Short-acting Antimuscarinic and Short-acting Beta2-agonist Use for Stable Chronic Obstructive Pulmonary Disease
Info: 4489 words (18 pages) Dissertation
Published: 10th Dec 2019
Short-acting Antimuscarinic and Short-acting Beta2-agonist Use for Stable Chronic Obstructive Pulmonary Disease
Table of Contents
Short-acting Beta2-agonist (SABAs)
Short-acting Antimuscarinics (SAMAs)
Purpose Statement
Chronic obstructive pulmonary disease (COPD) is one of the leading causes of death in the United States. Its chronicity and high prevalence result in massive utilization of healthcare resources. There is currently no cure, but proper management can improve symptoms, decrease exacerbations, and prolong survival. This paper will summarize current evidence on the use of short-acting antimuscarinics (SAMA) and short-acting beta-agonists (SABA) for management of occasional dyspnea in individuals with COPD.
Abstract
Objectives
To summarize current evidence on the effectiveness and safety of short-acting antimuscarinics and short-acting beta-agonists for management of occasional dyspnea in patients with chronic obstructive pulmonary disease (COPD).
Evidence Acquisition
Searched PubMed using the terms “pulmonary disease, chronic obstructive”, “short-acting antimuscarinic”, “short-acting anticholinergic” and “short-acting beta agonist.”
Evidence Synthesis
Evidence from current guidelines, systematic reviews, meta-analysis, clinical reviews, and randomized controlled trials were analyzed to compare short-acting antimuscarinics and short-acting beta-agonists in order to determine which of the current recommendations, if either, were superior to improve dyspnea with the best adverse effect profile.
Conclusion
Short-acting antimuscarinics (SAMAs) and short-acting beta agonists (SABAs) showed minimal difference in efficacy in terms of post-bronchodilator FEV1, post-bronchodilator FVC, evening post-bronchodilator PEF, symptoms of breathlessness, and patient preference. While combination therapies showed a small increase in the mean post-bronchodilator peak change from test day baseline, mean test day peak change in FVC, and decreased need for increased or added oral steroids when compared to SABAs, there was no difference between combination therapy and SABAs in terms of quality of life and symptoms of dyspnea.
Introduction
Chronic obstructive pulmonary disease (COPD) is a leading cause of morbidity and mortality worldwide and is expected to continue increasing in prevalence with the rise of exposure to occupational and environmental toxins.1 Three primary symptoms characterize COPD: dyspnea, chronic cough, and increased sputum production. The most common early symptom of the disease is exertional dyspnea.6,7 There is a slow progression of decline in lung function, which is considered the stable phase of the disease. The pathologic changes of COPD are primarily in the airways, but changes in lung parenchyma and pulmonary vasculature may also be seen.6
The decline in lung function is due to an inflammatory process, which results in obstruction and fibrosis of small airways, and eventually the destruction of lung parenchyma. Gas exchange abnormalities then occur, leading to hypoxemia and hypercapnia.1,7
Disease severity is staged by the global initiative for obstructive lung disease (GOLD) using percent predicted postbronchodilator FEV1 into GOLD stages 1-4. The disease also involves periods of acute exacerbation, with episodes of increased dyspnea, cough, and/or sputum production. These exacerbations along with lung function, is how GOLD assesses the disease for targeted treatment.6
Treating COPD is challenge faced across disciplines in medicine. While there is no cure at present for COPD, many drugs can provide symptomatic relief. There are multiple drugs approved for symptomatic treatment, and multiple modes of drug delivery. According to the GOLD guidelines, the first line recommended treatment for COPD patients suffering only from mild dyspnea is a short-acting bronchodilator.1,7 These include short-acting antimuscarinics (SAMAs) and short-acting beta2-agonist (SABAs).
Methods
To compare SABAs to SAMAs, studies that met the following criteria were identified:
1.Compared SABA or SAMA to a placebo or to the other
2. Conducted using adult patients with COPD
3. Did not include the use of any additional pharmacotherapy for COPD treatment, such as long-acting beta2-agonist/anti-muscarinics or inhaled corticosteroids
4.Were published within the last 25 years
5.Were in English
Articles were identified by searching PUBMED using the terms “pulmonary disease, chronic obstructive”, “short-acting beta agonist”, “short-acting antimuscarinic”, and “short-acting anticholinergic.” The search yielded 91 results. 54 articles that were not systematic reviews, meta-analyses, randomized controlled trials, or guidelines were excluded. Of the remaining 37 results, 15 were relevant.

Figure 1 Literature Selection Flowchart
Evidence Synthesis
The GOLD guidelines recommend short-acting bronchodilators for as-needed use in all symptomatic COPD patients with dyspnea.1,2 Regular use of a short-acting beta2-agonist (SABA), e.g. albuterol, levalbuterol, pirbuterol, or a short-acting anti-muscarinic (SAMA), e.g. ipratropium, has been shown to improve forced expiratory volume in 1 second (FEV1) and reduce symptoms.4 When used independently, SABAs and SAMAs are almost equally effective to improve symptoms of dyspnea, improve lung function in terms of FEV1, forced vital capacity (FVC), and peak expiratory flow (PEF), and increase exercise tolerance as measured with six minute walk distance (6MWD) and the shuttle walk test.5 The bronchodilator response to both types of therapy varies from day-to-day, which may have consequences such as unpredicatable symptoms, overuse of the medication, and unpredicatable long term mortality benefits.4,8
Short-acting Beta2-agonist (SABAs)
Short-acting beta2-agonists prevent bronchoconstriction by stimulating beta2-adrenergic receptors to increases cyclic AMP, which relaxes airway smooth muscle.6 Regular SABA use has been shown to be an effective management for stable COPD.5,7 A Cochrane systematic review of 13 randomized controlled trials compared SABA to placebo with primary outcomes spirometric testing, symptom scores, and quality of life found SABAs more effective than placebo in multiple areas. Results of spirometric testing favored SABA over placebo in both post-bronchodilator forced expiratory volume in 1 second (FEV1), with a weighted mean difference (WMD) of 0.140 liters (95% confidence interval (CI) 0.04,0.58), and forced vital capacity (FVC), with a WMD of 0.30 liters (95% CI 0.02,0.58). Evening post-bronchodilator PEF was significantly higher in participants using SABAs, with a WMD of 29.17L/min (95% CI 2.56, 70.94). Symptoms of breathlessness were significantly improved with SABAs, with a standardized mean difference (SMD) of 1.33 (95% CI 1.01, 1.65). Quality of life was measured using the chronic respiratory disease questionnaire (CRQ), with improvements in domains for both dyspnea (p=0.003) and fatigue (p=0.0003). SABAs were also superior to placebo in terms of patient preference, with almost nine time more patients more likely to prefer SABA to placebo (OR 9.04, 95% CI 4.64 to 17.61). Two included studies reported clinically and statistically significant improvements in the 6MW, with increases of 42 meters (p<0.018) and 35 meters (p<0.05). Side effects could not be adequately observed due to insufficient patient numbers and length of observation in the included studies. As this review did not compare SABAs to alternative treatments, the study concluded that while SABAs are an effective treatment for stable COPD, there are multiple effective treatments available and treatment choice can be made based on individual response, patient preference, and economic considerations.5
In another Cochrane systematic review, SABAs and SAMAs were directly compared. The mean post-bronchodilator peak change from test day baseline had a WMD of -0.00 liters (95% CI -0.00, 0.06), showing no significant difference between treatments. Mean test day peak change in FVC also showed no significant differences between treatments (WMD -0.01 liters, 95% CI 0.06, 0.03). Symptoms of breathlessness were not statistically significant between treatments (WMD 0.00, 95% CI -0.09, 0.09). Quality of life was also measured using the CRQ in this systematic review. There was a small, statistically significant difference in treatment groups favoring SAMAs in all domains of the CRQ, including dyspnea (WMD 0.16 units, 95% CI 0.09, 0.23), fatigue (WMD 0.13 units, 95% CI 0.02, 0.23), emotion (WMD 0.17 units, 95% CI 0.05, 0.29), and mastery (WMD 0.18 units, 95% CI 0.06, 0.30). Only two included studies reported data in terms of the 6MW mastery (WMD 62.60 meters, 95% CI -15.65, 140.85). One small, unblinded study found that SAMA use was associated with significant increase in the distance walked, while the other reported no significant difference. This review also compared combination use of ipratropium bromide plus a SABA to a SABA alone, which is discussed later in this paper.4
A prospective three-month cohort in Respiratory Medicine indicated that SABAs are commonly overused by COPD patients. Nearly half of the patients included in the study overused their as-needed SABA at least once during the three-month observation period, with 19% of participants overusing more than half of the days. Overuse, which is defined as greater than 8 actuations of a SABA per day while clinically stable, is associated with a greater degree of dyspnea and increased likelihood of using at-home oxygen when compared with non-overusers.8 Through a mechanism that is unclear, over or repeated use of SABAs can lead to decreased sensitivity to their bronchodilating effect and increased sensitivity to bronchoconstrictive stimuli.8,9
Special care must also be taken when prescribing a SABA to patients with both COPD and comorbidities. Patients with asthma have an increased sensitivity to bronchoconstrictive stimuli with overuse of a SABA. Diabetic patients have an increased risk of ketoacidosis with any beta-agonist use, as beta-agonist stimulation in the liver induces glycogenolysis, which raises blood sugar level.8 Older individuals treated with higher doses of beta2-agonist may experience somatic tremor.2 Patients susceptible to arrhythmias may experience resting sinus tachycardia from stimulation of beta2-adrenergic receptors.7,9 While it was suspected that SABA use may increase risk of myocardial infarction (MI), a 2003 clinical review concluded that there is no increased risk of acute MI associated with SABA use in COPD patients. The adjusted rate ratio for acute MI with a 12 month history of inhaled beta agonist was 1.12 (95% CI 0.95, 1.33) and 1.02 (95% CI 0.52, 2.00) for first time use.10
Short-acting Antimuscarinics (SAMAs)
Short-acting antimuscarinics (SAMAs) reduce bronchoconstriction by blocking acetylcholine’s bronchoconstrictor effects on M3 muscarinic receptors found in airway smooth muscle.2 In the previously discussed Cochrane systematic review comparing SABAs and SAMAs, SAMAs were found to have no statistically significant diference in terms of the mean post-bronchodilator peak change from test day baseline, mean test day peak change in FVC, and symptoms of breathlessness. They were found to improve CRQ scores in all domains when compared to SABAs. SAMAs were also found to be superior regarding increasing or adding systemic steroids. Systemic steroids, which are one of the next treatment steps for COPD according to the GOLD guidelines, were added or increased in fewer participants using ipratropium bromide compared to SABAs (Peto odds ratio (OR) 0.52, 95% CI 0.37, 0.74). The number needed to treat (NNT) for ipratroprium bromide in order to precent one person requiring a course of oral steroids was calculated to be 15 (95% CI 12 to 28).4
In addition to these advantages, ipratropium was also found to have the lowest total treatment costs (Figure 2), encompassing the cost of the drug itself, additional cost of the drug during increased use during exacerbations, and hospitalization costs when compared to albuterol and dual therapy with ipratropium and albuterol.11
Figure 2: Total Treatment costs over the 85-day follow up period for the three treatment groups
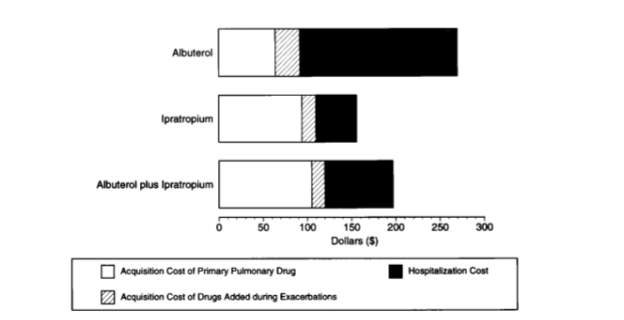
Friedman M, Serby CW, Menjoge SS, Wilson JD, Hilleman DE, Witek TJ, Jr. Pharmacoeconomic evaluation of a combination of ipratropium plus albuterol compared with ipratropium alone and albuterol alone in COPD. Chest. 1999;115(3):635-641.
In a a systematic review and meta-analysis of 17 trials including 14,783 participants, inhaled anticholinergics were compared with the control (control being a placebo, inhaled beta-agonist, or inhaled steroid beta-agonist combination) to determine if there was an increased risk of major cardiovascular events among COPD patients. Anticholinergics were found to be associated with a significantly increased risk of cardiovascular death, MI, or stroke (1.8% vs 1.2% for control; RR, 1.58 [95% CI, 1.21,2.06]; P<0.001). The mechanism in which SAMAs increase these risks is unknown.2 Findings consistent with this were found in a more recent cohort study. It concluded that compared to subjects with no exposure to anticholinergics within the past year, any exposure to anticholinergics within the past 6 months was associated with an increased risk of cardiovascular events (including acute coronary syndrome, heart failure, and cardiac dysarrythmia) (hazard ratio [95% CI] for less than or equal to four and greater than four 30-day equivalents: 1.40 [1.30-1.51] and 1.23 [1.13-1.36], respectively.13
The most commonly observed adverse effect of SAMAs is dryness of mouth.2,11 Like SABAs, there are multiple patient comorbidities which may limit use. In men with benign prostatic hyperplasia, risk of urinary retention increased by 40% with SAMA use.12 Comorbidities such as coronary artery disease or diabetes that increase risk of cardiovascular death must also be considered.
SAMA+SABA Dual Therapy
Another proposed treatment strategy is the use of SAMA and SABA together as a dual therapy. The previously discussed Cochrane systematic review comparing ipratropium bromide plus SABAs versus SABA alone, there was no significant benefit to using dual therapy (Table 1).4
Table 1Outcomes in Cochrane review of ipratropium bromide versus short acting beta2-agonist for stable chronic obstructive pulmonary disease
Abbreviations: FEV1, Forced expiratory volume in 1 second; HRQL, Health related quality of life
In the review, the mean post-bronchodilator peak change from test day baseline had a WMD of 0.07 liters (95% CI 0.05, 0.08), a small increase in the combination group compared to the group that used SABAs. Mean test day peak change in FVC also favored the combination group, with a WMD of 0.12 liters (95% CI 0.08, 0.17). Symptoms of breathlessness were not statistically significant between treatments (WMD 0.04, 95% CI -0.05, 0.13). Quality of life measured using the CRQ showed no statistically significant difference in any domains of the CRQ between combination and SABA, including dyspnea (WMD 0.01 units, 95% CI -0.06, 0.08), fatigue (WMD 0.02 units, 95% CI -0.09, 0.13), emotion (WMD 0.02 units, 95% CI -0.12, 0.16), and mastery (WMD 0.03 units, 95% CI -0.09, 0.14). Fewer subjects using the combination therapy needed increased or added systemic steroids compared to SABAs alone (Peto odds ratio 0.69, 95% CI 0.5, 0.94). The number needed to treat for combination therapy in order to prevent one person requiring a course of oral steroids was calculated to be 20 (95% CI 12 to 108). This study ultimately concluded that the advantage of regular long-term use of either bronchodilator alone or combination SAMA+SABA use is small, if the aim is to improve lung function, symptoms, and exercise tolerance. A treatment strategy proposed in this study suggested SABAs as the initial agent for therapy based solely on cost effectiveness. It also suggested conducting an “n of 1” trial of SABA and SAMA, to determine which gives the best relief of symptoms and continuing with the treatment of their choice, although this strategy does not have formal evaluation.4
In a randomized, double blind controlled trial comparing a SABA and a SAMA delivered together in one device to either drug alone, showed a 20-40% additional increase in bronchodilation in the dual therapy compared to either alone.14 In a randomized crossover study evaluating daily variation in lung function with combined albuterol and ipratropium, a statistically significant reduction in daily variability in FEV1 was found when with dual therapy compared to monotherapy (difference in coefficient of variation=0.007, p=0.019) and no treatment (difference in coefficient of variation=0.022, p=0.001).15This may be due to combining the different mechanisms of action in both drugs. The order in which the drugs were used did not impact outcome.They were equally effective regardless of whether a SAMA or a SABA was used first.14,15 However, patients did not report a significant difference in the severity of their symptoms.13 The drawbacks of dual therapy have not been thoroughly evaluated yet.
Strategies for Practice
A proposed strategy is to allow the patients trials with both a SABA and a SAMA as monotherapies, and let them choose which provides more relief. This strategy has yet to undergo formal evaluation, but provides a more cost effective alternative to dual therapy, encourages active involvement of the patient in their care, and may increase adherence to treatment.4 Ultimately, the choice of inhaler should be directed at achieving goals of treatment (Table 2), as well as be tailored to the individual depending on access, comorbidities, cost, and the patient’s preference.4,5
Conclusions
SAMAs and SABAs have little difference in efficacy in terms of post-bronchodilator FEV1,
post-bronchodilator FVC, evening post-bronchodilator PEF, symptoms of breathlessness, and patient preference.4,5 SAMAs, while favored in terms of patient evaluated quality of life as reported with the CRQ, have a significant correlation with increased risk of cardiovascular events, such as MI and stroke.4,13 Systemic steroids were added or increased in fewer participants using the evaluated SAMA compared to SABAs. SABAs have not been associated with an increased risk of cardiovascular death, however they are associated with an increased risk of respiratory death.8,9 The effectiveness of SABAs can decrease with overuse, a common occurrence in many COPD patients.4
Combination therapy showed a small increase in the mean post-bronchodilator peak change from test day baseline, mean test day peak change in FVC, and less need for increased or added oral steroids when compared to SABAs. There was no difference between combination therapy and SABAs in terms of quality of life and symptoms of dyspnea.4,15 Use of both drugs means patient is at risk for adverse effects of both, as well as a potential increase in cost.
As none of the treatments were significantly superior and all have noteworthy drawbacks, providers must use clinical judgement when deciding which therapy is most appropriate for their patient in order to achieve treatment goals as outlined in the GOLD guidelines (Table 2).1
Table 2 Goals for treatment of stable COPD (GOLD guidelines)
and REDUCE RISK
|
Lee H, Kim J, Tagmazyan K. Treatment of stable chronic obstructive pulmonary disease: the GOLD guidelines. Am Fam Physician. 2013;88(10):655-663, 663b-f.
References
1. Lee H, Kim J, Tagmazyan K. Treatment of stable chronic obstructive pulmonary disease: the GOLD guidelines. Am Fam Physician. 2013;88(10):655-663, 663b-f.
2. Singh S, Loke YK, Furberg CD. Inhaled anticholinergics and risk of major adverse cardiovascular events in patients with chronic obstructive pulmonary disease: a systematic review and meta-analysis. Jama. 2008;300(12):1439-1450.
3. Salpeter SR, Buckley NS, Salpeter EE. Meta-analysis: Anticholinergics, but not β-agonists, Reduce Severe Exacerbations and Respiratory Mortality in COPD. Journal of General Internal Medicine. 2006;21(10):1011-1019.
4. Appleton S, Jones T, Poole P, et al. Ipratropium bromide versus short acting beta-2 agonists for stable chronic obstructive pulmonary disease. Cochrane Database of Systematic Reviews. 2006(2).
5. Ram FSF, Sestini P. Regular inhaled short acting β2-agonists for the management of stable chronic obstructive pulmonary disease: Cochrane systematic review and meta-analysis. Thorax. 2003;58(7):580.
6. Han MK, Dransfield MT, Martinez FJ. Chronic obstructive pulmonary disease: Definition, clinical manifestations, diagnosis, and staging. In: Waltham, MA: UpToDate; 2017.
7. Global Initiative for Chronic Obstructive Lung Disease Pocket Guide to COPD Diagnosis, Management, and Prevention. 2017.
8. Fan VS, Gylys-Colwell I, Locke E, et al. Overuse of short-acting beta-agonist bronchodilators in COPD during periods of clinical stability. Respiratory Medicine. 2016;116:100-106.
9. Cazzola M, Page CP, Rogliani P, Matera MG. β2-Agonist Therapy in Lung Disease. American Journal of Respiratory and Critical Care Medicine. 2013;187(7):690-696.
10. Suissa S, Assimes T, Ernst P. Inhaled short acting beta agonist use in COPD and the risk of acute myocardial infarction. Thorax. 2003;58(1):43-46.
11. Friedman M, Serby CW, Menjoge SS, Wilson JD, Hilleman DE, Witek TJ, Jr. Pharmacoeconomic evaluation of a combination of ipratropium plus albuterol compared with ipratropium alone and albuterol alone in COPD. Chest. 1999;115(3):635-641.
12. Afonso AS, Verhamme KM, Stricker BH, Sturkenboom MC, Brusselle GG. Inhaled anticholinergic drugs and risk of acute urinary retention. BJU Int. 2011;107(8):1265-1272.
13. Ogale SS, Lee TA, Au DH, Boudreau DM, Sullivan SD. Cardiovascular events associated with ipratropium bromide in COPD. Chest. 2010;137(1):13-19.
14. In chronic obstructive pulmonary disease, a combination of ipratropium and albuterol is more effective than either agent alone. An 85-day multicenter trial. COMBIVENT Inhalation Aerosol Study Group. Chest. 1994;105(5):1411-1419.
15. Singh D, Zhu C-Q, Sharma S, Church A, Kalberg CJ. Daily variation in lung function in COPD patients with combined albuterol and ipratropium: Results from a 4-week, randomized, crossover study. Pulmonary Pharmacology & Therapeutics. 2015;31:85-91.
Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allRelated Content
All TagsContent relating to: "Biomedical Science"
Biomedical Science focuses on how cells, organs and systems function in the human body and underpins much of modern medicine. Biomedical Science applies parts of natural and/or formal sciences to help develop advances in healthcare.
Related Articles
DMCA / Removal Request
If you are the original writer of this dissertation and no longer wish to have your work published on the UKDiss.com website then please:

 Relieve symptoms
Relieve symptoms


