Literature Review on New Zealand's Thoroughbred Industry
Info: 10344 words (41 pages) Dissertation
Published: 19th Mar 2021
Tagged: Veterinary
LITERATURE REVIEW
NEW ZEALAND THOROUGHBRED INDUSTRY- OVERVIEW:
The New Zealand thoroughbred industry is the largest of the New Zealand equine industry generating about $ 1,635 million in value added contribution to the Gross Domestic Product (GDP) and it represents about 0.9% of the New Zealand real GDP for the year 2008/09 (IER, 2010b). The revenue is generated from the production of horses for racing, exportation of horses, overseas buyer investments, wagering, government wagering tax and returns to owners (IER, 2004).
The thoroughbred industry has two wings, the racing wing represented by the New Zealand thoroughbred racing and the breeding wing represented by the New Zealand thoroughbred breeders association (Fennessy, 2010). The industry has a long tradition of exporting horses and in the last ten years, about 1,500 thoroughbred horses were known to be exported annually as yearlings (Fennessy, 2010).
The table 1 and table 2 provide data on the number of horses racing within the season and data on the scale of the breeding industry.
Table 1: The New Zealand Thoroughbred racing industry. Source: New Zealand Thoroughbred Racing Fact Book (Racing, 2009).
| Season | Horses raced | Races run | Total stakes paid (prize money)
$ NZ million |
Total stakes paid (prize money)
% of 1999-2002 |
| Mean of 1999-2002 | 5,528 | 2,836 | $32.3 | 100 |
| 2002-03 | 5,469 | 2,810 | $35.0 | 108 |
| 2003-04 | 5,564 | 2,791 | $36.2 | 112 |
| 2004-05 | 5,706 | 2,817 | $37.2 | 115 |
| 2005-06 | 5,642 | 2,855 | $39.7 | 123 |
| 2006-07 | 5,566 | 2,863 | $49.4 | 153 |
| 2007-08 | 5,576 | 2,970 | $56.2 | 174 |
| 2008-09 | 5,826 | 3,088 | $58.4 | 181 |
| 2009-10 | NA | NA | $51.7 | 160 |
THE RACING INDUSTRY
The thoroughbred racing industry is governed by the New Zealand Thoroughbred Racing (NZTR). The NZTR is responsible for governance and management of the thoroughbred racing industry including drawing up and delivering its business plan as well as overseeing its funding distribution policy, rules and all integrity and race day functions, race programming and handicapping, registrations including the stud book, communications and industry marketing and education and development (IER, 2010a). More than 47,000 people are actively involved in production and training of more than 30,000 racehorses in New Zealand (IER, 2010a).
There are around 52 racecourses in New Zealand and 65 Racing (R.C) or Jockey clubs (J.C) ((NZTR), 2012). The major races held in New Zealand are the Karaka Million, Spring Classic, New Zealand 1000 Guineas, New Zealand 200 Guineas, New Zealand Cup, Telegraph Handicap, Wellington Cup, Thorndon Mile, Auckland Cup, New Zealand Derby and the New Zealand Oaks.
The racing division of the thoroughbred industry is responsible for generating more than $1,093 million in value added or 73% of the racing industry total with the Auckland, Waikato, and Manawatu- Wanganui regions generate the largest amount among the thoroughbred racing industry (IER, 2004) and this is depicted in Table 3.
Table 3: Value added contribution to Gross Domestic Product
| Value Added Generated by Racing | Initial | Total |
| Auckland region | $ 89,770,802 | $ 314,960,731 |
| Bay of Plenty Region | $ 8,663,168 | $ 30,000,389 |
| Canterbury Region | $ 26,066,466 | $ 91,190,570 |
| Gisborne Region | $ 764,548 | $ 2,678,851 |
| Hawkes bay Region | $ 9,543,967 | $ 33,237,498 |
| Manawatu- Wanganui Region | $ 36,093,153 | $ 126,370,247 |
| Marlborough Region | $ 394,867 | $ 1,374,817 |
| Northland Region | $ 4,572,578 | $ 16,008,366 |
| Otago region | $ 8,349,526 | $ 29,186,804 |
| Southland Region | $ 7,219,838 | $ 25,190,498 |
| Taranaki Region | $ 9,762,991 | $ 34,072,247 |
| Tasman- Nelson Region | $ 452,058 | $ 1,593,484 |
| Waikato Region | $ 83,141,394 | $ 291,944,263 |
| Wellington Region | $ 26,377,723 | $ 92,162,585 |
| West Coast Region | $ 1,046,372 | $ 3,621,551 |
| Total value Added Generated | $ 312,219,452 | $ 1,093,592,900 |
Table 2 provides data on the number of horses racing within the season.
Table 2: The New Zealand Thoroughbred racing industry. Source: New Zealand Thoroughbred Racing Fact Book (Racing, 2009).
| Season | Horses raced | Races run | Total stakes paid (prize money)
$ NZ million |
Total stakes paid (prize money)
% of 1999-2002 |
| Mean of 1999-2002 | 5,528 | 2,836 | $32.3 | 100 |
| 2002-03 | 5,469 | 2,810 | $35.0 | 108 |
| 2003-04 | 5,564 | 2,791 | $36.2 | 112 |
| 2004-05 | 5,706 | 2,817 | $37.2 | 115 |
| 2005-06 | 5,642 | 2,855 | $39.7 | 123 |
| 2006-07 | 5,566 | 2,863 | $49.4 | 153 |
| 2007-08 | 5,576 | 2,970 | $56.2 | 174 |
| 2008-09 | 5,826 | 3,088 | $58.4 | 181 |
| 2009-10 | NA | NA | $51.7 | 160 |
The total number of starts has risen by 2,465 over the last four years (IER, 2010a) which is depicted in chart 1 as shown below
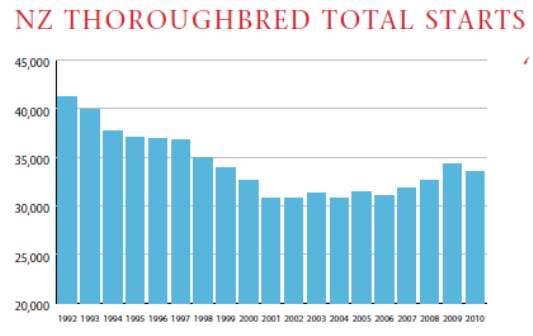
The success of the racing industry depends on the number of participants. The thoroughbred racing industry supports around 23,000 participants at various levels of employment with many of the participants undertaking more than one role (IER, 2004). The following table 3 depicts the participants in the thoroughbred racing industry (IER, 2004).
| Participant type | Numbers |
| Number of Breeders | 3, 156 |
| Number of breeders staff | 3,855 |
| Number of owners & syndicate owners | 6,664 |
| Number of breeders/ owners | 2,801 |
| Number of owners/ trainers | 696 |
| Number of public trainers | 361 |
| Number of permits to train holders | 379 |
| Number of track riders | 234 |
| Number of stablehands | 37 |
| Total participants in producing racing animal | 18,183 |
| Number of jockeys | 156 |
| Number of apprentice jockeys | 83 |
| Number of industry admin staff | 75 |
| Number of club secretaries | 65 |
| Number of committee members | 843 |
| Number of full-time race club staff | 506 |
| Number of part time & contractor staff | 1,249 |
| Number of volunteer race club staff | 996 |
| Number of race day Drs & Vets | 174 |
| Number of race club wagering staff | 601 |
| Number of TAB off- course staff | 408 |
| Total participants in producing the racing product | 5,156 |
| Total number of participants in the racing industry | 23,339 |
Table 4 depicts the number of licenses from the year 2007 to 2010. There are 22.4% less trainers registered with NZTR now than there were in 1999/ 2000 (IER, 2010a).
| Number of Licenses | 2007/08 | 2008/09 | 2009/10 |
| Public trainers | 353 | 374 | 370 |
| Permit to train | 372 | 413 | 409 |
| Owner trainer | 437 | 437 | 458 |
| Total number of trainers | 1162 | 1224 | 1237 |
| Jockeys | 112 | 131 | 113 |
| Apprentice Jockeys | 69 | 53 | 47 |
| Amateur riders | 26 | 43 | 41 |
| Total number of riders | 207 | 227 | 221 |
| Stablehands (non riding) | 570 | 709 | 744 |
| Stablehands (riding) | 421 | 506 | 528 |
| Trackriders | 133 | 151 | 175 |
| Total | 1124 | 1366 | 1447 |
THOROUGHBRED RACING INDUSTRY IN THE INTERNATIONAL STAGE
The New Zealand thoroughbred industry stands at the eleventh place in the international stage in terms of the number of horses starting in races each season and around eighth place in the number of foals produced (Fennessy, 2010). Table 4 gives an international comparison of the top thirteen thoroughbred racing and breeding industries (Fennessy, 2010).
Table 4: Comparison of the scale of international thoroughbred racing for 2007 with prize- money for 2008 (Fennessy, 2010).
| Country | Number of foals | Starters in flat races | Flat races run | Total prize money ($ US million, 2008) |
| USA | 36,071 | 67,930 | 51,132 | $1,165 |
| Japan | 7,516 | 24,143 | 17,476 | $1,220 |
| Australia | 18,255 | 31,419 | 19,382 | $421 |
| France | 5,384 | 8,726 | 4,156 | $335 |
| Great Britain | 5,839 | 11,323 | 5,659 | $158 |
| Canada | 2,632 | 7,482 | 5,057 | $127 |
| Hong Kong | Nil | 1,154 | 726 | $98 |
| Ireland | 12,633 | 3,458 | 958 | $84 |
| Argentina | 7,538 | 11,123 | 6,101 | $64 |
| Singapore | Nil | 1,099 | 704 | $41 |
| New Zealand | 4,338 | 5,489 | 2,734 | $40 |
| South Africa | 3,246 | 6,567 | 3,883 | $31 |
| Macau | Nil | 768 | 737 | $23 |
New Zealand-bred horses have been known to perform well on the international stage and thus there is always an increase in demand for them. New Zealand-bred horses have won 84 stake races in Australia in 2009/10, 6 better than the previous season and 3 better than the decades previous best of 81, achieved in 2000/01 season (IER, 2010a). Table 5 provides data in international New Zealand-bred group one winners in 2009/10 (IER, 2010a).
Table 5: International New Zealand-bred group one winners in 2009/10 (IER, 2010a).
| Country | Name of the horse |
| Hong Kong | Super Satin, Beauty Flash, Able One, Fellowship |
| Singapore | Race Ahead, Green Birdie, Intercept |
| South Africa | Gold Onyx |
| Australia | Efficient, Jessicabeel, Metal Bender, Miss Keepsake, Monaco Consul, More Joyous, Rangirangdoo, So You Think, Velocitea, Zabrasive |
THE BREEDING INDUSTRY
The thoroughbred breeding industry is governed by the New Zealand Thoroughbred Breeders’ Association (NZTBA). The first general meeting of the NZTBA was held in Wellington on March 16, 1948, and the first annual meeting was held on January 25, 1949 (NZTB, 2016). New Zealand ranks eighth in the number of foals produced (Fennessy, 2010).The stud book department is a part ofNZTR and it is involved in broodmare and stallion registrations, foaling returns, registering ownership for breeding and foal identification.
The breeding season by the industry in New Zealand is from September 1 to November 30 and this is done with the aim to have foals born as early in the season as possible so they are larger when sold as yearlings and thus cam maximise sales price but this season does not mirror the physiological breeding season (Waldron, Rogers, Gee, & Bolwell, 2011); (Hanlon, Stevenson, Evans, & Firth, 2012). Restrictions on the use of artificial insemination and imposition of a short commercial breeding season has a great impact on the reproductive efficiency of Thoroughbred mares bred in New Zealand (CW Rogers, 2016).
In the last 20 years there has been a phenomenon called consolidation that occurred in the New Zealand thoroughbred industry with the national broodmare herd decrease from 10,176 in 1989 to 6,488 in 2004 which is a 36% reduction in the number of mares being bred and a 45% reduction in the annual foal crop (CW Rogers, Gee, & Vermeij, 2009). There is also a reduction in the number of thoroughbred stallions standing at stud during this past 20 years and this reduction was found to be associated with greater sire efficiency within the industry with an annual increase in the number of mares covered by each stallion and thus providing support for the consolidation being associated with attempts to improve economic and reproductive performance (CW Rogers et al., 2009).
Table 6 provides data on the number of registered thoroughbred stallions, registered thoroughbred broodmares and thoroughbred foals present in New Zealand during different years. From the data provided in Table 6 we can conclude that there is a reduction (approximate) in the number of thoroughbred horses in New Zealand.
Table 6: Number of registered thoroughbred stallions, registered thoroughbred broodmares and thoroughbred foals present in New Zealand (IER, 2004); (IER, 2010b); (IER, 2010a).
| Type | 2004/05 | 2005/06 | 2006/07 | 2007/08 | 2008/09 | 2009/10 |
| Registered broodmares | 7012 | 7498 | 7332 | 7470 | 8326 | 7098 |
| Total mares served | 6872 | 6558 | 6623 | 6757 | 6483 | 6488 |
| Stallions | 204 | 196 | 201 | 177 | 166 | 167 |
| Foals | 4596 | 4518 | 4349 | 4126 | 4224 | 4132* |
*At the time of publication
Table 7 provides data on the New Zealand thoroughbred breeding, sales and exports statistics from 1995-2015
| Season | Stallions covering 10 or more mares | Mares served | Foals | Horses exported | Est. value of exports | NZ yearling sales aggregate |
| 1994-95 | 190 | 8587 | 5264 | 2065 | $30.1 million | |
| 1995-96 | 176 | 8340 | 5219 | 1987 | $29.1 million | |
| 1996-97 | 179 | 7947 | 5161 | 2027 | $31.2 million | |
| 1997-98 | 160 | 7718 | 4874 | 1827 | $44.4 million | |
| 1998-99 | 146 | 7224 | 4993 | 2175 | $48.7 million | |
| 1999-00 | 268 | 7316 | 4849 | 1937 | $110 million | $71.7 million |
| 2000-01 | 256 | 7738 | 4958 | 2000 | $115 million | 68.3 million |
| 2001-02 | 244 | 7611 | 5082 | 1914 | $115 million | $68.3 million |
| 2002-03 | 229 | 7204 | 5060 | 1763 | $115 million | $47.2 million |
| 2003-04 | 218 | 6903 | 4683 | 1797 | $115 million | $60.4 million |
| 2004-05 | 204 | 6848 | 4509 | 1803 | $120 million | $69.6 million |
| 2005-06 | 192 | 6558 | 4522 | 1831 | $125 million | $65.7 million |
| 2006-07 | 201 | 6623 | 4349 | 1888 | $130 million | $81.4 million |
| 2007-08 | 177 | 6757 | 4126 | 1670 | $145 million | $113.4 million |
| 2008-09 | 170 | 6349 | 4409 | 1578 | $130 million | $ 75 million |
| 2009-10 | 167 | 6493 | 4207 | 1465 | $140 million | $93.6 million |
| 2010-11 | 164 | 6103 | 4039 | 1607 | $150 million | $88.1 million |
| 2011-12 | 150 | 5826 | 3927 | 1601 | $135 million | $77.3 million |
| Season | Stallions covering 10 or more mares | Mares served | Foals | Horses exported | Est. value of exports | NZ yearling sales aggregate |
| 2012-13 | 140 | 5533 | 3828 | 1511 | $130 million | $72.3 million |
| 2013-14 | 141 | 5576 | 3469 | 1486 | $69.7 million | |
| 2014-15 | 132 | 5300 (est.) | 3550 (est.) | 1480 | $67.4 million |
NEW ZEALAND THOROUGHBRED PRODUCTION CYCLE, SALES & EXPORTS
The thoroughbred production in New Zealand operates on two levels with one focussing on the production of horses for the domestic market and the other focusses on the production of horses for export (CW Rogers, Gee, & Bolwell, 2014). There is a reduction in the production of individuals for the domestic market and the reason for this are the changes in the selection practices of thoroughbred broodmares over three generation intervals, short breeding season, longer gestation period, reduction in number of thoroughbred sires standing at stud and improper reproductive management of popular stallions at stud (CW Rogers et al., 2014).
The national yearling sales consist of four categories and they are held at the National sales complex at Karaka, Auckland. The four categories are premier yearling sale, select yearling sale, festival sale and carnival sale. Table 8 summarizes data for exports and the sales receipts from the national yearling sales series held each year in January- February (Fennessy, 2010).
Table 8: Data for exports and sales receipts from the national yearling sales series held each year in January- February. (Fennessy, 2010).
| Season | Number of horses exported | Estimated value of exports ($ NZ million) | New Zealand yearling sales aggregate ($ NZ million) | Average ( and median) prices of yearlings ($ NZ 000s) |
| 2001-02 | 1,914 | $115 | $55 | |
| 2002-03 | 1,763 | $115 | $47 | |
| 2003-04 | 1,782 | $115 | $60 | |
| 2004-05 | 1,806 | $120 | $70 | |
| 2005-06 | 1,831 | $125 | $66 | |
| 2006-07 | 1,888 | $130 | $81 | $79 ($50) |
| 2007-08 | 1,670 | $145 | $113 | $95 |
| 2008-09 | 1,578 | $130 | $75 | $63 ($30) |
| 2009-10 | NA | NA | $94 | $83 ($42) |
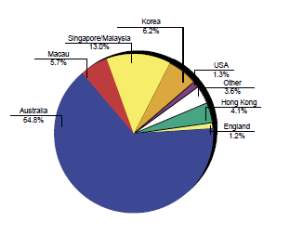
New Zealand is known to produce good quality Thoroughbreds that compete very well on the international stage with recent successes of New Zealand bred horses winning a number of group one races; including races in Honk Kong, Singapore, Dubai and Australia(IER, 2010b); (Fennessy, 2010).The export market has always remained stable (CW Rogers et al., 2014) and it is the feature of the New Zealand thoroughbred industry, with the local breeding industry only being sustainable as a result of the international demand for New Zealand horses, with 62% of exports being shipped to Australia, 11 % to Hong Kong and 14 % to Singapore in 2007-08 season (Fennessy, 2010). The New Zealand Thoroughbred industry relies more on international buyers to sustain market values, with 65 % of the total yearling sales revenue originating from overseas buyers (CW Rogers et al., 2009); (IER, 2010b).
Chart 2 depicts the value of Thoroughbred sold at auction by residence of purchaser for the year 2004 (IER, 2004).
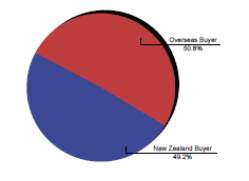
Chart 3 depicts the share of thoroughbred market internationally for the year 2004 (IER, 2004).


Sire and dam mating
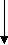
Birth of a foal

Weanlings

Yearling- sales
Breeding population
Mostly females and few males
 ————————————-
————————————-
Pre training
Ready to run 18 months’ old
Race training 2yrs
 ————————————
————————————

Race training 3yrs old
 ———————————-
———————————-
4yrs & older training/ racing
 ———————————–
———————————–
Export as future race horses or stud animals, Sale due to poor performance, Recreation/ sport horse. Meat works
 ———————————————————————-
———————————————————————-
Figure 2: The New Zealand thoroughbred production cycle
FACTORS AFFECTING OESTROUS CYCLE
Horses are seasonally polyestrous animals. The reproductive pattern which is seasonal is the result of a circannual endogenous rhythm that is governed by external factors such as photoperiod, temperature, nutrition and body condition (Nagy, Guillaume, & Daels, 2000).
PHOTOPERIOD:
Melatonin is a hormone which is mostly produced during the night time, is secreted by the pineal gland (Walsh, Prendergast, Sheridan, & Murphy, 2013). In seasonally breeding mammals like the horse, melatonin seems to play a major role in controlling the reproductive cycles (Karsch et al., 2013); (Sharp, 1980); (Burkhardt, 1947). The reproductive system depends on the recognition of the shortened duration of melatonin secretion as the longer days of spring approaches (Walsh et al., 2013). A reduced duration of melatonin is stimulatory and is found to decrease the inhibition of gonadotropin releasing hormone (GnRH) pulse frequency and thus it helps in activating the annual reproductive phase (Cleaver et al., 1990).
The production of melatonin can be manipulated with the help of artificial photoperiod and thus photoperiod is the most important external factor that can play a major role on the circannual endogenous reproductive rhythm (Ginther, 1992). Studies have shown that exposing mares to artificial light during the winter and early spring can hasten the onset of ovarian activity (Burkhardt, 1947). The appropriate response to this artificial photoperiod is not always directly proportional to exposure to long days (Nagy et al., 2000) and there are factors that enable photoperiod to influence the circannual reproductive rhythm and they are: (a) State of refractoriness to photoperiodic changes (b) the photoperiodic history (c) the existence of a photosensitive phase during the night (Nagy et al., 2000).
Mares will continue with their circannual rhythm irrespective of their stimulatory or inhibitory effects of the lighting conditions and this is called as photoperiodic refractoriness. It is difficult to completely eliminate this effect and this is supported by different studies.
Photoperiodic history shows the animals ability to respond to a specific photoperiod (Nagy et al., 2000). Studies carried out in ewes have shown that the artificial photoperiod is stimulatory if the animals were exposed to a 13 hour light if preceded by a 16 hour light photoperiod and the effect in inhibitory if preceded by a 10 hour light period (Robinson & Karsch, 1987). Such studies have not been carried out in horses so far (Nagy et al., 2000).
Studies have shown that mares have a photosensitive phase during the period of darkness. When anestrous mares were exposed to lights for 1-2 hours of light after 8 or 9.5 hours of darkness, it was found out that there was a stimulation in follicular growth (Palmer, Driancourt, & Ortavant, 1981); (Malinowski, Johnson, & Scanes, 1985); (Guillaume, Duchamp, Nagy, & Palmer, 2000). and this method works by interrupting the melatonin production by the pineal gland and therefore modulates the inhibitory effect of melatonin on GnRH production (Palmer & Guillaume, 1992).
The general recommendations for artificial photoperiod treatment are as follows (McCue, Logan, & Magee, 2007):
- A fixed length period of light exposure for 15-16 hours, allowing 8-9 hours of darkness may be used.The duration of light exposure is not necessary to be increased incrementally.
- Alternatively, adding 2.5-6 hours of additional light exposure after dusk was found to be effective. Automatic timers may be programmed to turn lights on at sunset and off at 2230-2300 hours.
- Light treatment should be initiated during the first week of December if the breeding operations are to begin in February.
- 100 lux should be the minimum light intensity to be used.
Administering artificial photoperiod to mares has its own management costs.A recent study was carried out where it was shown that blue light from light emitting diodes can suppress melatonin production in horses (Walsh et al., 2013). In this study, the mares were exposed to a light source from a light emitting diode which was directed either at a single or both eyes and blood samples were collected to analyze the melatonin levels. It was found out that production of melatonin can be inhibited by exposing a single eye to low wavelength blue light. Thus a new management technique can be developed where horses can be fitted with a light emitting device which will help in achieving the required artificial photoperiod while the horses are maintained outdoors, and this method reduces the management costs and improves the health and welfare of the horses (Walsh et al., 2013).
TEMPERATURE:
Not many studies have been carried out to understand the effects of environmental temperature on equine reproduction. A 10-year retrospective analysis of breeding records of a thoroughbred farm in Australia has shown that there was a notable difference between years in the time of the first ovulation. In this study, the minimum and maximum environmental temperatures in the weeks immediately prior to the first ovulation were similar in all years of the study thus enabling us to come to a conclusion that environmental temperature does play a role in the onset of reproductive activity (Guerin & Wang, 1994).
NUTRITION & BODY CONDITION:
Nutrition and body condition can play a major role on seasonal reproduction (Donadeu & Watson, 2007); (Nagy et al., 2000). The nutritional appropriateness of a broodmare can be assessed by using the Henneke body condition score (scale from 1 to 9) which is a subjective assessment method of body fat level (Morley & Murray, 2014). It is based on visual appraisal and palpable fat cover at six areas of the horse’s body (Gentry et al., 2004) as shown in the diagram below
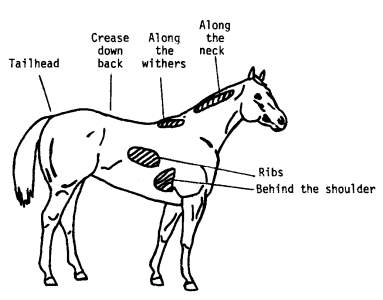
Diagram of areas palpated to estimate body fat and condition score (Gentry et al., 2004).
Certain key terms are used in the body condition score system and they are follows (“BCS: What body condition scores mean,” 2006):
- Crease- A gutter over the spine created by fat buildup on either side of the bone.
- Hooks- The pelvic hip bones that jut out to the side of the horse’s rump.
- Pins- Pelvic bones near the tail that poke out at the back of the horse’s rump.
- Spinous processes- Bony protrusions at the top of the vertebrae of the torso.
- Tailhead- The root of the tail where it blends in with the body; highest movable point on the tail.
- Transverse processes- Bony protrusions on each side of the vertebrae.
Description of individual condition scores (Henneke, Potter, Kreider, & Yeates, 1983)
| Score | Description |
| 1 (poor) | The animal is extremely emaciated, spinous processes, ribs, tail- head, and hooks and pins are prominent. Bony structures of withers, shoulders, and neck are easily noticeable. No fatty tissues can be felt |
| 2 (Very thin) | The animal is emaciated. A thin layer of fat over the base of spinous processes. Transverse processes of lumbar vertebrae feel rounded.
Spinous processes, ribs, tailhead, hooks, and pins are prominent. , shoulders and neck structures are faintly discernable |
| Score | Description |
|
Fat about halfway up spinous processes; transverse processes cannot be felt. Thin fat layer over ribs. Spinous processes and ribs are easily discernable. Tailhead prominent, but individual vertebrae cannot be visually identified. Hook bones appear rounded but not easily discernable. Pin bones not distinguishable. Withers, shoulders, and neck are accentuated. |
|
Ridge along the back. Faint outline of ribs discernible. Tailhead prominence depends on conformation; fat can be felt around it. Hook bones not discernable. Withers, shoulders, and neck are not obviously thin. |
|
Back is level. Ribs cannot be visually distinguished but can be felt easily. Fat around tailhead beginning to feel spongy. Withers appear rounded over the spinous processes. Shoulders and neck blend smoothly into the body. |
|
May have slight crease down back. Fat over ribs feels soft and spongy. Fat around tailhead feels soft. Fat beginning to be deposited along the sides of the withers, behind the shoulders and along the sides of the neck. |
|
May have crease down back. Individual ribs can be felt with noticeable filling between ribs with fat. Fat around tailhead is soft. Fat deposited along withers, behind shoulders, and along the neck. |
|
Crease down back. Difficult to feel ribs. Fat around tailhead very soft. The area along withers filled with fat. The area behind shoulder filled in flush. Noticeable thickening of the neck. Fat deposited along inner buttocks. |
|
Obvious crease down back. Patchy fat appearing over ribs. Bulging fat around tailhead, along withers, behind shoulders and along the neck. Fat along inner buttocks may rub together. Flank filled in flush. |
In a study carried out by Henneke, he found out that mares with a BCS of less than 5.0 when compared to mares with a BCS above 5.0 had a significantly longer average interval to first ovulation (Henneke, Potter, & Kreider, 1984).
The nutrient levels in the feed play a major role in controlling reproduction. Mares whose diets were supplemented with concentrates were found to ovulate earlier after the winter anestrous period (C. Van Niekerk & Van Heerden, 1972). A study was carried out understand the relationship between energy intake and body condition on the reproductive performance of nonpregnant mares and it was found out that a high- energy intake can shorten the interval to first ovulation in transitional mares with a low level of body fat but does not benefit mares in moderate or fat condition (Kubiak, Crawford, Squires, Wrigley, & Ward, 1987). Apart from energy content, the quality of dietary protein in the diet can influence the mares breeding season. A study was carried out to understand the effects of dietary protein on reproduction in the mare and it was found that mares ovulated approximately 3-6 weeks earlier when their diet contained high-quality protein when compared to mares on low- quality dietary protein (F. Van Niekerk & Van Niekerk, 1997).
Pasturing has a positive effect on the ovarian activity of mares (Nagy et al., 2000). The beneficial effects of pasturing on equine estrous activity was demonstrated in a study where it was found out that anestrous mares who were pastured on green grass from early May ovulated sooner in the spring than mares housed on a dry lot and feed hay (Carnevale, Hermenet, & Ginther, 1997).
AGE:
The reproductive efficiency of mares depends on its age. A study was carried out to understand the relationships of age to uterine function and reproductive efficiency in mares. In this study, the mares were divided into two age groups: young (5 to 7 years) and old (≥ 15 years). It was found out that in older mares there was an increase in endometrial inflammation, reduced pregnancy rate, increased embryo loss rate and increased the duration in fixation of the embryonic vesicle (Carnevale & Ginther, 1992).
Follicular activity is usually less in older mares (≥ 14 years) than in young mares( 5 to 7 years) during the transition period (Carnevale et al., 1997). A depletion of primordial follicles and an associated reduction in the number of viable follicles can be a possible reason for the reduced follicular activity in older mares (Carnevale, Bergfelt, & Ginther, 1994).
Age can also play a role on multiple ovulation in mares and it is backed by a study that was carried out to determine the effect of age on ovulation rate and distribution in thoroughbred mares (Morel & O’Sullivan, 2001). 828 mares were grouped into six age groups: A (3-5 years), B (6-8 years), C (9-11 years), D(12-14 years), E (15-18 years), F(18-22 years). The distribution of ovulation between the left and right ovary was not affected by age group, but the incidence of multiple ovulations increased with age.
REPRODUCTIVE PHYSIOLOGY OF THE MARE
The mare is a seasonal long day breeder with the onset of breeding season occurring in spring and this is associated with an increase in daylight, temperature and availability of pasture (Nagy et al., 2000).The natural breeding season occurs from April to October in the Northern Hemisphere and from October to March in the Southern Hemisphere. The breeding season imposed by the industry in New Zealand is from September 1 to November 30 and this is done with the aim to have foals born as early in the season as possible so they are larger when sold as yearlings and thus can maximize sale price but this season does not mirror the physiological breeding season (Waldron et al., 2011). Restrictions on the use of artificial insemination and imposition of a short commercial breeding season have a great impact on the reproductive efficiency of Thoroughbred mares bred in New Zealand (CWW Rogers, Gee, & Bolwell, 2016).
The Oestrous Cycle:
The average length of the estrous cycle in mares during the breeding season is about 21 to 22 days with 5 to 7 days of estrous (Aurich, 2011). The length of diestrous remains constant at 14 to 15 days irrespective of the change in season. The estrous cycle consists of two phases and they are the follicular phase and luteal phase (Brinsko, Blanchard, Varner, Schumacher, & Love, 2011). During the follicular phase, the mare is sexually receptive to the stallion and the reproductive tract is prepared to accept and transport spermatozoa and ovulation occurs. The luteal phase is characterized by a period where the mare is not receptive to the stallion, and the genital tract is ready to accept and nurture the conceptus.
Endocrine regulation of oestrous cycle:
The estrous cycle relies on hormones produced by the pineal gland, hypothalamus, pituitary gland, ovaries and endometrium (Brinsko et al., 2011). A simplified version of the hormonal regulation of estrous in the mare is shown in the image below. The hypothalamus produces Gonadotropin Releasing Hormone (GnRH) which is carried to the anterior pituitary via the hypothalamohypophyseal portal system. According to the different stages of the estrous cycle the GnRH pulse frequency changes and it is low during the luteal phase with a GnRH pulse lasting for approximately 120 minutes and on the day of ovulation, the GnRH pulse frequency lasts for approximately 30 minutes (Aurich, 2011).
The pituitary then produces Follicle Stimulating Hormone (FSH) and Leutinizing Hormone (LH). The equine pituitary contains gonadotroph cells that store and produces FSH or LH and they are two types the mono hormonal gonadotrophs and bihormonal gonadotrophs (Aurich, 2011). The heterogenicity in the pattern of LH and FSH storage within the gonadotroph population is responsible for the morphological basis for the differential regulation of LH and FSH secretion throughout the equine reproductive cycle (Tortonese et al., 2002). FSH is involved in recruiting follicles and LH is responsible for follicular maturation and production of estrogen, ovulation and luteinization.
FSH and LH then reach the ovaries via the systemic circulation. The maturing follicles
produce estrogen and the estrogen produced especially by the dominant follicle has a stimulatory effect and causes further release of LH. Once ovulation occurs there is a formation of corpus luteum (CL). The CL produces progesterone which inhibits or has a negative feedback on the release of LH from the anterior pituitary.
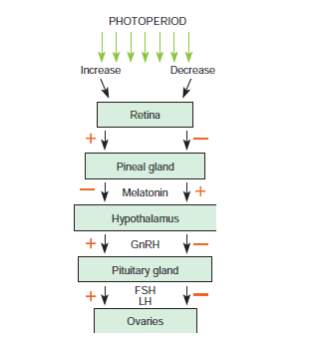
A simplified version of the hormonal regulation of the estrous cycle of the mare (Brinsko et al., 2011). The regular pattern of the estrous cycle of the mare is controlled by the interplay among the pineal gland, hypothalamus, pituitary, ovaries and endometrium. The role of the endometrium is depicted in the image below
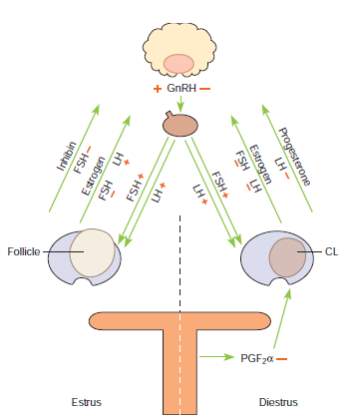
Schematic depiction of the hormonal control of ovarian activity in the mare (Brinsko et al., 2011).
FOLLICULAR PHASE:
The follicular phase is characterized by follicular growth and estrogen production and the mare exhibits behavioral signs of estrous. In the equine estrous cycle one or two distinct follicular waves can occur (Aurich, 2011). The first major follicular wave can occur during the early luteal phase which is usually anovulatory but sometimes ovulation does occur and this phenomenon is considered unique to the equine species (Aurich, 2011).
When the largest follicle reaches a size of about 13 mm in diameter the FSH reaches a plateau (Gastal, Gastal, Bergfelt, & Ginther, 1997). Later the concentration of FSH starts to reduce and it reaches a level where it is not able to support further growth of subordinate follicles but the level is sufficient to help in the growth of the dominant follicle and this phenomenon is called as follicle deviation or selection (Aurich, 2011). The largest and the second largest follicles have an average size of 22 and 19 mm respectively during time of follicle deviation and this stage of the dominant follicle is considered the decisive developmental stage and there is rapid activation of deviation mechanisms that block further development of the second largest follicle to a similar decisive size (Ginther, 2000).
The dominant follicle inhibits the further growth of smaller follicles by the production of estrogen and inhibin (Gastal, Gastal, & Ginther, 1999). LH helps in further growth of the dominant follicle after the beginning of follicular deviation (Ginther, 2000). The dominant follicle is now called the preovulatory follicle and it grows at a rate of 3 mm per day in diameter to 35 mm four days before ovulation and once they reach 40 mm at about 2 days before ovulation the follicular size reaches a plateau (Ginther, Gastal, Gastal, & Beg, 2008). There are also reports that the follicles can reach a size of 55 mm or more (Cuervo-Arango & Newcombe, 2008).
Most mares ovulate within 48 hours before the end of estrous; occasionally mares ovulate after the endo of estrous (Brinsko et al., 2011). Once the follicle ovulates the follicular fluid drains into the peritoneal cavity and the hormones present in the fluid is rapidly absorbed into the circulation and this is responsible for the increase in the concentration of inhibin on the day of ovulation (Bergfelt, Mann, Schwartz, & Ginther, 1991).
LUTEAL PHASE:
After ovulation occurs there is the formation of corpus luteum. This corpus luteum enlarges within the internal part of the ovary due to the peculiar anatomy in horses and they are pear like in shape and contain small compartments with rough surface textures (Kimura et al., 2005). The luteal cells in horses originate from the granulosa cells of the preovulatory follicle (C. Van Niekerk, Morgenthal, & Gerneke, 1975).
At the time of ovulation there is a sudden increase in the concentration of progesterone (da Costa, Branco, Pessa, Silva, & Ferreira-Dias, 2005), with maximal concentrations reached on the 8th day after ovulation and there is a decrease in the level of progesterone until the onset of luteolysis that begins approximately on the 14th day (Aurich, 2011) .
In the mare luteolysis occurs at around day 15-17 of the cycle and it is characterised by a marked decrease in circulating levels of progesterone (Aurich, 2011) accompanied with progressive decrease in the mean cross- sectional area of the corpus luteum from day 4 to day 19 of the cycle (Ginther, Gastal, Gastal, Utt, & Beg, 2007). The hormone responsible for luteolysis is PGF2α which is produced by the equine endometrium during the late diestrous phase and the PGF2α unlike in other species is secreted into the peripheral circulation making the equine corpus luteum highly sensitive to the hormone (Aurich, 2011). Studies have shown that the release of PGF2α in horses is stimulated by the oxytocin which is produced by specific secretory endometrial cells (Bae & Watson, 2003).
REFERENCES
(NZTR), N. Z. T. R. I. (2012). Clubs and Courses. Retrieved from https://www.nzracing.co.nz/RaceInfo/Clubs-And-Courses.aspx
Aurich, C. (2011). Reproductive cycles of horses. Animal Reproduction Science, 124(3), 220-228.
Bae, S.-E., & Watson, E. D. (2003). A light microscopic and ultrastructural study on the presence and location of oxytocin in the equine endometrium. Theriogenology, 60(5), 909-921.
BCS: What body condition scores mean. (2006). Equus, 347.
Bergfelt, D., Mann, B., Schwartz, N., & Ginther, O. (1991). Circulating concentrations of immunoreactive inhibin and FSH during the estrous cycle of mares. Journal of Equine Veterinary Science, 11(6), 319-322.
Brinsko, S. P., Blanchard, T. L., Varner, D. D., Schumacher, J., & Love, C. C. (2011). Manual of Equine Reproduction (Third ed.): Elsevier Health Sciences.
Burkhardt, J. (1947). Transition from anoestrus in the mare and the effects of artificial lighting. The Journal of Agricultural Science, 37(01), 64-68.
Carnevale, E., Bergfelt, D., & Ginther, O. (1994). Follicular activity and concentrations of FSH and LH associated with senescence in mares. Animal Reproduction Science, 35(3-4), 231-246.
Carnevale, E., & Ginther, O. (1992). Relationships of age to uterine function and reproductive efficiency in mares. Theriogenology, 37(5), 1101-1115.
Carnevale, E., Hermenet, M., & Ginther, O. (1997). Age and pasture effects on vernal transition in mares. Theriogenology, 47(5), 1009-1018.
Cleaver, B., Grubaugh, W., Davis, S., Sheerin, P., Franklin, K., & Sharp, D. (1990). Effect of constant light exposure on circulating gonadotrophin levels and hypothalamic gonadotrophin-releasing hormone (GnRH) content in the ovariectomized pony mare. Journal of reproduction and fertility. Supplement, 44, 259-266.
Cuervo-Arango, J., & Newcombe, J. (2008). Repeatability of preovulatory follicular diameter and uterine edema pattern in two consecutive cycles in the mare and how they are influenced by ovulation inductors. Theriogenology, 69(6), 681-687.
CW Rogers, E. K. G. C. F. B. (2016). Reproductive production constraints within the New Zealand racing industry. Proceeding of the New Zeealand Society of Animal Production, 76, 146-150.
da Costa, R. R., Branco, V., Pessa, P., Silva, J. R., & Ferreira-Dias, G. (2005). Progesterone receptors and proliferating cell nuclear antigen expression in equine luteal tissue. Reproduction, Fertility and Development, 17(6), 659-666.
Donadeu, F., & Watson, E. (2007). Seasonal changes in ovarian activity: lessons learnt from the horse. Animal Reproduction Science, 100(3), 225-242.
Fennessy, P. (2010). An overview of the New Zealand thoroughbred industry. Paper presented at the Proceedings of the New Zealand Society of Animal Production.
Gastal, E., Gastal, M., Bergfelt, D., & Ginther, O. (1997). Role of diameter differences among follicles in selection of a future dominant follicle in mares. Biology of Reproduction, 57(6), 1320-1327.
Gastal, E., Gastal, M., & Ginther, O. (1999). Experimental assumption of dominance by a smaller follicle and associated hormonal changes in mares. Biology of Reproduction, 61(3), 724-730.
Gentry, L. R., Thompson, D. L., Gentry, G. T., Del Vecchio, R. P., Davis, K. A., & Del Vecchio, P. M. (2004). The relationship between body condition score and ultrasonic fat measurements in mares of high versus low body condition. Journal of Equine Veterinary Science, 24(5), 198-203.
Ginther, O. (1992). Reproductive biology of the mare: basic and applied aspects.
Ginther, O. (2000). Selection of the dominant follicle in cattle and horses. Animal Reproduction Science, 60, 61-79.
Ginther, O., Gastal, E., Gastal, M., & Beg, M. (2008). Dynamics of the equine preovulatory follicle and periovulatory hormones: what’s new? Journal of Equine Veterinary Science, 28(8), 454-460.
Ginther, O., Gastal, E., Gastal, M., Utt, M., & Beg, M. (2007). Luteal blood flow and progesterone production in mares. Animal Reproduction Science, 99(1), 213-220.
Guerin, M., & Wang, X. (1994). Environmental temperature has an influence on timing of the first ovulation of seasonal estrus in the mare. Theriogenology, 42(6), 1053-1060.
Guillaume, D., Duchamp, G., Nagy, P., & Palmer, E. (2000). Determination of minimum light treatment required for photostimulation of winter anoestrous mares. JOURNAL OF REPRODUCTION AND FERTILITY-SUPPLEMENT-, 205-216.
Hanlon, D., Stevenson, M., Evans, M., & Firth, E. (2012). Reproductive performance of Thoroughbred mares in the Waikato region of New Zealand: 1. Descriptive analyses. New Zealand Veterinary Journal, 60(6), 329-334.
Henneke, D., Potter, G., & Kreider, J. (1984). Body condition during pregnancy and lactation and reproductive efficiency of mares. Theriogenology, 21(6), 897-909.
Henneke, D., Potter, G., Kreider, J., & Yeates, B. (1983). Relationship between condition score, physical measurements and body fat percentage in mares. Equine Veterinary Journal, 15(4), 371-372.
IER. (2004). Size and scope of the New Zealand racing industry. New Zealand Racing Board, Petone, New Zealand.
IER. (2010a). New Zealand thoroughbred racing fact book 2010. Wellington, New Zealand: New Zealand Thoroughbred Racing.
IER. (2010b). The size and scope of New Zealand racing industry. Report to the New Zealand Racing Board.
Karsch, F. J., Bittman, E. L., Foster, D. L., Goodman, R. L., Legan, S. J., & Robinson, J. E. (2013). Neuroendocrine basis of seasonal reproduction. Recent progress in hormone research, 40, 185-132.
Kimura, J., Hirano, Y., Takemoto, S., Nambo, Y., Ishinazaka, T., Himeno, R., . . . Yokota, H. (2005). Three‐dimensional Reconstruction of the Equine Ovary. Anatomia, histologia, embryologia, 34(1), 48-51.
Kubiak, J., Crawford, B., Squires, E., Wrigley, R., & Ward, G. (1987). The influence of energy intake and percentage of body fat on the reproductive performance of nonpregnant mares. Theriogenology, 28(5), 587-598.
Malinowski, K., Johnson, A., & Scanes, C. (1985). Effects of interrupted photoperiods on the induction of ovulation in anestrous mares. Journal of animal science, 61(4), 951-955.
McCue, P., Logan, N., & Magee, C. (2007). Management of the transition period: physiology and artificial photoperiod. Equine Veterinary Education, 19(3), 146-150.
Morel, M. D., & O’Sullivan, J. (2001). Ovulation rate and distribution in the thoroughbred mare, as determined by ultrasonic scanning: the effect of age. Animal Reproduction Science, 66(1), 59-70.
Morley, S. A., & Murray, J.-A. (2014). Effects of body condition score on the reproductive physiology of the broodmare: a review. Journal of Equine Veterinary Science, 34(7), 842-853.
Nagy, P., Guillaume, D., & Daels, P. (2000). Seasonality in mares. Animal Reproduction Science, 60, 245-262.
NZTB. (2016). About Us. Retrieved from http://www.nzthoroughbred.com/About-us.aspx
Palmer, E., Driancourt, M., & Ortavant, R. (1981). Photoperiodic stimulation of the mare during winter anoestrus. Journal of reproduction and fertility. Supplement, 32, 275-282.
Palmer, E., & Guillaume, D. (1992). Photoperiodism in the equine species—what is a long night? Animal Reproduction Science, 28(1-4), 21-30.
Racing, N. Z. T. (2009). New Zealand thoroughbred racing fact book 2009. Wellington, New Zealand: New Zealand Thoroughbred Racing.
Robinson, J., & Karsch, F. (1987). Photoperiodic history and a changing melatonin pattern can determine the neuroendocrine response of the ewe to daylength. Journal of Reproduction and Fertility, 80(1), 159-165.
Rogers, C., Gee, E., & Bolwell, C. (2014). Reproductive production constraints within the New Zealand racing industry. Journal of Equine Veterinary Science, 34(1), 164.
Rogers, C., Gee, E., & Bolwell, C. (2016). Reproductive production constraints within the New Zealand racing industry.
Rogers, C., Gee, E., & Vermeij, E. (2009). Retrospective examination of the breeding efficiency of the New Zealand Thoroughbred and Standardbred. Paper presented at the Proceedings of the New Zealand Society of Animal Production.
Sharp, D. (1980). Environmental influences on reproduction in horses. The Veterinary clinics of North America: large animal practice.
Tortonese, D. J., Gregory, S. J., Eagle, R. C., Sneddon, C. L., Young, C. L., & Townsend, J. (2002). The equine hypophysis: a gland for all seasons. Reproduction, Fertility and Development, 13(8), 591-597.
Van Niekerk, C., Morgenthal, J., & Gerneke, W. (1975). Relationship between the morphology of and progesterone production by the corpus luteum of the mare. Journal of reproduction and fertility. Supplement(23), 171-175.
Van Niekerk, C., & Van Heerden, J. (1972). Nutrition and ovarian activity of mares early in the breeding season. Journal of the South African Veterinary Medical Association, 43(4), 351-360.
Van Niekerk, F., & Van Niekerk, C. (1997). The effect of dietary protein on reproduction in the mare. III. Ovarian and uterine changes during the anovulatory, transitional and ovulatory periods in the non-pregnant mare. Journal of the South African Veterinary Association, 68(3), 86-92.
Waldron, K., Rogers, C., Gee, E., & Bolwell, C. (2011). Production variables influencing the auction sales price of New Zealand Thoroughbred yearlings. Paper presented at the Proceedings of the New Zealand Society of Animal Production.
Walsh, C., Prendergast, R., Sheridan, J., & Murphy, B. (2013). Blue light from light-emitting diodes directed at a single eye elicits a dose-dependent suppression of melatonin in horses. The Veterinary Journal, 196(2), 231-235.
This literature review has been written by a student and is published as an example. See our guide on How to Write a Dissertation Literature Review for guidance on writing your own.
Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allRelated Content
All TagsContent relating to: "Veterinary"
A veterinarian provides care to animals, including the diagnosis and treatment of any diseases or other conditions that animals may have. A veterinarian practices veterinary medicine, and can also be known as veterinary surgeons or veterinary physicians.
Related Articles
DMCA / Removal Request
If you are the original writer of this dissertation and no longer wish to have your work published on the UKDiss.com website then please:




