Functional Traits in Urban and Parkland Trees in the UK in Response to Increased Pressures on London Plane (Platanus x Hispanica)
Info: 7974 words (32 pages) Dissertation
Published: 16th Dec 2019
Tagged: Ecology
An Analysis of Functional Traits in Urban and Parkland Trees in the UK in Response to Increased Pressures on London Plane (Platanus x hispanica)
Abstract
In response to recently reported increased pest and disease pressure on a key large UK tree species (London plane) a trait based review of a total of nine commonly occurring broadleaved UK urban tree species was undertaken. Wood density and Specific Leaf Area was tested from trees located in an urban and a park setting for all nine species.
Table of Contents
1.2 Urban Trees in the UK; London Plane (Platanus x hispanica)
1.3 Functional Traits in Trees
2.3 Leaf Data Collection Methods
2.4 Branch Wood Data Collection Methods
2.5 Data Collection Limitations
2.6 Measuring Specific Leaf Area
1. Introduction
1.1 Trees in Urban Areas
The diverse range of benefits afforded by an urban forest to people living within urban conurbations are numerous and well documented (Escobedo et al, 2011; Ulmer et al, 2016). Factors such as population growth and climate change will continue to put increasing pressure and demand on the regulating, provisioning, supporting and cultural ecosystem services that urban trees play a crucial role in delivering (UK National Ecosystem Assessment, 2011). Some examples of the environmental, economic and social benefits that urban trees provide to inhabitants of urban conurbations are summarised in Table 1 below.
| Environmental Benefits | Social Benefits | Economic Benefits |
|
|
|
|
|
|
|
|
|
|
|
|
|
Table 1: Benefits provided by urban trees to inhabitants of urban conurbations
The multitude of ecosystem services provided to cities by trees makes tree planting an essential component of urban planning. However, the space and budget allocated for trees within modern infrastructure is often small, with the conditions for trees often being incredibly harsh (Forestry Commission, 2016). Therefore, the species that are selected for use in urban spaces must be efficient and suitable in terms of resilience and longevity within harsh urban conditions. This will maximise the efficacy of ecosystem services being delivered by the urban forest. At present, there is a gap in the understanding and information available regarding urban plant ecology (Lambrecht et al, 2016). There is also currently a lack of understanding and information available on how urban trees being used in the UK vary in growth rates and if there are useful relationships between allometric variables in our urban tree stock that could be used to aid urban planning (Monteiro et al, 2016). Further study will continue to improve the selection process and the understanding behind urban tree selection within urban forestry in the UK.
As well as being able to tolerate urban conditions, tree species selected as part of urban planting schemes must fulfil other criteria; diversifying the species mix within the urban forest is essential to ensure sustainability (Sjoman et al, 2016). Particularly within urban areas where environmental stress factors are highly concentrated, the importance of tree species selection is heightened as stressed trees are more likely to succumb to these pressures and reduce the efficacy of the urban forest in providing essential ecosystem services (Sturrock et al, 2011). Aesthetics, maintenance costs and ability to provide shade are also all factors often considered by landscape architects when selecting tree species for urban use. Although often, information pertaining to an individual species ability to fulfil specific criteria and tolerate specific site constraints and environmental stress factors is not readily available (Roy et al, 2016). A greater understanding of how tree species perform in specific site conditions and an insight into their overall adaptive trait strategy would enable the species selection process to be refined and improved for a wide range of urban forestry practitioners. Species selection for resilience has been repeatedly identified as one of the key measures to be used for building resilient urban forests for the future. However, despite this a need for more information to aid practitioners in urban tree selection processes to achieve this has also been highlighted (Conway and Vecht, 2015).
Resilience in the context of ecological networks and urban forests has been described by DEFRA, (2007) as; ‘capability to absorb, resist or recover from disturbances and damage, while continuing to meet its overall objectives of supporting biodiversity and providing ecosystem services’. It is the qualities or traits that enable resilience that are required to be present in the tree species that constitute our urban forests.
As well as resilience there is also scope to better understand the plasticity of tree species during environmental changes and the intra-species adaptability within genera. Garzon et al (2011) propose that species range predictions traditionally predicted with Species Distribution Models (SDMs) for the effects of climate change do not account for phenotypic plasticity. This could result in under or over exaggerating the risk presented to individual tree species during climate change and other environmental stress factors. Study carried out at a local plant/tree community level will provide greater insight into intra-species responses to environmental stress factors.
1.2 Urban Trees in the UK; London Plane (Platanus x hispanica)
In the United Kingdom, the most frequently occurring tree planted in urban environments across the UK is the London Plane (Platanus x hispanica). In London, this species accounts for over 50% of all trees within the capitals’ urban forest (Forestry Commission, 2017). The prevalence of this species, particularly in urban areas is due to its resilience and its ability to withstand harsh urban conditions. These are namely but not exhaustively; compacted and poor-quality soils, limited rooting space, cyclical pruning regimes and high levels of air pollution. Despite these harsh conditions London Planes can grow into large and healthy specimens which ensure that the essential ecosystem services that only mature trees can provide, are delivered to the city. McPherson et al (1999) have demonstrated through a cost benefit analysis study, that planting large tree species (such as London plane) in urban areas also provides annual net benefits 44% greater than medium tree species and 92% greater than small tree species. This suggests that improved tree selection can not only improve ecosystem services but can also introduce efficiency into budgets for tree planting and subsequent management.
As well as the established pressures that affect all trees in urban areas (such as climate change, increased levels of pests and disease) it has recently been identified that there are numerous emerging pests and pathogens that could be potentially devastating particularly for London Plane. Given the ubiquity of this species in UK urban spaces and planting schemes and the large contribution it is making to the health and function of the urban forest, it is essential to acknowledge these emerging and significant threats and to understand the resilience and plasticity of all tree species which currently constitute the UK urban forest in order to enable pro-active management. This will mitigate against the impacts from any potential wide spread loss of large and mature urban trees from within the UK urban forest tree stocks.
Tubby and Perez-Sierra (2015) have recently highlighted several established and emerging threats to London Plane in the UK. These include some pathogens that are native to the UK such as; Plane anthracnose (Apoignomonia veneta) and decay fungi such as Inonotus hispidus and Perenniporia fraxinea. However, there are also established pathogens in the UK of unknown provenance such as Massaria disease (Splanchnonema platani) and threats not yet in the UK, but that are having a significant impact elsewhere and present a genuine and potentially catastrophic threat, such as Xylella (Xylella fastidiosa) which is considered to be a serious threat to not only London Plane but also a wide range of UK broadleaved species.Canker stain of Plane (Ceratocystis platani) which affects Platanus spp. Specifically is another disease highlighted as being a potentially devastating threat to the UK’s plane trees given the extent of the tree loss it is responsible for to date in Europe.


Figure 1: Massaria (Forestry Commission, 2012) Figure 2: Ceratocystis platani (Horticulture Week, 2016)
In the UK at present, the most significant threats to Plane trees in terms of pests and disease are the established threat of Massaria and the potential impact of the arrival of Canker stain of Plane from mainland Europe. Since 2009 Massaria disease has been reported to be affecting Plane trees within the London area. The disease is a fungal pathogen and manifests as a lesion on the upper surface of mid-canopy limbs and causes failure and branch drop of affected limbs (Forestry Commission, 2017). This disease presents challenges for tree management teams and managers particularly due to the health and safety hazard of falling limbs and the necessity for climbing inspections to confirm the presence of the disease. The disease is found with increased frequency in hotter and drier climates which could become exacerbated in the UK in the future by the effects of climate change (Forestry Commission, 2008).
Canker stain of Plane (Ceratocystis platani) is a fast acting and destructive pathogen that can cause rapid tree mortality in affected Plane trees, it has been problematic in Europe since it arrived there from the USA during the second world war (Panconesi, 1999). This disease currently has no confirmed presence in the UK however, given the level of destruction the disease has caused in urban tree stocks in Europe there is concern surrounding a widespread loss of Plane trees in the UK and most notably within London should the disease be discovered in the UK (Forestry Commission, 2018).
There have been ongoing surveys monitoring for Canker stain of Plane in the UK taking place since 2013 and incoming Platanus spp. plants require a plant passport as detailed in the Canker Stain of Plane Contingency Plan, published by the Forestry Commission (Nianiaka, 2016).
A recent study by Pelleteret et al (2017) has also used PCR and DNA sequencing techniques to confirm the presence of Ceratocystis platani in suspected cases of the disease in urban Plane trees in Switzerland. However, as with all the previously mentioned established and emerging threats to Plane trees in the UK, the outcome in the event of an outbreak or increase in frequency in these pathogens is potentially severe. Brasier (2008) also suggests that given our historic and current policies on international trading in plants and our proximity to mainland Europe, we should not consider our planted or native trees to be entirely biologically secure. It is suggested that as well as implementing measures to reduce the likelihood of large scale pest and disease outbreaks in the UK urban forest, a proactive approach to urban tree selection should be used to increase diversity in the tree stock and to identify the species and characteristics required to create as sustainable and robust a resource as possible.
1.3 Functional Traits in Trees
Functional traits can manifest within plants and trees in several ways e.g. as morphological, phenological or behavioural characteristics. These characteristics directly influence a plants performance or ‘fitness’ and can be used to characterise plant community responses to changes in the environment (Nock et al, 2016). In the context of arboriculture and urban forest management Goodness et al (2016) states that functional traits are strongly linked to the regulating, supporting, provisioning and cultural ecosystem services that urban trees provide to the built environment. It is important for this relationship between traits and ecosystem services to be used and developed and for a trait based approach to be used to inform urban forest management and planting decisions to help build resilient urban forests.
The characteristics or functional traits in trees can also be indicative of its overall growth and adaptation strategies and of how it interacts and responds to its biotic and abiotic environment. These strategies play a key role in determining how and why a tree can grow where it does and how successful it can grow in that location. An analysis of the primary organs of a tree i.e. the leaves, stem and roots can provide an indication of the adaptive strategies that a tree may be using in order for it to survive in its environment. Reich (2014) proposes that the environmental site factors and constraints, combined with how a plant trades off and utilises functional traits, will result in an overall slow, medium or fast strategy in terms of how a plant acquires and processes available resources. This strategy also strongly influences the type of ecosystem services the tree will deliver to its environment. An understanding of where a plant or tree sits within this spectrum will enable trees to be matched to sites far more effectively.
Due to plant functional traits being commonly used and adept at assessing the drivers behind species abundance, ecosystem services and dynamic phenotype adaptation to environmental conditions, there is a wealth of plant functional trait data available in databases from multiple regions (Kattge et al, 2011). This data is also being more frequently recommended for use in aiding the making of policy and management decisions for large areas of land and the associated trees and vegetation, in order to maximise ecosystem services (Lavorel et at, 2010). The use of trait data in this way has also recently highlighted the importance of small scale, local level studies such as this project, that acknowledge and include intra specific trait dynamics and local community level variation (Albert et al, 2010). It is also important that the data being collected i.e. the traits being selected for analysis are able to provide good insight into the relationship between the plants or trees and their adaptation to their environment.
1.4 Specific Leaf Area
The leaves (Figure 2) are one of the most important and key organs of a tree, the way in which they function also contributes significantly to the functioning of the entire ecosystem that a tree is part of. Leaf traits are strongly linked to a range of abiotic and biotic factors and ecosystem services (Figure 3), this connection makes them adept at indicating a how a plant is responding to environmental stresses occurring within it at an individual at a local level and can be a very useful trait to monitor for tree managers (Liu et al, 2017).


Figure 2: London Plane leaves (My landscapes, 2017) Figure 3: Plane trees providing shade (tbeartravels, 2018)
Specific Leaf Area (SLA) can be defined as the ratio of leaf area to leaf mass, with leaves growing in higher light concentrations having thicker leaves partly due to extra palisade cells being present. This in turn increases the number of chloroplasts and overall photosynthetic potential of the leaves (Evans and Porter, 2001).
SLA also directly influences the rate at which a trees canopy expands and grows. This relationship is due to SLA strongly influencing the total leaf area of a trees canopy and consequently determining the light use efficiency and photosynthetic capability of the tree (Kumar et al, 2012). SLA is also a key parameter to consider in relation to tree growth as it will strongly influence how much leaf area will be created per unit of overall biomass. This relationship also has implications in terms of climate change, as an increase in CO2 typically causes an increase in overall leaf mass and a reduction in SLA (Kimball et al, 2002).
By monitoring SLA in the tree species commonly occurring in the UK urban forest it will be possible to establish if the species are adapting to environmental stresses between a park and urban site and if the strategy observed is consistent with existing knowledge of leaf traits. Greenwood et al have demonstrated that at a global level, high SLA can be a reliable indicator of a tree species more susceptible to mortality in drought conditions. However, they also demonstrate that high SLA is indicative of a faster growth rate and increased carbon storage. Further study of species at a local level will help to understand how these factors apply to UK urban forest management.
1.5 Wood Density
The wooden structure of a tree (See Figure 3) must simultaneously perform multiple essential functions. These include but are not limited to; providing a high vantage point for leaves to compete for light, structural support for the entire physical structure, transporting water from the roots to the leaves and transporting food around the entire organism to ensure it remains alive, healthy and functioning (Thomas, 2000).
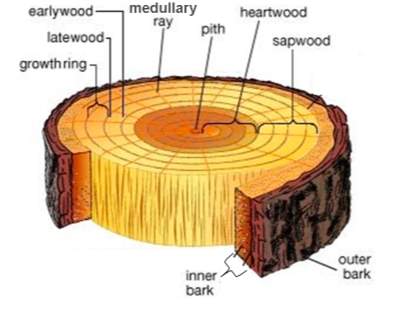
Figure 3: Diagram of tree cross section (Woodstairs, 2018)
Wood density is an important parameter when considering suitability and efficiency in trees as it is essential in ensuring the survival and competitive ability of a species within its environment. This trait also gives a clear insight into the investment trade-off that is occurring between the construction of solid wood and xylem tissue (Fajardo, 2016). Wood density is also an important trait at a global level as knowledge of how wood density varies between species allows for more accurate estimations of the carbon being sequestered within forests and urban tree stocks (Denman et al, 2007).
Wood density is a particularly complex trait and can be influenced by several different factors (including SLA). This is due to overall plant or tree growth being strongly influenced primarily by water and nutrient availability. In turn, water availability and the hydraulic efficiency and connectivity in the woody tissues from the roots to the leaves will have an effect on the leaf phenotype (Machado et al, 2007).
In this study, by monitoring wood density in different locations particularly in conjunction with another trait related to water, carbon and nutrients such as SLA, an insight into the ecological strategy of a species can begin to be profiled. The intention is to ascertain if the trees commonly constituting the UK urban forest are conforming to the existing model of wood and leaf traits i.e. tree species with denser, slower growing wood being associated with higher biomechanical and hydraulic efficiency and species with less dense wood having a faster growth rate at the expense of mechanical strength and having a higher susceptibility to drought (Chave et al, 2009). Previous studies involving wood functional traits have largely concentrated on data gathered from within a forest setting e.g. Hoeber et al (2014). Local level urban tree data will provide a useful insight into this trait.
2. Research
2.1 Aims
In response to current UK urban forest management issues, and following a review of the relevant and current literature regarding UK tree species selection and plant functional traits the aims and hypotheses for this project are;
The overall aims of this project are to undertake a trait based survey and review of some of the most commonly occurring tree species currently constituting the UK urban forest particularly in response to the increased pressure on London plane. The intention is to determine if a trait based approach can inform planting choices for suitable replacement species for urban use.
The specific traits selected for investigation are wood density and specific leaf area. In carrying out this research the aim is to highlight any trait trends within species and to look for associations between how functional traits vary in different site conditions i.e. in a park tree or urban tree, to try to understand if functional traits in trees are indicative of a tree species’ plasticity in response to environmental stress factors present in an urban location.
- There will be a statistically significant difference in specific leaf area between trees in an urban location and those in a park location.
- There will be a statistically significant difference in specific leaf area at a species level between trees in an urban location and those in a park location.
- There will be a statistically significant difference in wood density between trees in an urban location and those in a park location and at a species level.
- There will be a statistically significant difference in wood density at a species level between trees in an urban location and those in a park location.
2.2 Methods
The methods used to complete the research for this project were carried out in accordance with methods used in similar research and are considered to currently be best practice. A project specific risk assessment (Appendix A) was produced prior to any research work commencing and was adhered to at all times.
To complete this project leaf and branch wood samples were collected firstly from London Plane and subsequently from eight further tree species. The species used to collect leaf and branch wood samples were chosen to reflect some of the more commonly occurring species in the UK urban forest and are listed below;
- London Plane (Platanus x hispanica)
- English oak (Quercus robur)
- Turkey oak (Quercus cerris)
- Red oak (Quercus rubra)
- Common lime (Tillia x europaea)
- Horse chestnut (Aesculus hippocastanum)
- False acacia (Robinia pseudoacacia)
- Sycamore (Acer pseudoplatanus)
- Norway maple (Acer platanoides)
Six leaf samples and six branch wood samples were collected for each species on the list. Each individual leaf and branch sample was collected from a different tree to ensure adequate replication. A species name and number labelling system was devised to enable future identification once individual samples had been dried, stored and processed. All samples were collected with the necessary permissions and in accordance with an approved risk assessment.
2.3 Leaf Data Collection Methods
Leaf samples were collected during the summer months (June – August) of 2017. The parkland tree leaf samples were collected at Hampstead Heath, North London with the help and supervision of the arboriculture team. The urban tree leaf samples were collected from street trees in the London Borough of Croydon, with the help and supervision of the Tree and Woodlands Officer.
Samples were collected from the ground by hand without the use of any climbing equipment. A description of the site conditions, canopy size, DBH and the presence of any pests, pathogens or decay fungi/physical defects were also recorded, the survey sheets and results for this project can be seen in (Appendix).
Once collected, the leaves were individually bagged and labelled to ensure samples could be accurately identified once the samples were dry. Leaf
samples were then photographed the same day (whilst fresh) on a blank piece of white paper next to a 30cm rule for scale, the use of a photographed rule enabled accurate measurements when the photographs were later analysed in ImageJ (see Photos 2, 3 and 4).


Photo 1: Branch sample collection Photo 2: Leaf sample being photographed
2.4 Branch Wood Data Collection Methods
Branch wood samples were collected between the months of September – December 2017. The parkland samples were collected at Hampstead Heath, North London with the help and supervision of the arboriculture team. The urban tree samples were collected from street trees from within the London Borough of Croydon, with the help and supervision of the Tree and Woodlands Officer.
The branch wood samples were collected from both low and mid canopy branches that could be either collected with a handsaw from the ground or with the use of a pole pruner from the ground. (No climbing equipment or power tools were used in the collection of these samples). The samples were chosen from branches at random from within a tree that would provide a sample that fulfilled the following criteria;
- Samples were at least 3 cm in diameter and were cut from live branch wood.
- The branch being cut could provide a sample at least 10cm in length at the required diameter.
- The sample was collected at least 10cm away from a branch union.
- The sample being cut did not contain significant knots, cankers or defects.
Once a branch was cut, a sample piece was selected, cut to length and labelled (see photo 1). Once labelled, the samples were stored in a cool and dry place and were processed either on the day of collection or the following day. This was done to ensure drying, shrinking and breakage was kept to a minimum to ensure as accurate a measurement of wood density as possible was recorded.
2.5 Data Collection Limitations
During the data collection element of the project there were several constraints and limitations. Where possible the experimental plan was adapted to eliminate these, where this was not possible then they were mitigated against as much as was reasonably practicable.
When collecting leaf and wood samples to measure SLA and wood density it was not possible to always collect paired data i.e. a leaf and wood sample from the same tree. This was due to access and permission not always being possible. If similar work was to be conducted in future, it would be preferable to carry this out in areas where permission to collect paired data was in place.
When collecting the leaf and wood samples, samples were collected from a ‘park’ and an ‘urban’ location. Park trees were collected from parks from within either wooded or grassland areas from within the same park (Hampstead Heath). The urban samples were collected from within 2 separate locations (Boroughs of Croydon and Lewisham). For future work it would be preferable to collect the 2 paired datasets from 2 separate locations.
2.6 Measuring Specific Leaf Area
Specific Leaf Area (SLA) for each individual leaf was calculated by dividing the area (cm2) of one side of a fresh leaf by its oven dried weight (g).
In order to calculate the area for each leaf a software programme (ImageJ) was used to analyse each photographed leaf (Schneider et al, 2012). This software enabled images to be manipulated, analysed and measured and provided an accurate measurement of the area of each leaf necessary for the calculation of SLA (see Photo 3).
Once the leaves had been labelled, photographed and the areas determined in ImageJ, they were placed in their individual labelled bags and oven dried at 70°c. Once dried, they were weighed (see photo 5) and the oven dried weight was used to complete the SLA calculation. Final SLA values for all leaves in this project can be seen in (Appendix).


Photo 3: Leaf area being measured using ImageJ Photo 4: Leaf area being measured using ImageJ
2.7 Measuring Wood density
Wood density was determined by first calculating sample piece volume (adjusted for the density of water at 20oc) then calculating oven dried mass of the same sample. Final reported values were oven dried mass per fresh volume (g cm-3) detailed methods are listed below;
To measure wood density the collected branch wood samples were stripped of all outer bark and phloem and were then cut into small pieces (approx. 3cm x 3cm). The fresh volume of these pieces was then calculated by submerging each piece into a container of water on a weighing scale and recording the displacement in grams (g) (see photo 6).


Photo 5: Dried leaves being weighed Photo 6: Measuring branch sample volume
Once the fresh volume had been calculated, the wood samples were transferred to an oven and dried for at least 72 hrs at approximately 70 oc until completely dry. The final wood density of the sample was reported as (g cm-3) The stem wood density of all samples for this project can be seen in (Appendix).
3 Results
Once all leaf and wood samples were processed and the final data required for the project was collected, it was then analysed using Minitab 18 and Microsoft Excel.
3.1 Specific Leaf Area Results
The entire SLA data set (N = 108) was analysed using a General Linear Model ANOVA (Analysis of Variance). Mean values were then tested with a Tukey pairwise comparison for significant differences between SLA, species and location. This test demonstrated that the dataset as a whole demonstrated a statistically significant difference (P= 0.023) between SLA and tree location i.e. park or urban tree.
However only three tree species; (Aesculus hippocastanum, Tillia x Europea and Quercus cerris) showed a statistically significant difference at a species level in their mean SLA values. Mean SLA values for individual species can be seen in (Graph 1) and mean SLA values for each location can be seen in (Graph 2).
An Anderson Darling test for normality was also carried out on the dataset residuals which confirmed that the data was normally distributed and was also statistically significant (P= 0.023).
A two sample t-test also demonstrated that there was a significant difference (P=0.029) in SLA between a park or urban location but not between species.
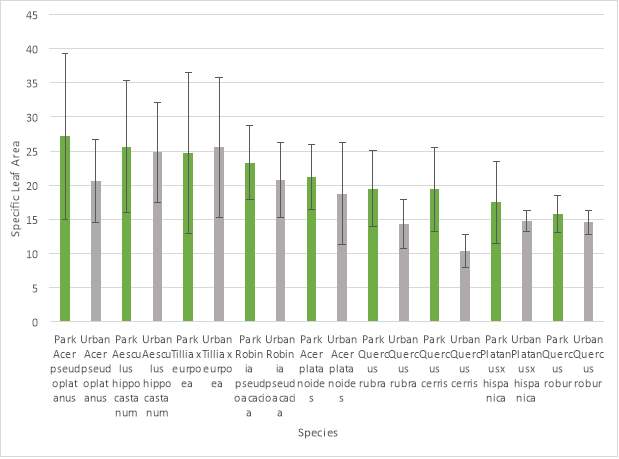 Graph 1: Mean SLA results (m2 kg-1) for species and location (error bars show standard deviation).
Graph 1: Mean SLA results (m2 kg-1) for species and location (error bars show standard deviation).
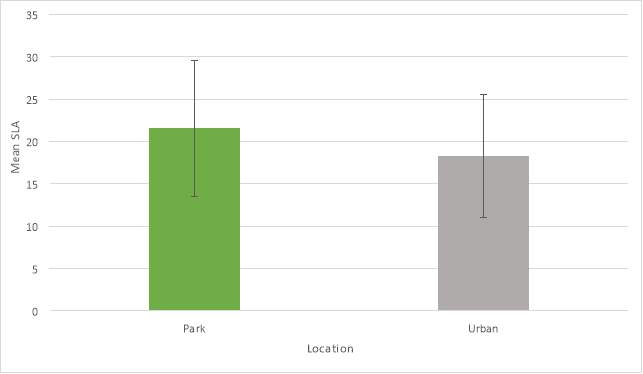
Graph 2: Mean SLA results for location (error bars show standard deviation)
3.2 Wood Density Results
The entire wood density data set (N = 108) was analysed using a General Linear Model ANOVA (Analysis of Variance). Mean values were then tested with a Tukey pairwise comparison for significance between wood density with species and location. This test demonstrated that the dataset as a whole did not show any statistically significant associations between wood density, species and tree location. However, a 2-sample t-test did show a statistically significant (P=0.394) difference between wood density and location but not between wood density and species.
Of the nine-tree species analysed only common lime (Tillia x europea) showed a significant association between wood density and species. Common lime also demonstrated the lowest wood density of any species sampled. Mean wood density values for individual species can be seen in (Graph 3) and mean wood density values summarised for each location can be seen in (Graph 4).
An Anderson Darling test for normality was also carried out on the dataset residuals which confirmed that the data was normally distributed (P= 0.089).
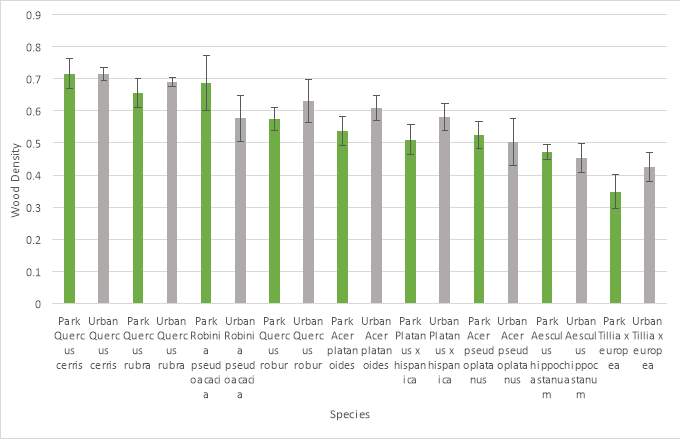 Graph 3: Mean wood density (g cm -3) of species and location (error bars show standard deviation).
Graph 3: Mean wood density (g cm -3) of species and location (error bars show standard deviation).
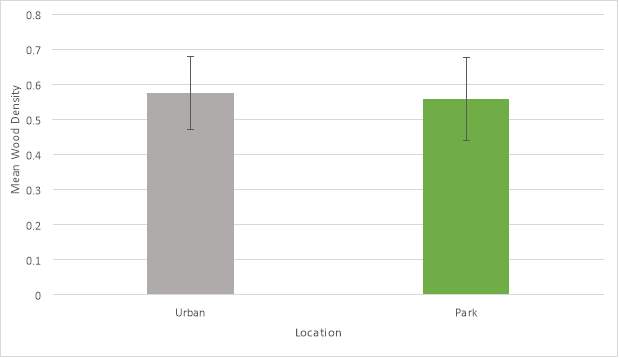
Graph 4: Mean wood density values for location (error bars show standard deviation).
4 Discussion
The leaf data results showed a statistically significant difference (P= 0.023) in SLA between the trees in a park and urban location. Therefore the proposed hypothesis from this study can be accepted. This suggests that there is an adaptive response by this trait in response to environmental stress increases in an urban area and that SLA can be a reliable indicator of this process. At a species level, only Aesculus hippocastanum, Tillia x europea and Quercus cerris showed a statistically significant difference in mean SLA values. These species are characterised by high wood density and low SLA (Q. cerris) and low wood density and high SLA (A. hippocastanum, T. x europea).
The wood density data set did not show a statistically significant difference between density, species and location. In this case the null hypothesis should be accepted.
A statistically significant difference (P=0.394) was demonstrated between wood density in a park and urban location by a two-sample t-test, this hypothesis can also be accepted. This suggests that wood density can be a trait indicative of adaptation between sites as environmental stresses increase. However, due to wood density being a highly complex trait further more detailed and controlled study would be required to build on these findings.
The results from this project show alignment with previous research regarding functional traits in trees in response to environmental stress factors. Greenwood et al (2017) have demonstrated that tree species with low wood density and high SLA are the most prone to succumb to drought induced mortality. This is one of the most prevalent urban environmental stress factors to consider when selecting and managing urban trees. Of the species surveyed for this project those with low SLA and high wood density such as Quercus,
5 Conclusions
6 References
Albert, C. H., Thuiller, W., Yoccoz, N. G., Douzet, R., Aubert, S., Lavorel, S. (2010). ‘A multi-trait approach reveals the structure and the relative importance of intra – vs. interspecific variability in plant traits’. Functional ecology, Vol 24, (6), pp. 1192 – 1201.
Alvey, A. A. (2006) ‘Promoting and preserving biodiversity in the urban forest’ Urban Forestry & Urban Greening, 5, pp. 195-201.
Chave, J., Coomes, D., Jansen, S., Lewis, S, L., Swenson, N, G., Zanne, A, E. (2009) ‘Towards a worldwide wood economics spectrum’ Ecology Letters, Vol 12, pp.351-366.
Coder, K. D, (1996) ‘Identified Benefits of Community Trees and Forests’, University of Georgia Cooperative Extension Service, Forest Resources Publication FOR96-39
Denman, K. L., Brasseur, G., Chidthaisong, A., Ciais, P., Cox, P. M. (2007) ‘Couplings between changes in the climate system and biogeochemistry’ Climate Change 2007: The Physical Science Basis, pp. 663-745.
Dwyer, J. F., McPherson, E. G., Schroeder, H. W., Rowntree, R. (1992) ‘Assessing the Benefits and Costs of the Urban Forest’, Journal of Arboriculture 18(5), pp. 227 – 234.
Escobedo, J., Kroeger, T., Wagner. (2011) ‘Urban forests and pollution mitigation: Analysing ecosystem services and disservices’ Environmental pollution, Vol 159 (8-9), pp. 2078-2087.
Evans, J. R., Poorter, H. (2001) ‘Photosynthetic acclimation of plants to growth irradiance: The relative importance of specific leaf area and nitrogen portioning in maximising carbon gain’, Plant, Cell and Environment, Vol 24, pp. 755-767.
Fajardo, A. (2016) ‘Wood density is a poor predictor of competitive ability among individuals of the same species’ Forest Ecology and Management, Vol 372, pp. 217-225.
Forestry Commission. (2008) Forest Research Pathology Advisory Note (No.7) Problems on Plane Trees. Surrey: Forest Research.
Forestry Commission. (2012) Massaria Disease of Plane Trees. [online image] Available at: http://www.treetree.co.uk/massaria-disease-of-planes.html [Acessed:02 March 2018].
Forestry Commission. (2017) ‘Massaria Disease’. Available at: https://www.forestry.gov.uk/fr/massaria. (Accessed:12th December 2017).
Forestry Commission. (2016) ‘Urban forestry and society’. Available at: http://www.forestry.gov.uk/fr/urbanforestryandsociety. (Accessed: 19th October 2016).
Garzon, M. B., Alia, R., Robson, T.M., Zavala, M. A. (2011) ‘Intra-specific variability and plasticity influence potential tree species distributions under climate change’, Global Ecology and Biogeography, Vol 20, pp.766-778
Goodness, J., Andersson, E., Andersson, P, M, L., Elmqvist, T. (2016) ‘Exploring the links between functional traits and cultural ecosystem services to enhance urban ecosystem management’, Ecological Indicators Vol 70, pp.597-605
Greenwood, G., Ruiz-Benito, P., Martinez-Vilalta, J., Lloret, F., Kitzberger, T., Allen. C. D., Fensham, R., Laughlin, D. C., Kattge, J., Bonisch, G., Kraft, N. J. B., Jump, A.s. (2017). ‘Tree mortality across biomes is promoted by drought intensity, lower wood density and higher specific leaf area’, Ecology Letters, 20, pp. 539-553.
Horticulture Week. (2016) Implications of plane wilt for UK arboriculture are “huge”. [online image] Available at: https://www.hortweek.com/implications-plane-wilt-uk-arboriculture-huge-sacre/arboriculture/article/1402836 [Accessed 02 March 2018].
Heisler, G. M. (1986) ‘Energy savings with trees’ Journal of Arboriculture, 12 (5), pp.113-125.
Hoeber, S., Leuschner, C., Kohler, L., Arias-Aguiler, D., Schuldt, B. (2014). ‘The importance of hydraulic conductivity and wood density to growth performance in eight tree species from a tropical semi dry climate’, Forest Ecology and Management, Vol 330, pp.126-136.
Kattge, J., Diaz, S., Lavorel, I, C., Prentice, P., Leadley, G., Bonisch, G. (2011) ‘TRY – a global database of plant traits’ Global Changes in Biology, Vol 17, (9), pp.2905 – 2935.
Kimball, B. A., Kobayashi, K., Bindi, M. (2002) ‘Responses of Agricultural Crops to Free-Air CO2 Enrichment’ Advances in Agronomy, Vol 77, pp. 293-368.
Kumar, U., Singh, P., Boote, K. J. (2012) ‘Chapter two – Effect of Climate Change Factors on Processes of Crop Growth and Development and Yield of Groundnut (Arachis hypogaea L.)’, Advances in Agronomy Vol 116, pp.41-69.
Kuo, F. E. (2003) ‘The role of Arboriculture in a Healthy Social Ecology’, Journal of Arboriculture, 29(3), pp. 148 – 155.
Kuo, F. E., Sullivan. W. C. (2001) ‘Aggression and Violence in the Inner City – Effects of Environment via Mental Fatigue’, Environment and Behaviour, 33 (4), pp. 543 – 571.
Lambrecht, S.C., Mahieu. S., Cheptou, P. O. (2016) ‘Natural selection on plant physiological traits in an urban environment’, Acta Oecologica, 77, pp. 67-74.
Lavorel, S., Grigulis, K., Lamarque, P., Colace, M. P., Garden, D., Girel, J., Pellet, G., Douzet, R. (2010) ‘Using plant functional traits to understand the landscape distribution of multiple ecosystem services’ Journal of Ecology, Vol 99, (1), pp. 135-147
Liu, Z., Zhu, Y., Li, F., Jin, G. (2017) ‘Non-destructively predicting leaf area, leaf mass and specific leaf area based on a linear mixed-effect model for broad leaf species’ Ecological Indicators, Vol 78, pp.340-350.
Machado, S. R., Rodella, R. A., Angyalossy, V., Marcati, C. R. (2007). ‘Structural variations in root and stem wood of Styrax (Styracacea) from Brazilian forest and cerrado’, IAWA, pp. 173.
McPherson, G., Simpson, J, R., Peper, P, J., Maco, S, E., Xiao, G. (1999) ‘Benefit Cost Analysis of Modesto’s Municipal Urban Forest’, Journal of Arboriculture, Vol 25 (5), pp. 235-248.
Monteiro, M. V., Doick, K. J., Handley, P. (2016) ‘Allometric relationships for urban trees in Great Britain’ Urban Forestry & Urban Greening, 19, pp. 223-236
My landscapes. (2018). London Plane Leaves. [online image]. Available at: https://www.mylandscapes.co.uk. Accessed [1st April 2018].
Nock, C. A., Vogt, R.J., Beisner, B. E. (2016) Functional Traits. Chichester: John Wiley & Sons Ltd.
Nowak, D. J., Crane, D. E. (2001) ‘Carbon storage and sequestration by urban trees in the USA’, Environmental Pollution, 116, pp. 381-389.
Panconesi. (1999) ‘Canker Stain of Plane Trees: A Serious Danger to Urban Plantings in Europe’ Journal of Plant Pathology, Vol 81, (1), pp. 3-15.
Pelleteret, P., Crovadore, J., Cochard, B., Pasche, S., Bovigny, P, Y., Chablais, R., Lefort, F. (2017) ‘Urban London Plane tree dieback linked to fungi in the Botryosphaeriaceae’ Urban Forestry & Urban Greening, Vol 22, pp. 74-83.
Schneider, C. A., Rasband, W. S., Eleceiri, K. W. (2012) ‘NIH Image to ImageJ: 25 years of image analysis’ Nat. Methods, 9, pp. 671-675.
Tbeartravels. (2018). London Plane Providing Shade. [online image]. Available at: https://tbeartravels.com/2014/08/22/northern-germany-dresden-part-2/. Accessed: 1st April 2018].
Thomas, P, A. (2000) Trees Their Natural History 2nd edn. Cambridge: Cambridge University Press.
Tubby, K.V., Perez-Sierra, A. (2015) ‘pests and pathogen threats to plane (Platanus) in Britain, The International Journal of Urban Forestry, 37 (2) pp. 85-98
Ulmer, J., Wolf, K., Backman, D., Tretheway, R., Blain, C., O’Neil-Dunne, J., Frank, L. (2016) ‘Multiple health benefits of urban tree canopy: The mounting evidence for a green prescription’, Health & Place 42, pp. 54 – 62.
Wolf, K. (1998) ‘Urban Forest Values: Economic Benefits of Trees in Cities’, University of Washington College of Forest Resources, Factsheet No. 29.
Woodland Trust (2012) ‘Trees in our towns the role of trees and woodland in managing urban water quality and quantity’. Available at: http://www.woodlandtrust.org.uk/water. Accessed 19th October 2017).
Woodstairs. (2018). How To Evaluate Wood. [online image]. Available at: https://www.woodstairs.com/how-to-evaluate-wood/ [Accessed: 18th February, 2018]
7 Appendices
Appendix A. Data collection methods risk assessment sheets.
Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allRelated Content
All TagsContent relating to: "Ecology"
Ecology is a field of biology that concentrates on the study of the relationships and interactions between living organisms (including humans) and their environments.
Related Articles
DMCA / Removal Request
If you are the original writer of this dissertation and no longer wish to have your work published on the UKDiss.com website then please:




