ± standard deviation (SD). All variables were put through a Shapiro-Wilk normality tests to determine normal distribution. Accordingly, a two-way repeated measure analysis of variance (ANOVA), was conducted for statistical evaluation with the factors ‘test’ and ‘time’ using SPSS (Version 22, SPSS Inc., Chicago). Significance was accepted at the level of p<0.05. Mauchly’s test of sphericity was completed to determine whether the assumption of sphericity was violated or not. Where significance was established, Bonferroni-corrected post-hoc analysis was undertaken as outlined by Vincent & Weir (2012). Effect sizes were calculated using the equation, 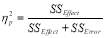 .
.
Results:(255)
AIx and AIx(75):
The means and SD of AIx and AIx(75) between the two exercise conditions are represented in figure 1 and table 2, respectively. A two-way repeated measures ANOVA conducted on the AIx data indicated there to be no significant interaction (F(3,15)=1.414, p=.278, 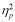 =.220) between condition and time. Similarly, there were no significant differences found between HITT and CE conditions (F(1,5)=.012, p=.916,
=.220) between condition and time. Similarly, there were no significant differences found between HITT and CE conditions (F(1,5)=.012, p=.916, 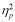 =.002) or testing times (F(3,15)=1.133, p=.368,
=.002) or testing times (F(3,15)=1.133, p=.368, 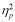 =.185).
=.185).
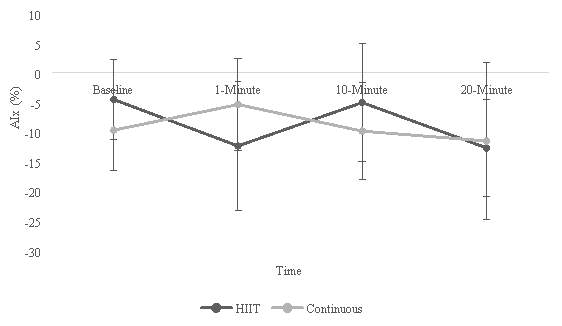
Figure 1: Comparison of AIx means and SD between HIIT and CE.
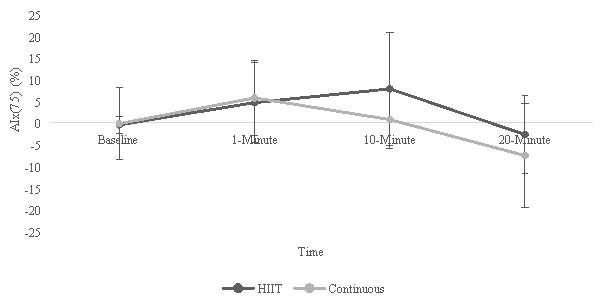
For AIx(75) the two-way repeated ANOVA showed there to be no statistically significant interactions (F(3,15) =.554, p=.653, 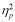 =.100) between the two exercise conditions and time. In terms of differences between HITT and CE conditions there was also no significance found (F(1,5)=.462, p=.543,
=.100) between the two exercise conditions and time. In terms of differences between HITT and CE conditions there was also no significance found (F(1,5)=.462, p=.543, 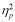 =.079). However, there were statistically significant differences seen in time (F(3,15)=6.791, p=.004,
=.079). However, there were statistically significant differences seen in time (F(3,15)=6.791, p=.004, 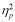 =.376). Bonferroni-corrected post-hoc analysis revealed there to be a significant difference (p=.007) in AIx(75) between baseline and 1-minute post-exercise as well as significance (p=.013) demonstrated between time 10-minutes and 20-minutes rost-exercise.
=.376). Bonferroni-corrected post-hoc analysis revealed there to be a significant difference (p=.007) in AIx(75) between baseline and 1-minute post-exercise as well as significance (p=.013) demonstrated between time 10-minutes and 20-minutes rost-exercise.
Figure 2: Comparison of AIx(75) means and SD between HIIT and CE.
PWV:
Figure 3 represents the means and SD of PWV between the HIIT and CE conditions. There were no statistically significant interactions seen between the two conditions and the times of testing (F(3,12)=1.407, p=.289, 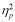 =.260). Likewise, the two-way repeated measures ANOVA reported there to be no statistically significant differences between HIIT and CE conditions (F(1,4)=1.814, p=.249,
=.260). Likewise, the two-way repeated measures ANOVA reported there to be no statistically significant differences between HIIT and CE conditions (F(1,4)=1.814, p=.249, 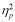 =.312). However, statistically significant differences were seen between times (F(3,12)=4.829, p=.020,
=.312). However, statistically significant differences were seen between times (F(3,12)=4.829, p=.020, 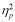 =.547), post-hoc testing using Bonferroni correction revealed an approach in significance (p=.086) between time 1-minute and 10-minutes post-exercise.
=.547), post-hoc testing using Bonferroni correction revealed an approach in significance (p=.086) between time 1-minute and 10-minutes post-exercise.
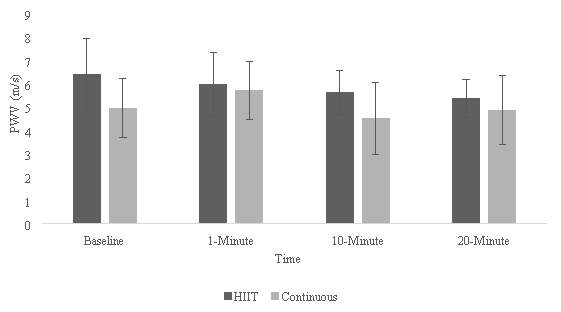
Figure 3: Comparison of PWV means and SD between HIIT and CE.
Discussion:(815)
AIx and AIx (75): (393)
The current study found there to be no significant interactions between condition and time in AIx and AIx(75), both of which presented with large effect sizes (Cohen, 1988; Cohen, 1992). It could be speculated that with larger statistical power through sample size these findings may have presented with significant results. In terms of condition there were no significant differences reported for AIx and AIx(75), however, only AIx(75) presented with a large effect size according to Cohen’s (1988) categories. Additionally, there were no significant differences in time for AIx, however, there was a statistically significant result exposed in time for AIx(75).
Though there was absence of significance, there are indications of acute decreases in pulse wave pressure post-exercise. This matches the findings of Heffernan et al. (2007) and Dischl et al. (2011) who reported significantly lower AIx with a delay of ~15-20 minutes’ post-exercise. They also discovered that AIx(75) was significantly enlarged directly post-exercise with a steady decline equal, or lower than baseline within 30 minutes (Heffernan et al., 2007; Dischl et al., 2011). This was concurrent with that seen in CE with a much lower drop compared to baseline in AS measured through AIx(75), however, HIIT AIx(75) peaked at 10-minutes and remained further increased during recovery (Heffernan et al., 2007). After extended comprehensive exercise, AIx(75) has been reported to be decreased within 15-minutes post-exercise (Vlachopoulo et al., 2010; Pressler et al., 2011). This insinuates that HR amended AIx(75) and AIx have comparable time courses during post-exercise recovery but with postponed reaction patterns, dependent on duration and intensity of exercise (Hanssen et al., 2015). As such, amending AIx for HR, the current study discovered roughly identical time courses for AIx(75) compared to AIx (Hanssen et al., 2015), especially for the CE condition.
The fundamental mechanisms of reduced pressure wave augmentation in recovery of post-exercise are multifaceted (Hanssen et al., 2015). Munir et al., (2008) discovered that a decrease in pressure wave reflection is partnered with increased blood flow and dilatation of muscular arteries. Moreover, enlarged quantities of nitro-glycerine infusion activated comparable alterations in AIx compared to an increase in exercise intensities (Munir et al., 2008). Thus, it could be suggested that a crucial mechanism underlying post-exercise decrease in pressure augmentation is nitric oxide stimulated dilatation of the muscular arteries through shear stress and augmented blood flow (Hanssen et al., 2015).
PWV: (357)
Statistical analysis revealed a non-significant interaction between condition and time when investigating PWV. Additionally, there were no significant differences between conditions, however, large effect sizes were presented for both results according to Cohen’s (1988) effect size categories, tallying the trend that significance could have been possible with an increased sample. Furthermore, there was a significant difference seen in time, thus, the main observation was that exercise, independent of the modalities, decrease PWV. These findings conflict those of Guimaraes et al. (2010) who demonstrated that AS was improved only during HIIT as calculated by a decrease in PWV, advocating that level of disease and difference in exercise intensity could be the answer to inconsistent results (Guimaraes et al., 2010).
This study, however, ran parallel to Ranadive et al. (2012) and Siasos et al. (2016) who discovered no significant differences in PWV relative to baseline measurements in either HIIT or CE conditions. Although, Kingwell et al., (1997) showed a decrease in PWV after a single session of moderate CE, within 30-minutes. Within this study PWV was measured up to 20-minutes post-exercise, as such, these contradictory results could be credited to the differing protocols (Siasos et al. 2016).
The mechanisms linked to AS are endothelial dysfunction, hyperplasia and hypertrophy of smooth muscle cells, abasement of the elastic matrix and upsurge in collagen content (Benetos et al., 2002; Zieman, Melenovsky & Kass, 2005). However, Peng, Haldar, Deshpande, Irani and Kass’s (2003) experimental study established that the degree of wall swelling stimulated by shear stress affects the alteration in A through mechano-biomechical signalling that can be detrimental to vasorelaxation. Consequently, increase in duration or intensity of exercise should be essential to help significantly improve AS in CVD patients (Guimaraes et al., 2010). Nevertheless, this was not evident in this investigation and could be explained by the sample group used. In healthy participants, with less stiffness of the arteries, a short duration of low intensity CE could be satisfactory to develop arterial function (Cameron & Dart, 1994; Tanaka et al., 2000), thus, could explain why no significant differences were seen between conditions but were seen in time for AS, as measured through PWV.
Limitations: (65)
As demonstrated by the effect sizes throughout this investigation (Cohen, 1988; Cohen, 1992) it could be propositioned that increase in size of sample could have resulted in statistically significant results for the variables measured in the current study. An additional limitation to this study comes from the protocol, it could be argued that true HIIT was not achieved, instead high volume interval training was attained…..
- Not fasted or hydration controlled for.
- Baseline not same AIx and PWV
Conclusion:(131)
In summary, the current study endeavoured to investigate the acute effects of HIIT versus moderate intensity CE on vascular health. Although there were no significance interactions or differences between conditions for AIx, AIx(75) and PWV, the results discovered support the notion that exercise, by-and-large, can help in the decrease AS. As such, the results presented speculate that with greater statistical power through sample size, HIIT has the potential to accomplish, although less noticeable, acute effects on AS. However, the negatives outweigh the positives
Based on the aforementioned, the two null hypotheses presented can be accepted.
H0 – There will be no significant differences in AIx or AIx(75) after completing a HIIT versus moderate intensity CE.
H0 – There will be no significant differences in PWV after completing a HIIT versus moderate intensity CE.
Word count: xxxx words. (3000) 2081 (941) 3022
278
Additional mechanisms speculated to be involved in the unbiased effects of exercise on central stiffening of the arteries are the lessening of endothelon-1 release and increased volumes of circulating catecholamine (Wray et al., 2007).
References:
American College of Sports Medicine. (1994). ACMS position on exercise for patients with coronary artery disease. Med Sci Sports Exerc, 26, i–v.
American College of Sports Medicine. (2004). ACSM position on exercise and hypertension. Med Sci Sports Exerc, 36, 533–553.
American College of Sports Medicine. (2013). ACSM’s guidelines for exercise testing and prescription. Baltimore: Lippincott Williams & Wilkins.
Avolio, A. P., Deng, F. Q., Li, W. Q., Luo, Y. F., Huang, Z. D., Xing, L. F., & O’rourke, M. F. (1985). Effects of aging on arterial distensibility in populations with high and low prevalence of hypertension: comparison between urban and rural communities in China. Circulation, 71(2), 202-210.
Baulmann, J., Weber, T., & Mortensen, K. (2010). Messmethoden der arteriellen Gefäßsteifigkeit. Journal für Hypertonie-Austrian Journal of Hypertension, 14(2), 18-24.
Baulmann, J., Herzberg, C. P., & Störk, T. (2013). Die Renaissance von Pulswellengeschwindigkeit, Augmentation und zentralem Aortendruck als Determinanten des kardiovaskulären Risikos. Die Medizinische Welt–aus der Wissenschaft in die Praxis, 64(1), 30-33.
Belz, G. G. (1995). Elastic properties and Windkessel function of the human aorta. Cardiovascular Drugs and Therapy, 9(1), 73-83.
Benetos, A., Adamopoulos, C., Bureau, J. M., Temmar, M., Labat, C., Bean, K., … & Safar, M. (2002). Determinants of accelerated progression of arterial stiffness in normotensive subjects and in treated hypertensive subjects over a 6-year period. Circulation, 105(10), 1202-1207.
Blair, S. N., Kohl, H. W., Paffenbarger, R. S., Clark, D. G., Cooper, K. H., & Gibbons, L. W. (1989). Physical fitness and all-cause mortality: a prospective study of healthy men and women. Jama, 262(17), 2395-2401.
Boutcher, S. H. (2010). High-intensity intermittent exercise and fat loss. Journal of obesity, 2011, 1-10.
Boutouyrie, P., Tropeano, A. I., Asmar, R., Gautier, I., Benetos, A., Lacolley, P., & Laurent, S. (2002). Aortic stiffness is an independent predictor of primary coronary events in hypertensive patients. Hypertension, 39(1), 10-15.
Burr, J. F., Rowan, C. P., Jamnik, V. K., & Riddell, M. C. (2010). The role of physical activity in type 2 diabetes prevention: physiological and practical perspectives. The Physician and sportsmedicine, 38(1), 72-82.
Burr, J. F., Beck, J. L., & Durocher, J. J. (2017). The relationship of high-intensity cross-training with arterial stiffness. Journal of Sport and Health Science, 1-6.
Cameron, J. D., & Dart, A. M. (1994). Exercise training increases total systemic arterial compliance in humans. American Journal of Physiology-Heart and Circulatory Physiology, 266(2), H693-H701.
Chirinos, J. A., Kips, J. G., Jacobs, D. R., Brumback, L., Duprez, D. A., Kronmal, R., … & Segers, P. (2012). Arterial wave reflections and incident cardiovascular events and heart failure: MESA (Multiethnic Study of Atherosclerosis). Journal of the American College of Cardiology, 60(21), 2170-2177.
Cochran, A. J., Percival, M. E., Tricarico, S., Little, J. P., Cermak, N., Gillen, J. B., … & Gibala, M. J. (2014). Intermittent and continuous high‐intensity exercise training induce similar acute but different chronic muscle adaptations. Experimental physiology, 99(5), 782-791.
Cohen, J. (1988) Statistical power analysis for the behavioral sciences. 2nd edn. Hillsdale, NJ.: Lawrence Erlbaum Associates.
Cohen, J. (1992). A power primer. Psychological bulletin, 112(1), 155-159.
Dischl, B., Engelberger, R. P., Gojanovic, B., Liaudet, L., Gremion, G., Waeber, B., & Feihl, F. (2011). Enhanced diastolic reflections on arterial pressure pulse during exercise recovery. Scandinavian journal of medicine & science in sports, 21(6), e325-e333.
Elias, M. F., Dore, G. A., Davey, A., Abhayaratna, W. P., Goodell, A. L., & Robbins, M. A. (2011). Norms and reference values for pulse wave velocity: one size does not fit all. The Journal of Bioscience and Medicine, 1(1), 1-10.
Fisher, G., Brown, A. W., Brown, M. M. B., Alcorn, A., Noles, C., Winwood, L., … & Allison, D. B. (2015). High intensity interval-vs moderate intensity-training for improving cardiometabolic health in overweight or obese males: a randomized controlled trial. PloS one, 10(10), 1-15.
Fleenor, B. S., & Berrones, A. J. (2015). Arterial Stiffness: Implications and Interventions. Cham: Springer.
Fletcher, G. F., Froelicher, V. F., Hartley, L. H., Haskell, W. L., & Pollock, M. L. (1990). Exercise standards. A statement for health professionals from the American Heart Association. Circulation, 82(6), 2286-2322.
Fletcher, G. F., Balady, G., Blair, S. N., Blumenthal, J., Caspersen, C., Chaitman, B., … & Pollock, M. L. (1996). Statement on exercise: benefits and recommendations for physical activity programs for all Americans. Circulation, 94(4), 857-862.
Fletcher, G. F., Ades, P. A., Kligfield, P., Arena, R., Balady, G. J., Bittner, V. A., … & Gulati, M. (2013). Exercise standards for testing and training. Circulation, 128(8), 873-934.
Fox, E. L., Bartels, R. L., Billings, C. E., O’Brien, R., Bason, R., & Mathews, D. K. (1975). Frequency and duration of interval training programs and changes in aerobic power. Journal of Applied Physiology, 38(3), 481-484.
Gibala, M. J., Little, J. P., MacDonald, M. J., & Hawley, J. A. (2012). Physiological adaptations to low‐volume, high‐intensity interval training in health and disease. The Journal of physiology, 590(5), 1077-1084.
Gregg, E. W., Cauley, J. A., Stone, K., Thompson, T. J., Bauer, D. C., Cummings, S. R., … & Study of Osteoporotic Fractures Research Group. (2003). Relationship of changes in physical activity and mortality among older women. Jama, 289(18), 2379-2386.
Guimaraes, G. V., Ciolac, E. G., Carvalho, V. O., D’Avila, V. M., Bortolotto, L. A., & Bocchi, E. A. (2010). Effects of continuous vs. interval exercise training on blood pressure and arterial stiffness in treated hypertension. Hypertension Research, 33(6), 627-632.
Hambrecht, R., Hilbrich, L., Erbs, S., Gielen, S., Fiehn, E., Schoene, N., & Schuler, G. (2000). Correction of endothelial dysfunction in chronic heart failure: additional effects of exercise training and oral L-arginine supplementation. Journal of the American College of Cardiology, 35(3), 706-713.
Hanssen, H., Nussbaumer, M., Moor, C., Cordes, M., Schindler, C., & Schmidt-Trucksäss, A. (2015). Acute effects of interval versus continuous endurance training on pulse wave reflection in healthy young men. Atherosclerosis, 238(2), 399-406.
Hayashi, K., Sugawara, J., Komine, H., Maeda, S., & Yokoi, T. (2005). Effects of aerobic exercise training on the stiffness of central and peripheral arteries in middle-aged sedentary men. The Japanese journal of physiology, 55(4), 235-239.
Haykowsky, M. J., Liang, Y., Pechter, D., Jones, L. W., McAlister, F. A., & Clark, A. M. (2007). A meta-analysis of the effect of exercise training on left ventricular remodeling in heart failure patients: the benefit depends on the type of training performed. Journal of the American College of Cardiology, 49(24), 2329-2336.
Hazell, T. J., MacPherson, R. E., Gravelle, B. M., & Lemon, P. W. (2010). 10 or 30-s sprint interval training bouts enhance both aerobic and anaerobic performance. European journal of applied physiology, 110(1), 153-160.
Heffernan, K. S., Jae, S. Y., Echols, G. H., Lepine, N. R., & Fernhall, B. (2007). Arterial stiffness and wave reflection following exercise in resistance-trained men. Medicine and science in sports and exercise, 39(5), 842-848.
Heidenreich, P. A., Trogdon, J. G., Khavjou, O. A., Butler, J., Dracup, K., Ezekowitz, M. D., … & Lloyd-Jones, D. M. (2011). Forecasting the future of cardiovascular disease in the United States. Circulation, 123(8), 933-944.
Helgerud, J., Hoydal, K., Wang, E., Karlsen, T., Berg, P., Bjerkaas, M., … & Hoff, J. (2007). Aerobic High-Intensity Intervals Improve VO2max More Than Moderate Training. Medicine and science in sports and exercise, 39(4), 665-671.
Humphrey, R. (2006). Clinical applications: the exercise caloric challenge. ACSM’s Health & Fitness Journal, 10(2), 40-41.
Kass, D. A., Saeki, A., Tunin, R. S., & Recchia, F. A. (1996). Adverse influence of systemic vascular stiffening on cardiac dysfunction and adaptation to acute coronary occlusion. Circulation, 93(8), 1533-1541.
Ketelhut, R. G., Franz, I. W., & Scholze, J. (2004). Regular exercise as an effective approach in antihypertensive therapy. Medicine and science in sports and exercise, 36(1), 4-8.
Ketelhut, S., Milatz, F., Heise, W., & Ketelhut, R. G. (2016). Influence of a high-intensity interval training session on peripheral and central blood pressure at rest and during stress testing in healthy individuals. Vasa, 45(5), 373-377.
Kingwell, B. A., Berry, K. L., Cameron, J. D., Jennings, G. L., & Dart, A. M. (1997). Arterial compliance increases after moderate-intensity cycling. American Journal of Physiology-Heart and Circulatory Physiology, 42(5), H2186.
Laurent, S., Boutouyrie, P., Asmar, R., Gautier, I., Laloux, B., Guize, L., … & Benetos, A. (2001). Aortic stiffness is an independent predictor of all-cause and cardiovascular mortality in hypertensive patients. Hypertension, 37(5), 1236-1241.
Laursen, P. B., & Jenkins, D. G. (2002). The scientific basis for high-intensity interval training. Sports Medicine, 32(1), 53-73.
Laursen, P. B. (2010). Training for intense exercise performance: high‐intensity or high‐volume training? Scandinavian journal of medicine & science in sports, 20(2), 1-10.
Lavie, C. J., Arena, R., Swift, D. L., Johannsen, N. M., Sui, X., Lee, D. C., … & Blair, S. N. (2015). Exercise and the cardiovascular system. Circulation research, 117(2), 207-219.
Lee, I. M., Sesso, H. D., Oguma, Y., & Paffenbarger, R. S. (2003). Relative intensity of physical activity and risk of coronary heart disease. Circulation, 107(8), 1110-1116.
Linke, A., Schoene, N., Gielen, S., Hofer, J., Erbs, S., Schuler, G., & Hambrecht, R. (2001). Endothelial dysfunction in patients with chronic heart failure: systemic effects of lower-limb exercise training. Journal of the American College of Cardiology, 37(2), 392-397.
McArdle, W. D., Katch, F. I., & Katch, V. L. (2010). Exercise physiology: nutrition, energy, and human performance. Baltimore: Lippincott Williams & Wilkins.
McEniery, C. M., Hall, I. R., Qasem, A., Wilkinson, I. B., Cockcroft, J. R., & ACCT Investigators. (2005). Normal vascular aging: differential effects on wave reflection and aortic pulse wave velocity: the Anglo-Cardiff Collaborative Trial (ACCT). Journal of the American College of Cardiology, 46(9), 1753-1760.
Milatz, F., Ketelhut, S., & Ketelhut, R. G. (2017). Acute Effects of Moderate Continuous Training on Stress Test-Related Pulse Pressure and Wave Reflection in Healthy Men. High Blood Pressure & Cardiovascular Prevention, 24(1), 61-67.
Mitchell, G. F., Parise, H., Benjamin, E. J., Larson, M. G., Keyes, M. J., Vita, J. A., … & Levy, D. (2004). Changes in arterial stiffness and wave reflection with advancing age in healthy men and women. Hypertension, 43(6), 1239-1245.
Montero, D. (2015). The association of cardiorespiratory fitness with endothelial or smooth muscle vasodilator function. European journal of preventive cardiology, 22(9), 1200-1211.
Munir, S. M., Jiang, B., Guilcher, A., Brett, S., Redwood, S., Marber, M. S., & Chowienczyk, P. (2008). Exercise reduces arterial pressure augmentation through vasodilation of muscular arteries in man. Am J Physiol Heart Circ Physiol, 294, H1645-H1650.
Murtagh, E. M., Boreham, C. A., Nevill, A., Hare, L. G., & Murphy, M. H. (2005). The effects of 60 minutes of brisk walking per week, accumulated in two different patterns, on cardiovascular risk. Preventive medicine, 41(1), 92-97.
Nitzsche, N., Weigert, M., Baumgärtel, L., Auerbach, T., Schuffenhauer, D., Nitzsche, R., & Schulz, H. (2016). Acute Effects of Different Strength Training Protocols on Arterial Stiffness in Healthy Subjects. International Journal of Sports Science, 6(5), 195-202.
Park, S., Rink, L. D., & Wallace, J. P. (2006). Accumulation of physical activity leads to a greater blood pressure reduction than a single continuous session, in prehypertension. Journal of hypertension, 24(9), 1761-1770.
Peng, X., Haldar, S., Deshpande, S., Irani, K., & Kass, D. A. (2003). Wall stiffness suppresses Akt/eNOS and cytoprotection in pulse-perfused endothelium. Hypertension, 41(2), 378-381.
Perk, J., De Backer, G., Gohlke, H., Graham, I., Reiner, Z., Verschuren, M., … & Deaton, C. (2012). European Association for Cardiovascular Prevention & Rehabilitation (EACPR); ESC Committee for Practice Guidelines (CPG). European Guidelines on cardiovascular disease prevention in clinical practice (version 2012). The Fifth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice (constituted by representatives of nine societies and by invited experts). Eur Heart J, 33(13), 1635-701.
Perry, C. G., Heigenhauser, G. J., Bonen, A., & Spriet, L. L. (2008). High-intensity aerobic interval training increases fat and carbohydrate metabolic capacities in human skeletal muscle. Applied Physiology, Nutrition, and Metabolism, 33(6), 1112-1123.
Pressler, A., Hanssen, H., Dimitrova, M., Krumm, M., Halle, M., & Scherr, J. (2011). Acute and chronic effects of marathon running on the retinal microcirculation. Atherosclerosis, 219(2), 864-868.
Rakobowchuk, M., Tanguay, S., Burgomaster, K. A., Howarth, K. R., Gibala, M. J., & MacDonald, M. J. (2008). Sprint interval and traditional endurance training induce similar improvements in peripheral arterial stiffness and flow-mediated dilation in healthy humans. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology, 295(1), R236-R242.
Rakobowchuk, M., Stuckey, M. I., Millar, P. J., Gurr, L., & MacDonald, M. J. (2009). Effect of acute sprint interval exercise on central and peripheral artery distensibility in young healthy males. European journal of applied physiology, 105(5), 787-795.
Ranadive, S. M., Fahs, C. A., Yan, H., Rossow, L. M., Agiovlasitis, S., & Fernhall, B. (2012). Comparison of the acute impact of maximal arm and leg aerobic exercise on arterial stiffness. European journal of applied physiology, 112(7), 2631-2635.
Rognmo, Ø., Hetland, E., Helgerud, J., Hoff, J., & Slørdahl, S. A. (2004). High intensity aerobic interval exercise is superior to moderate intensity exercise for increasing aerobic capacity in patients with coronary artery disease. European Journal of Cardiovascular Prevention & Rehabilitation, 11(3), 216-222.
Ross, L. M., Porter, R. R., & Durstine, J. L. (2016). High-intensity interval training (HIIT) for patients with chronic diseases. Journal of Sport and Health Science, 5(2), 139-144.
Safar, M. E., & London, G. M. (2000). Therapeutic studies and arterial stiffness in hypertension: recommendations of the European Society of Hypertension. Journal of hypertension, 18(11), 1527-1535.
Schnohr, P., O’Keefe, J. H., Marott, J. L., Lange, P., & Jensen, G. B. (2015). Dose of jogging and long-term mortality: the Copenhagen City Heart Study. Journal of the American College of Cardiology, 65(5), 411-419.
Shiraev, T., & Barclay, G. (2012). Evidence based exercise: Clinical benefits of high intensity interval training. Australian family physician, 41(12), 960 – 962.
Siasos, G., Athanasiou, D., Terzis, G., Stasinaki, A., Oikonomou, E., Tsitkanou, S., … & Tousoulis, D. (2016). Acute effects of different types of aerobic exercise on endothelial function and arterial stiffness. European journal of preventive cardiology, 23(14), 1565-1572.
Smulyan, H., Asmar, R. G., Rudnicki, A., London, G. M., & Safar, M. E. (2001). Comparative effects of aging in men and women on the properties of the arterial tree. Journal of the American College of Cardiology, 37(5), 1374-1380.
Soares, F. H. R., & de Sousa, M. B. C. (2013). Different types of physical activity on inflammatory biomarkers in women with or without metabolic disorders: a systematic review. Women & health, 53(3), 298-316.
Swain, D. P., & Franklin, B. A. (2002). Is there a threshold intensity for aerobic training in cardiac patients?. Med Sci Sports Exerc, 34, 1071-1075.
Tanaka, H., DeSouza, C. A., & Seals, D. R. (1998). Absence of age-related increase in central arterial stiffness in physically active women. Arteriosclerosis, thrombosis, and vascular biology, 18(1), 127-132.
Tanaka, H., Dinenno, F. A., Monahan, K. D., Clevenger, C. M., DeSouza, C. A., & Seals, D. R. (2000). Aging, habitual exercise, and dynamic arterial compliance. Circulation, 102(11), 1270-1275.
Tanasescu, M., Leitzmann, M. F., Rimm, E. B., Willett, W. C., Stampfer, M. J., & Hu, F. B. (2002). Exercise type and intensity in relation to coronary heart disease in men. Jama, 288(16), 1994-2000.
Tjønna, A. E., Lee, S. J., Rognmo, Ø., Stølen, T. O., Bye, A., Haram, P. M., … & Kemi, O. J. (2008). Aerobic interval training versus continuous moderate exercise as a treatment for the metabolic syndrome. Circulation, 118(4), 346-354.
Tol, B. A., Huijsmans, R. J., Kroon, D. W., Schothorst, M., & Kwakkel, G. (2006). Effects of exercise training on cardiac performance, exercise capacity and quality of life in patients with heart failure: A meta‐analysis. European journal of heart failure, 8(8), 841-850.
Tordi, N., Mourot, L., Colin, E., & Regnard, J. (2010). Intermittent versus constant aerobic exercise: effects on arterial stiffness. European journal of applied physiology, 108(4), 801-809.
Trapp, E. G., Chisholm, D. J., Freund, J., & Boutcher, S. H. (2008). The effects of high-intensity intermittent exercise training on fat loss and fasting insulin levels of young women. International journal of obesity, 32(4), 684-691.
Tschentscher, M., Eichinger, J., Egger, A., Droese, S., Schönfelder, M., & Niebauer, J. (2016). High-intensity interval training is not superior to other forms of endurance training during cardiac rehabilitation. European journal of preventive cardiology, 23(1), 14-20.
Vincent, W. J., & Weir, J. P. (2012). Statistics in Kinesiology – 3rd edition (3rd ed.). Champaign, IL: Human Kinetics Publishers.
Vlachopoulos, C., Aznaouridis, K., O’rourke, M. F., Safar, M. E., Baou, K., & Stefanadis, C. (2010). Prediction of cardiovascular events and all-cause mortality with central haemodynamics: a systematic review and meta-analysis. European heart journal, 31(15), 1865-1871.
Vlachopoulos, C., Aznaouridis, K., & Stefanadis, C. (2010). Prediction of cardiovascular events and all-cause mortality with arterial stiffness: a systematic review and meta-analysis. Journal of the American College of Cardiology, 55(13), 1318-1327.
Vlachopoulos, C., Kardara, D., Anastasakis, A., Baou, K., Terentes-Printzios, D., Tousoulis, D., & Stefanadis, C. (2010). Arterial stiffness and wave reflections in marathon runners. American journal of hypertension, 23(9), 974-979.
Warburton, D. E., Nicol, C. W., & Bredin, S. S. (2006a). Health benefits of physical activity: the evidence. Canadian medical association journal, 174(6), 801-809.
Warburton, D. E., Nicol, C. W., & Bredin, S. S. (2006b). Prescribing exercise as preventive therapy. Canadian Medical Association Journal, 174(7), 961-974.
Wenger, H. A., & Macnab, R. B. (1975). Endurance training: the effects of intensity, total work, duration and initial fitness. The Journal of sports medicine and physical fitness, 15(3), 199-211.
Wilkinson, I. B., MacCallum, H., Flint, L., Cockcroft, J. R., Newby, D. E., & Webb, D. J. (2000). The influence of heart rate on augmentation index and central arterial pressure in humans. The Journal of physiology, 525(1), 263-270.
World Health Organization. (2011). Global Status Report on Non-Communicable Diseases 2010. Description of the Global Burden of NCDs, Their Risk Factors and Determinants, 2011. Google Scholar.
Wray, D. W., Nishiyama, S. K., Donato, A. J., Sander, M., Wagner, P. D., & Richardson, R. S. (2007). Endothelin-1-mediated vasoconstriction at rest and during dynamic exercise in healthy humans. American Journal of Physiology-Heart and Circulatory Physiology, 293(4), H2550-H2556.
Zieman, S. J., Melenovsky, V., & Kass, D. A. (2005). Mechanisms, pathophysiology, and therapy of arterial stiffness. Arteriosclerosis, thrombosis, and vascular biology, 25(5), 932-943.
Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allRelated Content
All TagsContent relating to: "Fitness"
Fitness is the physical condition of being fit and healthy. Being physically fit relates to an ability to perform physical or sporty activities, cardiorespiratory endurance, and muscular strength. Fitness is achieved through exercise and nutrition.
Related Articles
DMCA / Removal Request
If you are the original writer of this dissertation and no longer wish to have your work published on the UKDiss.com website then please:




