Musculoskeletal Loads during Stationary Cycling: Effects of Pedal Modifications for Knee Osteoarthritis
Info: 33997 words (136 pages) Dissertation
Published: 17th Feb 2022
ABSTRACT
Knee OA is a deteriorating joint disease affecting 27 million people in the US and current exercise prescriptions may be improved with new knowledge of their effects on muscle forces and joint contact loads. Cycling rather than other exercise modalities is generally considered an alternative for people with knee OA. If these research objectives were achieved, clinicians would have additional tools related to joint contact loads for treating people with OA with an cycling exercise while controlling progression of OA. The long-term goal of this research is to provide a scientific basis for planning, evaluation and improvement of subject-specific rehabilitation for subjects with knee osteoarthritis (OA).
The principles governing relationships between muscle forces, joint contact loads and movements in people with knee OA, have not been discovered. Determining how to adjust movements to optimize joint contact loads is difficult because experiments do not account for these loads. In combination with experimental approaches, muscle-actuated inverse dynamic simulations provide a scientific framework to estimate important variables and identify cause-and-effect relationships. These activities challenge existing paradigms for exercise prescriptions by including movements specifically designed for decreasing knee joint contact loads.
The research objective is to investigate muscle forces and joint contact loads that are experienced by the knee during cycling. The overall hypothesis is a combination of biomechanical cycling modifications that contribute to altered muscle forces and a reduction in knee joint contact loads in subjects with and without knee OA during cycling; this may be mitigated with a novel pedal design. The overall purpose of this research was to discover relationships between muscle forces, joint contact loads, cycling and OA-friendly cycling modifications for improving exercise prescriptions. The following objectives were addressed: 1) determine the effects of lateral pedal wedges and toe-in on joint biomechanics during cycling and 2) examine the potential of optimization to design subject-specific cycling modifications for decreasing knee joint contact loads.
PREFACE
This dissertation presents three studies conducted using inverse dynamic simulations of muscle forces, joint contact loads and optimization analyses to modify and develop effective cycling prescriptions for knee osteoarthritis. Each chapter is written as a separate technical paper and an overview of the goals and methods employed in each study has been provided. Additionally, each chapter provides an in-depth discussion of the study’s findings as well, as how these findings can be used to answer the scientific questions posed. Chapter 5 provides a summary of the results of the three studies in this dissertation and outlines on how they can be applied to develop better treatment options for individuals at risk for osteoarthritis progression.
TABLE OF CONTENTS
Click to expand Table of Contents
Chapter I Introduction
1.1 Defining Osteoarthritis (OA)
1.1.1 Knee Osteoarthritis
1.1.2 Causes of Osteoarthritis
1.2 Treatment of Osteoarthritis
1.2.1 Modifiable Risk Factors: Weight Management and Physical Activity
1.2.2 Exercise Prescription for OA
1.2.3 Exercise Prescription: Cycling
1.3 Gait Characteristics of Knee Osteoarthritis
1.3.1 Compressive Forces
1.3.2 Knee Adduction Moment
1.4 Statement of the Problem
1.4.1 Muscle Forces
1.4.2 Joint Contact Loads
1.5 Preliminary Work
1.5.1 Selection of human subjects and Data collections
1.6 Computational Modeling: Subject-Specific Musculoskeletal Modeling
1.6.1 OpenSim and Musculoskeletal Modeling Software
1.6.2 Biomechanical Models
1.6.3 Previous Research’s Biomechanical Modeling of the Knee and a Solution
1.6.4 Development and Analysis of Muscle-Actuated Inverse Dynamic Simulations
1.7 Overview and Specific Studies
1.7.1 Study 1: Pedal Modifications Change Muscle Forces Differently in Osteoarthritis (OA) and Healthy Subjects during Stationary Cycling: Implications for Osteoarthritis Exercise Prescription
1.7.2 Study 2: Lower-Limb Joint Contact Loads Adapt Differently in Individuals with and without Knee Osteoarthritis during Cycling with Pedal Modifications
1.7.3 Study 3: Investigate Changes in Knee joint Contact Loads Due to Variations in Muscle Activity during Cycling
Chapter II Pedal Modifications Change Muscle Forces Differently in Individuals with and without Knee Osteoarthritis (OA) during Stationary Cycling: Implications for Osteoarthritis Exercise Rehabilitation
Abstract
2.1 Introduction
2.2 Methods
2.2.1 Subject Demographics
2.2.2 Experimental Motion Capture Data Collection
2.2.3 Development and Analysis of Muscle-Actuated Inverse Dynamic Simulations
2.2.4 Evaluation of Muscle Forces and Test of Hypotheses
2.2.5 Validation of Models and Simulation Results
2.3 Results
2.3.1 Mean and peak Muscle Force Changes Across Conditions
2.3.2 Mean and Peak Muscle Force Differences between Cohorts
2.3.3 Validation of Models and Simulation Results
2.4 Discussion
Chapter III Lower-Limb Joint Contact Loads Adapt Differently in Individuals with and without Knee Osteoarthritis during Cycling with Pedal Modifications
Abstract
3.1 Introduction
3.2 Methods
3.2.1 Subject Demographics
3.2.2 Experimental Motion Capture Data Collection
3.2.3 Development and Analysis of Muscle-Actuated Inverse Dynamic Simulations
3.2.4 Estimation of Joint Contact Loads
3.2.5 Tests of the Hypothesis
3.2.6 Validation of the Model and Simulation Results
3.3 Results
3.3.1 Lateral Wedge Peak Joint Contact Load Differences across Conditions
3.3.2 Toe-in Peak Joint Contact Load Differences across Conditions
3.3.3 Peak Joint Contact Loads Differences between Cohorts
3.4 Discussion
Chapter IV Investigating Changes in tibiofemoral joint contact loads due to variations in muscle activity during stationary cycling
Abstract
4.1 Introduction
4.2 Methods
4.2.1 Subject Demographics
4.2.2 Development and Analysis of Muscle-Actuated Inverse Dynamic Simulations
4.2.3 Static Optimization and Joint Reaction Analysis
4.2.4 Creating the Objective Function
4.2.5 Exploring the Muscle Activation and their Effects on the Joint Contact Loads
4.2.6 A Muscle Coordination Pattern Minimizing Compressive Tibiofemoral Force
4.2.5 Change in Tibiofemoral Forces Due to Maximized Muscle Activations
4.3 Results
4.3.1Minimize Muscle Activations
4.3.2 Minimize Joint Contact Forces
4.3.3 Maximizing the muscle force to minimize the joint loads
4.4 Discussion
Chapter V Conclusion
5.1 Significance of Research
5.2 Research Innovation
5.3 Fundamental Contributions
5.4 Future Work
List of References
Appendix
Vita
LIST OF TABLES
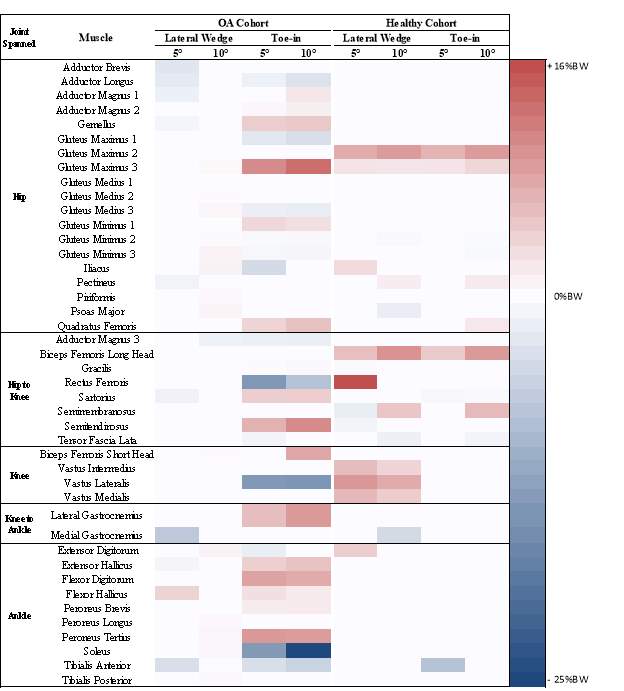
Table 1: A muscle force change identity grid showing the significant differences within each cohort in all lower extremity muscles compared to neutral pedal condition, for the mean muscle forces. Red indicates an increase, while blue indicates a decrease compared to neutral. The darker the color the greater the increase or decrease.
Table 2: A muscle force change identity grid showing the significant differences within each cohort in all muscles compared to neutral, for the peak muscle forces. Red indicates an increase, while blue indicates a decrease compared to neutral; while, the darker the color the greater the increase or decrease.
Table 3: Mean and peak muscle force significant differences between OA (+) and Healthy (-) in all conditions. Red indicates knee OA cohort was greater within that conditions, while blue indicates healthy cohort was higher within that conditions; the darker the color the greater the difference.
Table 4: The joint contact loads change identity grid showing the significant differences within each cohort in all contact loads compared to neutral, for the peak contact loads. Red indicates an increase compared to neutral, while blue indicates a decrease compared to neutral; while, the darker the color the greater the increase or decrease.
Table 5: The peak joint contact loads significant differences between OA (+) and Healthy (-) across all conditions. Red indicates knee OA cohort was greater within the corresponding condition, while blue indicates healthy cohort was higher within that condition. The darker the color the greater the difference.
Table 6: A change identity grid showing the differences in the joint contact loads for the specific muscles that changed from the normal joint reaction analysis. Results are shown for each cohort for step 1’s muscle variation, for the subjects with and without knee OA in all muscles used stands for a muscle weighting on 1, while don’t stands for. Red indicates an increase compared to joint reaction analysis, while blue indicates a decrease compared to joint reaction analysis. The darker the color the greater the increase or decrease.
LIST OF FIGURES
Figure 1: Illustrations showing the direction of the net external knee adduction moment (KAM) represented by the blue curved arrows toward the midline or medial aspect of the body or corresponding knee joint this is a result from a traditional inverse dynamics analysis determining the net forces responsible for the movement. The important fact in the difference between the adduction-abduction contact moments for this research is the net KAM is determined without taking into account for the internal muscle forces.
Figure 2: Showing what the a) lower limb looks like when there is a change in alignments, b) shows a closer look as to what happens at the knee joint when the joint changes from normal alignment to a more varus or adducted alignment.
Figure 3: Many factors contribute to knee joint contact loads associated with OA disease progression and disability during movements. The transformations between experimental EMG patterns and coordinated multi-joint movement (shaded region) are complicated. Furthermore, to make exercise prescription decisions, clinicians must predict joint load changes after equipment adjustments. Exercise prescription alters musculoskeletal geometry and multi-joint dynamics and these changes are not easily measured. The long-term goal of our work is to provide a scientific basis for exercise prescription that reduces harmful knee joint contact loads.
Figure 4: Four-step procedure to generate a subject-specific inverse dynamic simulation that reproduces experimental data.
Figure 5: Example subject during a) data collection and example b) subject-specific musculoskeletal model for muscle force estimation with the added patella.
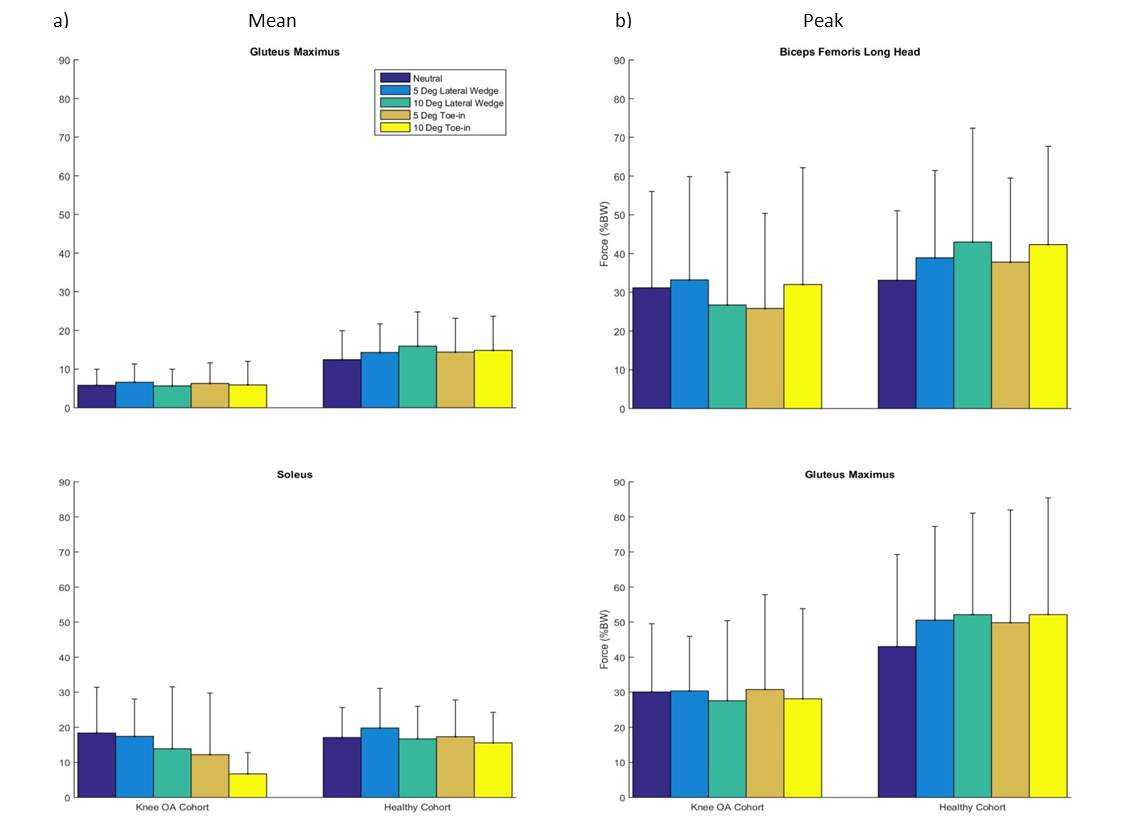
Figure 6: Some of the muscles that showed the most change across the conditions (at least 3 conditions compared to neutral) with a threshold set at greater than 10%BW, observed for a) mean and b) peak muscle forces normalized by %BW.
Figure 7: Comparison of muscle activations during cycling with the neutral pedal condition (at 80W and 60 RPM) and EMG from Neptune, et al., 1997 from elite male cyclists (at 127W and 60 RPM).
Figure 8: An example of a few muscle activations for a) subjects with OA and b) Subjects without OA. This tested the sensitivity of the joint contact loads by altering the muscle activations weightings from normal static optimization (weighting of 1) shown in blue. The red line is a weighting of 0 indicating being able to use the muscle as much as it wants to carry out the cycling movement. The green line is with a weighting of 100 penalizing the muscle with a and not allowing it to be used. Lastly, the black line is for maximizing the muscle activations.
Figure 9: Subjects with (left) and without (right) knee OA’s optimized adduction-abduction moment (top) and the compressive load(bottom) results shown, from the minimized knee joint contact loads and the maximized muscle forces in order to minimize joint contact loads. Minimum knee contact loads shown in red and maximized muscle forces shown in green were plotted against previously calculated joint contact loads from joint reaction analysis shown in blue.
Definition of Terms
Chapter I Introduction
1.1 Defining Osteoarthritis (OA)
Over 47.5 million adults in the United States have a physical disability, with roughly 8.6 million cases of arthritis being the leading primary cause of physical disability [1, 2]. These patients are commonly left with disabling pain leading to a loss of mobility and an overall reduced quality of life [3, 4]. Osteoarthritis (OA) is the most common form of arthritis; it affects millions of people worldwide. OA is a common joint disease that most often affects the aging population. Although it is more common in older adults and is often viewed as a wear-and-tear disease, though it is not accurate to state that the joints are merely eroding. OA is a disease of the entire joint, characterized by the breakdown of the cartilage, bony deviations in the effected joint, weakening of tendons, ligaments, and inflammation of the joint cavity.
Although OA can affect any joint in the body, the joints most commonly affected are the weight-bearing joints of the lower extremities, mainly the knees and hips [4]. With OA, the cartilage and bones within a joint begin to break down. These changes usually develop slowly and gradually worsen. OA can cause severe joint pain, swelling and stiffness. In some cases, it also causes reduced function and disability; some people are no longer able to carry out activities of daily living and, in some cases, are not able to work. Severe cases may require joint replacement surgery, particularly for the knee or hip joint.
1.1.1 Knee Osteoarthritis
Knee osteoarthritis, is a harmful, deteriorating joint disease that significantly alters the quality of life [5] for an estimated 27 million people in the United States [6-8]. Knee OA mostly affects the aging population. Of those affected, 37.4% are 60 years of age or older [6, 9]. Of those with doctor-diagnosed OA, roughly one quarter report problems with carrying out tasks of everyday living [8, 10]. OA is characterized by decreased neuromuscular control, weakened lower extremities and knee joint instability, with symptoms developing slowly over time [11]. To manage OA, industry costs exceeds $128 billion [10] creating an alarming clinical burden of 36 million ambulatory care visits and 750,000 hospitalizations per year [7] resulting in high socioeconomic costs [3]. OA is a clinical problem with very few treatment options beyond pain management until the disease progresses enough to require invasive and expensive surgery [12].
1.1.2 Causes of Osteoarthritis
Though the causes of OA are not completely understood, there are some commonly known risk factors including age [13-15], female gender [16-18], muscle weakness [19-21], genetics [14], injury [22], overuse [23, 24] and obesity [13]. OA is the leading cause of disability in the aging population. The goal of osteoarthritis treatment is to reduce pain and improve joint mobility. Currently there is no absolute cure for OA, though there are a few treatments that attempt to slow the progression of OA before it reaches a more advanced state. Some risk factors are modifiable and can help in alleviating OA joint pain and improving joint function (mobility).
1.2 Treatment of Osteoarthritis
Doctors usually treat OA with a combination of therapies that may include, but are not limited to, the following: physical activity, medications, physical therapy (for example muscle strengthening), weight loss, supportive devices and when pain management is no longer an option, surgery. In addition to medical treatment, people with OA can gain confidence in managing their condition with strategies that over the last few decades have proven to reduce pain and disability, so they can pursue the activities important to them. Patients with OA can alleviate their pain and improve joint function by learning and adapting to simple and effective arthritis management strategies aimed towards weight management and increasing physical activity.
1.2.1 Modifiable Risk Factors: Weight Management and Physical Activity
Based on existing evidence, it is crucial for people that are overweight to try to lose weight. Excess weight is detrimental to personal health and speeds progression of OA. Obesity is one of the single most modifiable risk factors in the development and progression of OA. Weight loss has been proven to reduce the debilitating symptoms that OA patients commonly encounter [25-27]. A study that was conducted by Messier et al. [26] demonstrated that for every pound of body mass a person loses, the compressive load across the knee joints is reduced by four pounds. Other researchers have noted that weight loss by means of diet and exercise resulted in improvements of joint function, mobility and reduced (or decreasing) pain [25, 27].
In recent years, the Osteoarthritis Research Society International (OARSI) has created a list of 25 recommendations for the treatment of patients with OA of the hip or knee [28, 29]. These recommendations come from evidence-based research and have been proven to alleviate some of the symptoms of OA. Two of the highly recommended non-pharmacological treatments are regular aerobic muscle-strengthening exercises and weight loss for overweight individuals [28, 29].
1.2.2 Exercise Prescription for OA
Different forms of exercise such as walking and cycling are commonly prescribed by healthcare professionals to reduce potentially harmful knee joint contact loads and are effective exercise prescriptions for treatment in populations with knee OA [28, 29]. Walking modifications have proven to be successful in diminishing knee pain by the use of a toe-in gait or lateral shoe wedges [30-32].
1.2.3 Exercise Prescription: Cycling
Bicycling allows people to increase their aerobic capacity during exercise without harmful joint loads in the lower extremities, as most of the body weight is supported by the seat of the bicycle, relieving the load-bearing joints. One study found that in subjects with knee OA, low-intensity cycling and high intensity cycling are essentially equivalent in increasing aerobic capacity, improving joint function and decreasing pain [33]. For subjects with OA, cycling’s health benefits include potentially reduced OA-related loads on the diseased joint tissues. However, there is little scientific evidence of the magnitude and location of these reduced joint contact loads on the knee. Despite the fact that cycling remains an integral component of exercise for subjects with OA, the potentially beneficial changes in muscle forces and associated joint contact loads is not well understood. Therefore, current exercise prescriptions may be improved with new knowledge of muscle forces and their effects on joint contact loads. This loading plays a crucial role in the progression of joint degeneration and severity of pain during exercise and activities of daily living [5, 34]. Thus, providing a better understanding of how different cycling movements affect the muscle forces and joint contact loads may be necessary for improving rehabilitation strategies.
Many authors have reported on lower-extremity joint kinematics during cycling [35-40]. While cycling motions are relatively similar due to the cyclical nature of cycling, there are differences that exist depending on the pedal cadence, workload and pedal width, seating arrangement and cycling modifications. These modifications alter the body’s orientation and joint positions, which in turn affect the entire body [41] and change the kinematic and kinetics configuration such as the joint angles and joint moment arms by making small adjustments to the cycling orientation. All of these modifications affect human biomechanics, changing the muscle lengths and moment arms and affecting the kinematics (and kinetics) in the muscles, which in turn affect the joint contact loads.
1.3 Gait Characteristics of Knee Osteoarthritis
It is easy to understand knee OA gait characteristics because the changes, which the OA patient experiences with an adapted gait have been, studied more than cycling. It is important to start studying the effects of cycling modification to understand what is happening at the knee joint for subjects with and without knee OA. Any alteration from the normal body orientations affects the biomechanics and orientation of the joint angles and moment arms that in turn affect the muscle length and joint contact loads. At the time of this writing, few or no studies have reported biomechanical variables of subjects with knee OA during cycling. There are also no studies that have tested both subjects (non-elite athletes) with and without knee OA in the same study. Subjects with knee OA have adapted different knee kinematics when compared to subjects without knee OA, having decreased or slower walking speeds, less total range-of-motion (ROM) in the affected limb and an increased peak net external knee adduction moment (KAM) compared to healthy subjects [5, 31, 32].
1.3.1 Compressive Forces
Peak compressive force acting on the knee joint during walking may reach up to approximately four times the body weight [26]. This shows that the mechanical loading has the potential to be adversely affect the joint by causing greater knee joint forces during walking. In a population with a more varus alignment at the knee, the joint contact loads during walking were shown to be about 3 times body weight [42]. In the OA population, the knee joint contact loads during walking were shown to be about 3.7 times body weight [26]. Messier et al. [26], studied the knee joint loading of older adults with knee OA. The results of the study showed that in comparison to the matched controls, the knee OA group experienced about a 25% reduction in compressive force across the knee; however, those patients had a decreased walking speed compared to their healthy cohort. Another study reported that subjects with medial compartment knee OA encountered 4% higher knee joint reaction forces during walking compared to their matched healthy counterparts [5]. While the differences reported appear to be small, the results suggest a relationship may exist between medial compartment OA and compressive knee joint contact loads. However, there is no pivotal evidence that has yet appeared in the literature about the relationship between knee OA, muscle forces and joint contact loads during cycling.
It is easy to assume that an increase in compressive joint loads has a harmful effect on the knee joint. Additionally, others studies have found that people with knee OA have been successful at lowering the loads on the affected limb in an attempt to reduce the pain during gait [5, 43]. Finally, a study by Chakravarty et al. [44] showed that middle and older aged long-distance runners did not have any change or alteration in OA progression compared to healthy non-runners studied over an 18 year period. These findings suggest that the response to joint loading may depend on the health of the knee joint cartilage. It can be argued that chronic compressive loads on the knee joint are not necessarily responsible for the onset of knee OA, but rather they have a large influence on the progression of OA once the disease has been acquired.
1.3.2 Knee Adduction Moment
Most patients with OA suffer from the most common form, which is medial compartment degeneration and pain, which occurs about 10 times more often than lateral compartment disease [45]. This is likely due to greater medial loading during activities of daily living [31]. Due to this fact, changes in knee kinematics and kinetics associated with knee OA have led to gait adaptations in order to maintain stability, causing a significant increase in the net external knee adduction moment (KAM) [11]. Presence, severity and progression of medial knee OA has been correlated with the first peak of the net external KAM, which is commonly used as a measure of medial knee loading [3, 42]. The weight bearing or compressive load that is centered on the medial compartment of the knee is produced by the KAM, which is opposed by an internal KAM. This net external KAM (Figure 1), acts to adduct the knee during stance into a deformity of the knee which alters the joint from normal (neutral) to a more inward angle (varus or adduction; Figure 2) position [46]. A condition that opens the joint space on the lateral aspect of the knee while compressing the medial joint space of the knee. During gait, this mechanical abnormality shifts the mechanical loading from the previously even load on the knee to a medial compartment load, which will in turn cause greater joint space narrowing. In gait (walking), there are typically two peaks present in the KAM. The first peak, which is associated with weight acceptance during the stance phase of walking, appears to have the largest influence on the knee OA population [43].
Other studies have found a relationship between the magnitude of KAM and the progression of knee OA [5, 44]. For example, Mundermann et al. [5], performed a gait analysis on 42 patients with bilateral medial compartment knee OA and 42 matched controls. The participants walked in their own shoes at a self-selected pace. The results of the study showed that the patients with more severe knee OA demonstrated about 11% larger first peak KAM compared to their matched counterparts and about 28% greater subjects with less severe OA. The authors also reported that even though the walking speeds were self-selected, the speeds were not different between groups, meaning that the differences seen in the KAM cannot be attributed to different walking speeds. Shull et al. [31, 32], conducted a study that attempted to reduce the KAM by having knee OA subjects walk with a toe-in foot progression angle. The authors found that this method of walking reduced the first peak KAM by about 13%, but the second peak KAM and the knee flexion moment remained unaffected. This study provided promising results for a simple technique to reduce the KAM during walking and it may be a potential solution for OA subjects and gait modifications for improvement of the lower limbs. Literature suggests that a relationship of the net external KAM factors has also been shown to be related to the severity and progression of knee OA [5, 46].
1.4 Statement of the Problem
How applying modifications to repetitive motions such as cycling exercise to achieve beneficial changes in muscle forces and joint contact loads connection to OA severity and progression is an open question. Exercise prescription based on joint contact loads is problematic because there is currently no scientific basis for determining how individuals’ exercise prescriptions contribute to joint contact loads leading to knee OA progression. The muscle tests performed during a physical exam, kinematic, kinetic and electromyography (EMG) data obtained from clinical motion analysis are not sufficient to identify the biomechanical source of an individual’s knee OA progression risk or to predict the consequences of exercise prescription. This limitation exists because transformations from EMG patterns to motion are extremely complex (Figure 3) and effects of exercise prescription on musculoskeletal geometry and multi-joint dynamics are not easily measured in an experiment.
Determining how to adjust exercise movements to optimize joint contact loads is difficult because experiments do not account for the aforementioned loads. For decades, experimental approaches have advanced the understanding of neuromuscular control, muscle strength, human motion within joints and the human bodies’ functional capacity. Progress is essentially limited and affected by two factors: 1) important variables (e.g., muscle forces and joint contact loads) are not generally measureable in experiments and 2) cause-and-effect relationships (e.g., motion contributions to joint contact loads) are difficult to establish from experiments. A scientific framework is needed, in combination with experiments, to uncover relationships between muscle forces, joint contact loads and purposeful movements in subjects with OA during rehabilitation. Muscle-actuated inverse dynamic simulations provide scientific framework that complements experimental approaches by estimating important variables and identifying cause-and-effect relationships. This research challenges existing paradigms for exercise prescriptions by including movements that are specifically designed for decreased knee joint contact loads. In turn, these activities help bridge the gap between the experimental approaches that are often used by physicians, physical therapists and scientists focused on improving rehabilitation. In addition to the engineers, mathematicians and computational scientists that utilize computer simulations to improve research studies of movement modification and lead to new rehabilitation solutions.
This dissertation is transformative because (i) these models and simulations are subject-specific rather than one-size-fits-all and (ii) they combine clinical and engineering approaches with rehabilitation exercise in a comprehensive way that has yet to be done. Efforts to incorporate results of dynamics-based analyses of human movement into clinical practice have important limitations. Most simulations have trusted upon basic or more general models, based on experimental data collection from an inadequate number of adult-sized cadavers. No study before now has used a subject-specific musculoskeletal model, in combination with an inverse dynamic simulation, to assess patients with medial compartment knee OA. While investigating and mitigating the magnitude of the muscle forces and joint contact loads experienced in the lower extremities during stationary cycling. This application of subject-specific models and computer simulations can be used to custom-tailor cycling rehabilitation programs.
1.4.1 Muscle Forces
Although cycling remains an essential rehabilitation component with exercise therapy for subjects with knee OA, the associated changes in muscle forces are not well understood. Cycling modification exercises might possibly improve and strengthen associated muscle forces to maintain or improve mobility while improving joint biomechanics of the unhealthy knee joint [42]. Nevertheless, there is little to no scientific evidence that portrays the muscle forces in subjects with knee OA during exercise [47, 48]. Thus, existing exercise treatments may be considerably improved and further developed with new knowledge of how modified cycling affects the associated muscle forces.
1.4.2 Joint Contact Loads
To better treat and understand patients with knee OA in the future, it is necessary to quantify the joint contact loads and how these loads change with cycling modifications. Studying knee loading through computational models is often used with gait analysis to determine musculoskeletal loading in vivo because there is no way to obtain a non-invasive measurement [49]. Knee loading has been previously thought to be a major contributor in the degeneration of articular cartilage associated with the progression of OA and it has been found that patients exhibit increased knee joint contact loads during gait [50].
There are several studies investigating various gait modifications, but few focus on cycling modifications and far fewer are able to effectively estimate the muscle forces and joint contact loads. Internally rotating and laterally abducting the ankle (altering the foot kinematics) using a cycling pedal modification can potentially reduce the net external KAM in subjects with and without knee OA, though the effect on muscle force and joint contact loads are unknown [31, 32]. Many others have studied the effects of different cycling modifications to reduce the KAM, including lateral pedal wedges [51], toe-in cycling [51, 52], pedal cadence [35, 36, 38, 53, 54], work rate [53, 54] and different cycling positioning [41]. However, at the time of this writing there are no studies on the effects of cycling modification and the potential for reduced joint contact loads for subjects with and without knee OA.
Inverse dynamics has traditionally been used to estimate the net joint loads such as the KAM during motion, but this approach is limited because it does not account for muscle forces, which may differ even more following cycling modifications. Determining the joint contact loads that account for these muscle forces provides for a better understanding of the joint mechanics and overall musculoskeletal function [49]. By investigating joint contact loads under cycling conditions, these modifications may be better understood for use in early intervention treatment strategies for patients with knee OA.
1.5 Preliminary Work
Experimental studies have been previously performed [51, 52] with this data, where surrogate measures of OA were examined. Specifically, the peak knee adduction angle and net internal peak knee abduction moment were calculated in Visual 3D. The internal knee abduction moment, is used as a measure for the medial compartment loading of the knee. This study found promising results in which a decrease in the knee adduction moment occurred using the lateral wedge. Another study [52], examined the effects of toe-in pedal modifications and found that the peak knee adduction angles decreased, but the internal knee abduction moment did not decrease, for subjects with and without knee OA. This preliminary work indicates a decrease in the internal knee abduction moment for lateral pedal wedges rather than toe-in. The muscles forces contributing to knee OA and joint contact loads are unknown.
This previous study led to the working hypothesis that lateral pedal wedges and toe-in will reduce knee joint contact loads by changing the distribution of muscle forces in all subjects during cycling. The purpose of this research is to determine the changes in magnitude of the muscle forces and joint contact loads experienced at the knee for subjects with medial compartment knee OA. Additionally, it will allow us to discover relationships between muscle forces, joint contact loads and cycling to develop OA-friendly cycling modifications and subject-specific exercise prescriptions.
1.5.1 Selection of human subjects and Data collections
Previous experimental data has been collected [51, 52] at the Biomechanics and Sport Medicine Laboratory on University of Tennessee-Knoxville’s campus from patients (6 male and 7 female) with medial compartment knee OA as well as healthy control subjects (6 male and 5 female). Subjects participated in five different cycling conditions on a stationary bicycle. The conditions varied by adjustments made to a customized instrumented foot pedal that recorded the pedal reaction forces. The five conditions with pedal adjustments included: 1) neutral position, 2) 5° toe-in pedal, 3) 10° toe-in pedal, 4) 5° pedal wedges and 5) 10° pedal wedges. See Chapters 2, 3 and Gardner et al., [51, 52] for additional details on the selection of human subjects and the data collection process.
1.6 Computational Modeling: Subject-Specific Musculoskeletal Modeling
Computational modeling of human movement is used to relate various aspects of the human anatomy and physiology to movement. In the last few years and decades, biomechanical models have led to tremendous advances in computer technology that have subsequently prompted greater development of more complex biomechanical models with greater accuracy and computationally more efficient analyses [55, 56]. Through computational modeling, researchers are able to develop subject-specific simulations that relate joint kinematics and kinetics to muscle force production and function. Unlike EMG analysis where muscle activation is linearly related to muscle force, simulations are able to account for the musculotendon properties such as muscle activation and contraction dynamics, force-length and force-velocity relationships and moment arms analysis to appropriately model non-linear relationships between muscle activation and force production. Such simulations are utilized in investigating the cause-and-effect relationship between joint motion and muscle function [57-61].
1.6.1 OpenSim and Musculoskeletal Modeling Software
Musculoskeletal modeling software programs allow users to select from a bank of models and create subject-specific simulations to explore a variety of research questions. OpenSim is a software program that provides users with a mathematical and computational modeling framework to analyze everything from designing prosthetic devices and studying how they will function in the body, to assessing the outcomes of surgical procedures like tendon lengthening in cerebral palsy patients. It is unique in that it is user friendly but also allows the user to increase model complexity to answer difficult problems related to human movement.
1.6.2 Biomechanical Models
The biomechanical models and computational tools developed yield broad applications. Numerous studies have been performed to record neuromuscular excitation patterns, understand muscles contraction dynamics, characterize musculoskeletal geometry and quantify multi-joint movements and kinematics. However, linking the detailed knowledge of these elements of the neuro-musculoskeletal system to create an integrated understanding of normal and pathological movement remains a major challenge in the application of biomechanics to a wide range of clinical problems and basic science research.
This study used dynamic modeling and simulation to identify the cycling pedal modification parameters that contribute to knee (and other) muscle forces and joint contact loads related to the pain and disability of individuals with knee OA and to explain the functional consequences of pedal modifications in decreasing these forces. Subject-specific, muscle-actuated, inverse dynamic simulations were created that reproduced experimentally measured and collected data from both knee OA and healthy subjects while cycling. The simulations were analyzed to determine how pedal modifications, lateral pedal wedges and toe-in angles alter foot position and influence knee joint contact loads for subjects with and without knee OA. This analysis will clarify the decrease (or increase) in muscle forces, joint contact loads during modified cycling, and they will enable us to answer the clinically important questions posed in studies 1-3 (Section 1.7). Ultimately, this work will test the utility of simulation-based medicine to identify optimal cycling modifications and facilitate the design of safe, subject-specific exercise prescriptions aiming to increase mobility while decreasing injury and the progression of knee OA.
1.6.3 Previous Research’s Biomechanical Modeling of the Knee and a Solution
The knee joint in the human body is made up of many components including ligaments, articular cartilage, menisci and muscles, which are capable of bearing and transferring the weight bearing load during activities of everyday life. Unfortunately, the knee joint is vulnerable to disease and injury, because of the large mechanical loading (weight bearing) that it is subjected to. With this fact in mind, it becomes crucial for identifying and quantifying the joint contact loads placed on the anatomical tissues that surrounds the human knee, which is critical for understanding and studying joint diseases like OA. Studies are inherently limited by how difficult it is to measure clinically important quantities such as muscle forces and joint contact loads. Therefore, muscle-actuated inverse dynamic musculoskeletal models are becoming a more practical approach for determining how musculoskeletal features interact with each other in order to generate movement. Overall, these simulations combine a subject-specific inverse dynamic skeletal model with muscles, but they rarely contain articular contact models due to their increased complexity and high computational expense. OpenSim is readily available open source software, developed inside a multibody dynamics framework; this enables the user to construct and simulate a musculoskeletal model, with the visualization of experimentally measured and simulated motion and allows for the extraction of useful information (i.e. muscle forces and joint contact loads) from the simulations [57]. Musculoskeletal models are used to examine the joint kinematics in order to predict muscle forces and joint contact loads during various movements and modifications.
However, one potentially problematic area is that the knee is usually simplified as a one degree of freedom joint and neglecting the ligaments that provide a constraint to the frontal plane of the knee. Previous musculoskeletal models allow researchers to investigate medial-lateral knee joint contact force during activities like walking [62, 63]. Some musculoskeletal modeling procedures require intricate, multi-step analyses, or the use of both full-body simulations and finite element models [64-68]. Finite element models rely on a proper representation of the joint surfaces and require expensive and highly invasive imaging techniques that may be unavailable.
Predictions of medial-lateral knee joint contact loads in subjects while using musculoskeletal modeling with generic geometry it is potentially inaccurate when the model does not accurately represent the specific subject. Certain specifications and adjustments made to subject-specific modeling parameters may improve accuracy and results [69]. Two such parameters are the frontal-plane knee alignment and medial-lateral contact these two locations are most likely influenced by the model, which is predicting the medial-lateral contact forces. By altering the line of action in which the muscle forces are articulating relative to each joint compartment that they are acting on. The frontal-plane knee alignment has the potential to effect the knee joint loading [68, 70-72] and can vary up to 3.75° in individuals without obvious varus-valgus [73]. Current modeling techniques have limitations that prevent the realistic representation of the frontal-plane alignment in subjects. For example, the generic models usually restrain the motion of the knee in the frontal plane [66, 69, 74, 75] and on the other hand, the models that are based on geometry obtained from diagnostic imaging are of non-weight bearing subjects [64, 69]. In addition, when medial-lateral contact is estimated through single points, the mechanical loading of the knee is directly influences by the location of the points. A common assumption is usually made that the medial-lateral contact locations are centered at the midline of the affected knee sitting between the femoral condyles in normal or natural knees [74], though inconsistency in the alignment and joint deterioration may change these locations and ultimately affect the magnitude experienced in the knee. To address an essential need for a better and improved knee joint contact loads that provides an accurate representation of the human body and the anatomy of the knee. The model could be altered by adding an additional constraint in order to have it resemble a realistic knee joint and will be constrained like normal human knee joint motion [76].
1.6.4 Development and Analysis of Muscle-Actuated Inverse Dynamic Simulations
The development of muscle-actuated, inverse dynamic simulations that accurately characterize the movement patterns of individuals with medial compartment knee OA offers tremendous potential to advance the prescription of cycling for these individuals. This study is designed so that many of the scientific objectives can be accomplished by making incremental modifications to the existing dynamic models. Over the past several years, engineers have helped to develop core methodologies and a powerful software framework to create subject-specific inverse dynamic simulations that reproduce experimental data.
OpenSim is used [57] to create, alter and evaluate models of many different musculoskeletal structures. The technique for creating and investigating an inverse dynamic simulation of subject-specific movements consists of several steps (Figure 4). Step 1 modifies an existing, generic model of the musculoskeletal system as needed to account for differences in the subject’s size and muscle moment-generating capacity. The musculoskeletal geometry of the generic model is scaled based on subject’s data collected from experimental motion capture.
Step two, processes the subject’s measured kinematics and pedal reaction forces data for “inverse dynamic tracking.” An optimal inverse kinematics problem is solved to minimize the distance between markers on the subject and markers on the model.
Step three, uses inverse dynamics and step two’s calculated kinematics as well as the experimental collected pedal reaction forces to determine the net joint moments.
Step four, the static optimization tool steps through each time step of a generalized motion and calculates the muscle activations that is generated from the experimental kinematics. The equations of motion relate the simulations accelerations to the muscle moments of the model. The musculoskeletal geometry relates the muscle moments to the muscles force generating capacity of the moments. The muscles contraction dynamics relate the muscle forces to the muscle activations. For more information on this step, please refer to section 4.2.3.
Step 5 compares the muscle excitation patterns, joint moments and pedal reaction forces determined from the simulation to the experimental data to verify that the solution obtained from the tracking algorithm provides a reasonable representation of the subject’s movement. A simulation is suitable for analysis if the joint angles and other variables that are not explicitly tracked, such as the joint moments, pedal reaction forces and muscle excitations, are within ±2 standard deviations of the subject’s measured between-trial variability. If the simulation does not meet these criteria, appropriate adjustments to the tracking parameters are made and step 4 is repeated.
The simulation is then ready to be used for knee OA rehabilitation purposes during a final step by performing an analysis of the particular patient’s joint contact loads during cycling. A calculation is made of the resulting forces and moments transferred between consecutive bodies because of all loads (including muscle forces) acting at joints of interest (e.g., knee). These resulting forces and moments, often referred to as joint reactions or joint contact loads, are necessary to quantify bone-on-bone forces at the joints. These joint reactions are a first order approximation of the joint loading conditions and are used to assess the subject’s joint contact loads as others have done for joint implant loads [77] with differing OA severities [78].
This dissertation utilizes high quality experimental motion capture data of individuals (OA and healthy subjects) performing with five different pedal modifications to conduct simulation-based research on muscle forces and joint contact loads while cycling. This research is divided into three different studies introduced in the following section.
1.7 Overview and Specific Studies
Arthritis is the leading cause of physical disability in the U.S. and OA is the most prevalent form of arthritis causing disabling pain and loss of mobility for almost 27 million adults (14% of all aged 25 and older) worldwide. OA costs in the US, Canada, UK, France and Australia account for 1–2.5% of each country’s gross domestic product. Disability and healthcare costs of OA can be drastically reduced by novel subject-specific intervention programs (i.e. exercise) that benefit subjects with OA by slowing disease progression and delaying disability. However, there is a significant need for evidence-based prescription targets that directly address joint contact loads during exercise. Cycling rather than other exercise modalities is generally considered an alternative for subjects with knee OA, but there is a lack of scientific knowledge characterizing and maximizing the rehabilitation benefits (i.e. altered muscle forces and reduced joint contact loads) of cycling for subjects with knee OA.
The long-term goal of this research is to provide a scientific framework for planning, evaluation and improvement of subject-specific rehabilitation for individuals with knee OA. Previous research has shown significant relationships between gait retraining modifications and lower-extremity joint contact loads. Research has utilized subject-specific modeling and simulation along with predictive tools using feedback control to design new movements that aim to minimize joint contact loads for walking and multi-directional sporting movements. Ultimately, no studies have combined cycling biomechanics with computer simulation to investigate optimal exercise prescriptions for individuals with knee OA.
The research objective is to investigate how cycling exercise modifications lead to different joint contact loads in subjects with knee OA. This study proposes to use a combination of clinical motion analysis, subject-specific musculoskeletal models, muscle-actuated inverse dynamic simulations and optimization to determine what features and their variations lead to improved joint contact loads for better exercise prescriptions.
The overall hypothesis is that a combination of biomechanical movement features, achieved in part by a novel pedal design, contributes to reduced joint contact loads in individuals with and without knee OA during cycling. This dissertation will address the following three studies:
1.7.1 Study 1: Pedal Modifications Change Muscle Forces Differently in Osteoarthritis (OA) and Healthy Subjects during Stationary Cycling: Implications for Osteoarthritis Exercise Prescription
Goal: Answer the question of how muscle forces change for subjects with and without OA using novel pedal designs to modify foot position during cycling.
- Pedal modifications will cause a change (increase or decrease) in the mean and peak muscle forces compared to the neutral pedal condition for each cohort.
- Mean and peak muscle forces for subjects with knee OA will be different (higher or lower) compared to healthy subjects without knee OA during cycling in each pedal condition.
Methods: Create subject-specific, muscle-actuated, inverse dynamic simulations that reproduce experimentally measured data from both populations during cycling with different pedal modification conditions and quantify the muscle force estimates.
Significance: This work will demonstrate how pedal modifications change subjects with and without knee OA muscle forces adversely
1.7.2 Study 2: Lower-Limb Joint Contact Loads Adapt Differently in Individuals with and without Knee Osteoarthritis during Cycling with Pedal Modifications
Goal: Answer the question of how muscle forces found in Study 1 will affect the joint contact loads while cycling in different pedal modifications.
- Pedal modifications will cause a change (increase or decrease) in the peak joint contact load compared to the neutral pedal condition for each cohort.
- Peak joint contact load for subjects with knee OA will be higher compared to healthy subjects without knee OA during cycling in each pedal condition.
Methods: Examine the previously created subject-specific, muscle-actuated, inverse dynamic simulations that reproduce experimentally measured data from both populations during cycling with different pedal modification conditions and compare the joint contact loads.
Significance: This work will discover how pedal modifications alter the subjects with and without knee OA joint contact loads adversely.
1.7.3 Study 3: Investigate Changes in Knee joint Contact Loads Due to Variations in Muscle Activity during Cycling
Goal: Determine the effect of varied muscle coordination on knee joint contact loads and the potential for a subject to decrease tibiofemoral forces while cycling by adopting a muscle coordination strategy that minimizes the important tibiofemoral contact loads.
Methods: A series of tasks to create a modified static optimizations framework that will be formulated to reduce knee joint contact loads by altering weights and muscle activations. Significance: This work will define what muscle activations that would achieve optimal (i.e., minimized) knee joint contact loads.
Together these studies will test the utility of simulation-based medicine to identify optimal pedal condition modifications and facilitate the design of safe, subject-specific exercise prescriptions aiming to increase mobility while decreasing harmful knee joint contact loads causing pain and disability for subjects with knee OA. They will also aid in determining the roles that muscle forces play in cycling kinematics in the healthy versus the OA cohorts in order to better develop future research for designing more effective subject-specific rehabilitation prescription.
Chapter II
Pedal Modifications Change Muscle Forces Differently in Individuals with and without Knee Osteoarthritis (OA) during Stationary Cycling: Implications for Osteoarthritis Exercise Rehabilitation
Abstract
Osteoarthritis (OA) is a severe clinical problem affecting an estimated 27 million adults in the United States with the only clear treatment options being pain management. Cycling is an integral component of exercise for individuals with knee OA, while the muscle forces during cycling remain unknown. Thirteen subjects with knee OA and eleven healthy subjects performed a cycling protocol five pedal conditions (neutral, 5° lateral wedge, 10° lateral wedge, 5° toe-in and 10° toe-in). Six hundred muscle-actuated inverse dynamic simulations (2 cohorts, 5 conditions, 5 trials in each condition) were performed to estimate muscle force differences between conditions. There were many changes in the mean and peak muscle forces for the subjects with and without knee OA across pedal modifications compared to neutral pedal condition. For the mean muscle force, subjects with knee OA had 44 (lateral wedge) and 56 (toe-in) significant changes. Healthy subjects had 40 (lateral wedge) and 35 (toe-in) significant changes. For the peak mean force, changes between subjects for the neutral condition there was 26 significant differences while the lateral wedge had 58 and toe-in condition had 68 significant differences between cohorts. In conclusion, this study showed while cycling in different pedal modifications, the subjects with knee OA experienced a greater effect of mean and peak muscle forces compared to subjects without knee OA. Toe-in pedal modifications showed to have had a greater impact on the knee OA cohort. Muscles either increased or decreased forces depending on the joints they spanned.
2.1 Introduction
Osteoarthritis (OA) is a degenerative joint disease significantly affecting the quality of life [5] for an estimated 27 million adults in the United States [6]. OA involves the breakdown and wear on the joint cartilage causing pain and disability [79]. Knee OA mostly affects the aging population of which 37.4% are 60 years of age or older [6, 9]. Roughly one quarter of those with doctor-diagnosed OA, reported problems with carrying out activities of daily living [8, 10]. Management of OA is a $128 billion industry [7, 10], that creates an alarming clinical burden with 36 million and 750,000 ambulatory care visits and hospitalizations per year [7]; resulting in high socioeconomic costs [3]. OA is a clinical problem with very few options beyond pain management until the disease progression to a more extreme state ultimately require a more expensive and invasive surgery [12].
There are several different therapies for knee OA: surgical treatments, non-pharmacological treatment and pain management. Surgical treatments include total knee arthroplasty (TKA) or high tibial osteotomy, which are used when non-invasive options no longer able to alleviate pain [80-83]. Surgeries are highly invasive and expensive [84] with TKA costs alone exceeding $11 billion in 2005 [84, 85]. A less invasive and cheaper non- pharmacological therapy is exercise prescription (i.e., cycling and walking) [86] to reduce pain and improve joint function [87]. In subjects with OA, low-intensity cycling is just as effective in decreasing pain, increasing joint function and improving aerobic capacity, as high-intensity cycling [33, 88]. For walking, movement modifications have been extensively studied and found to successfully reduce pain and joint moments (i.e. internal knee abduction moment) associated with knee OA [5, 30, 89, 90]. However, for cycling, movement modifications have not been studied to the same extent. One of our studies found peak internal knee abduction moments decreased when using lateral wedges during stationary cycling [51]. The internal knee abduction moment is used as a surrogate measure for the medial compartment knee loading. Another one of our studies found peak knee adduction angles decreased, but peak internal knee abduction moments did not decrease, for subjects with and without knee OA when cycling with increased toe-in angle [52]. These results suggest that movement modifications during cycling alter kinematics and internal knee abduction moments in knee OA patient and healthy controls alike, but the critical roles muscle forces play during modified cycling exercise therapy remain largely unknown.
Cycling remains an integral component of exercise therapy for individuals with knee OA, despite the fact, that potentially beneficial changes in muscle forces are not well understood for cycling. Since the lower limb muscles, play a more important role in cycling, as cycling requires these muscles to work together and harder than normal gait to carry out and complete the cycling movement. Therefore, it is even more important to examine the roles that associated muscles play in all cycling pedal modifications. Modified cycling exercise may potentially strengthen muscles to improve mobility while optimizing the biomechanics of the diseased knee joint. However, there is little scientific evidence characterizing muscle forces in individuals with knee OA during cycling [47, 48]. One study [12] examined walking modifications (i.e. Toe-in) rather than cycling in OA subjects, found significant muscle force differences were evident within individual subjects, however, there was no consistent muscle force change within the cohort as a whole. This may be due to a self-selected muscle pattern change that were not uniform for gait modification predominantly, since, subjects adopt different gait kinematics in order to decrease an individual’s overall associated knee pain. Therefore, current exercise prescriptions may be improved with new knowledge of how modified cycling affects muscle forces.
This study used dynamic musculoskeletal simulation, in combination with biomechanical movement analysis, to evaluate the muscle force changes for participants with knee OA and healthy counterparts during stationary cycling using four different pedal modifications and a neutral control condition. First, we hypothesized that pedal modifications would cause a change (increase or decrease) in the mean and peak muscle forces compared to the neutral pedal condition for each cohort. Secondly, we hypothesized that mean and peak muscle forces for subjects with knee OA would be different (higher or lower) compared to healthy subjects without knee OA during cycling in each pedal condition. Identifying muscle force differences between OA and the healthy cohorts will contribute to our understanding and help bridge the gap to improving cycling modifications that allow for subject-specific rehabilitation.
2.2 Methods
2.2.1 Subject Demographics
Thirteen subjects with knee OA (7 female, 6 males | 56.85.2 yrs. | 83.222.3 kg | 1.75 0.14 m | BMI:25.63.6
kgm2) and 11 healthy subjects without knee OA (5 female, 6 males | 50.09.7 yrs. | 80.223.1 kg | 1.75 0.12 m | BMI:25.95.4
kgm2), voluntarily participated in this study [52]. Knee OA cohort had medial compartment tibiofemoral OA in at least one of their knees. To be included in this study, subjects had to be between the ages of 50 to 65 years and have a BMI of 35 kg/m2 or less. The knee OA cohort had to be radiographically diagnosed with medial compartment knee OA, with or without patella-femoral knee OA, by a rheumatologist with a grade 1 to 4 on the Kellgren-Lawrence scale. Exclusion criteria consisted of any lower-extremity joint replacement, arthroscopic surgery or intra-articular injections 3 months prior to testing and/or systemic inflammatory arthritis. All subjects were asked to stop pain medication at least 2 days before testing. For more criteria on inclusion and exclusion, please refer to Gardner, et al., 2015 [52]. and Gardner, et al., 2016 [51].
2.2.2 Experimental Motion Capture Data Collection
Prior to the test, participants were fitted to the cycle ergometer in a seating arrangement that standardized seat height, seat depth and trunk angle. Participants performed a warm-up on the cycle ergometer for 3 minutes to asses proper positioning and observation of pain levels. For the test, five trials of cycling in each of the five pedal conditions (Neutral (0°), 5° Lateral wedge, 10° Lateral Wedge, 5° Toe-in and 10° Toe-in) were performed for 2 minutes each at a pedal cadence of 60 RPM and an 80W work rate. A two-minute rest period was provided between trials.
Biomechanical motion capture analysis was used to record kinematics during the cycling trials. Vicon motion capture analysis system (240 Hz) was used to obtain three-dimensional (3D) kinematics during stationary cycling. Participants wore tight fitting spandex shorts and a T-shirt during data collection. To identify joint centers, anatomical markers were placed bilaterally on the 1st and 5th metatarsals, medial and lateral malleoli, medial and lateral epicondyles, left and right greater trochanters, left and right iliac crests and left and right acromion processes. Semi-rigid thermoplastic shells, each with four non-collinear tracking markers, were attached to the trunk, pelvis, thighs and shanks using hook and loop wraps. Three individual tracking markers were placed on the posterior and lateral sides of the heel of standard lab shoes (Noveto, Adidas) worn by all subjects.
An instrumented cycle ergometer (Excalibur Sport, Lode and Groningen, Netherlands) was used during testing. The ergometer used a customized bike pedal, which allowed recordings of 3D pedal reaction forces and moments using two 3D force sensors (Type 9027C, Kistler, Switzerland) connected with two charge amplifiers (Type 5073A and 5072A, Kistler, Switzerland). Sensors could be placed in either the left or the right pedal, depending on the desired limb to be analyzed. A non-instrumented pedal with the same size and mass was used on the opposite side for symmetry and dynamic balancing.
2.2.3 Development and Analysis of Muscle-Actuated Inverse Dynamic Simulations
Subject-specific models were created using the generic, full-body musculoskeletal model [91] in OpenSim, which contains 10 segments, 23 degrees of freedom and 92 muscle–tendon actuators [58, 92-94] (Figure 5). The pelvis position and orientations were defined as 6 degrees of freedom relative to the ground. The head, arms and torso were represented as one segment and connected to the pelvis by a ball-and-socket joint [95]. The hip was modeled as a ball-and-socket joint [96]. The knee was modeled as a planar joint with tibiofemoral and patellofemoral translational constraints as a function of knee flexion [96]. The ankle and subtalar joints were modeled as separate revolute joints [97]. Each muscle–tendon actuator was modeled as a Hill-type muscle in series with a tendon based on published muscle-tendon parameters [58, 92-94, 96]. The model was altered further by adding constraints to the patella in order to model the knee so it articulated in contact with the femur and to allow the muscles to wrap around the patella [76]. The patella functioned to allow the quadriceps forces to act along the patellar ligament line of action. This alteration improved the relationship between knee kinetics, kinematics and muscle forces beyond the original knee model [96] and allowed for a better estimation of the muscle forces.
The procedure for creating and analyzing muscle-actuated inverse dynamic simulation of each individual’s movement consisted of a four-step procedure. First, we took a generic musculoskeletal model placed markers on the model in the same corresponding location as the data collection process then we scaled the generic model in order to account for differences in the subject’s height, mass and muscle moment-generating capacity due to muscle moment arms [57]. The model was scaled by using the musculoskeletal geometry of the generic model and the marker data collected from experimental motion capture analysis (Figure 5). Step two, used inverse kinematics too to solve for the associated model’s joint angles while trying to minimize errors concerning experimental and model markers. Step three, used inverse dynamics along with the inverse kinematics results from the previous step and experimental pedal reaction forces to determine the net joint moments. Step four, used static optimization [56] to determine the muscle activations and corresponding muscle forces that generate the net joint moments. We chose to use the inverse dynamics approach for our large number of simulations (24 subjects, 5 conditions with 5 trials per condition = 600 trials) needed because this approach is a well-established and computationally efficient method for estimating muscle forces during movement.
2.2.4 Evaluation of Muscle Forces and Test of Hypotheses
We evaluated our first hypothesis regarding the changes in mean and peak muscle forces within each cohort by conducting a repeated measures one-way analysis of variance (ANOVA; p
2.2.5 Validation of Models and Simulation Results
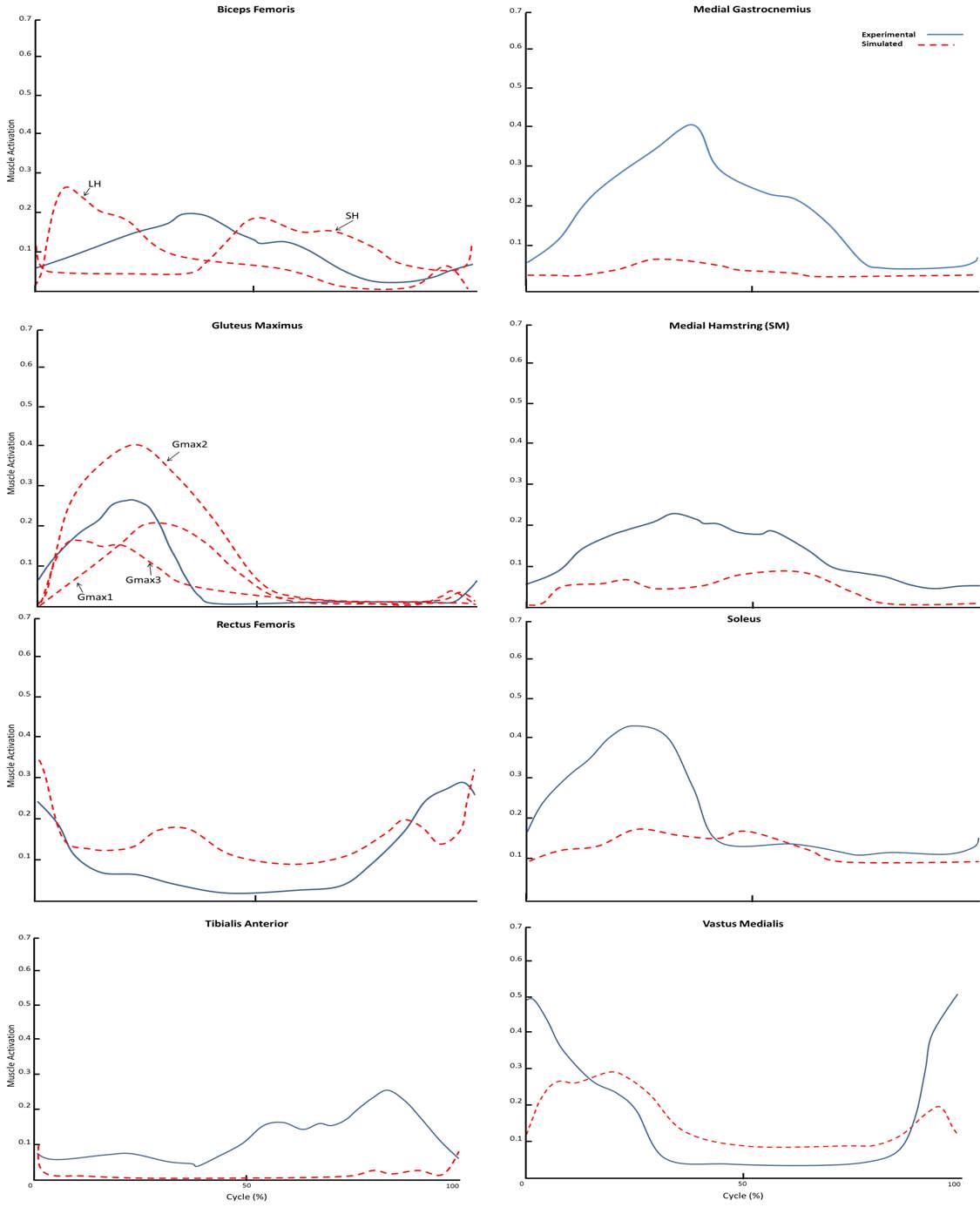
Experimental data, was used during all phases in the development of the inverse dynamic simulations in order to validate the models and results. During scaling of the models, the errors between experimental and model markers were examined and model marker positions were adjusted to minimize marker errors. Each marker was placed in the correct location to match the experimental data. Following inverse kinematics, the maximum and root mean square (RMS) marker errors were examined and marker-tracking weights were adjusted to reduce, these errors. In addition, the resulting hip, knee and ankle joint angles and net joint moments were examined and compared to those from the Visual 3D motion capture analysis software and joint angle data from the literature [98]. These steps were performed to make sure the model was capable of adequately estimating muscle forces with static optimization [99]. Since muscle forces, during cycling in OA subjects and pedal modifications remain unknown. Following static optimization, muscle activations for healthy subjects in neutral cycling were compared to EMG data from the literature (also healthy subjects in neutral cycling) [98]. It is important to note, the data in our study was collected using the same pedal cadence (60 RPM) but a reduced work rate (80 W) compared with the data from Neptune, et al., 1997 [98] used during our muscle force validation processes.
2.3 Results
2.3.1 Mean and peak Muscle Force Changes Across Conditions
There were many changes in the mean muscle forces for all subjects across pedal modifications compared to the neutral pedal condition. This confirmed our hypothesis that modifications would lead to a change in mean muscle forces (Table 1). Subjects with knee OA had 44 significant (pFigure 6a, Table 1). Subjects with knee OA had 56 significant (pFigure 6a, Table 1). The Biceps Femoris Short Head, showed the largest decrease of 1%BW. There were not as many changes in the peak muscle forces (relative to mean muscle force changes) for all subjects across pedal modifications compared to the neutral pedal condition. Nonetheless, our hypothesis was still confirmed that pedal modifications lead to a change in peak muscle forces (Table 2).
2.3.2 Mean and Peak Muscle Force Differences between Cohorts
There were many differences in the mean muscle forces between subjects with and without knee OA in each pedal condition and confirmed our hypothesis that cohort differences would lead to differences in mean muscle forces (Table 3). The neutral pedal condition revealed 26 significant (phealthy) difference of 12%BW. The Gluteus Maximus, showed the largest negative (OA
2.3.3 Validation of Models and Simulation Results
The models and simulations were validated with experimentally measured data. During scaling of the models, model marker positions were adjusted to reduce the error between experimental and model markers in order to be below the acceptable threshold (≤ 2 cm). Following inverse kinematics, marker-tracking weights were adjusted in order to reduce these errors below an acceptable threshold (maximum error ≤ 3 cm and RMS error ≤ 2 cm). In addition, the lower-limb kinematics compared well to those from Visual 3D [51, 52] for this study and published kinematic data [98]. Following inverse dynamics, the lower-limb kinetics compared well to those from Visual 3D and published kinematic data [98]. The muscle activations compared well to published EMG data [98] considering the similarities in pedal cadence and differences in work rate (Figure 7).
2.4 Discussion
This study examined the muscle force changes for subjects with and without knee OA during stationary cycling using five different pedal conditions. The findings support our hypotheses that pedal modifications change the mean and peak muscle forces within cohorts and those differences exist between the OA and healthy cohorts. Our findings suggest that subjects with knee OA have a greater potential when compared to healthy subjects (without knee OA) to significantly change their muscle forces overall with pedal modifications. Additionally, toe-in pedal modifications caused the largest changes for subjects with knee OA. Significant muscle force differences existed between subjects with and without knee OA. Interestingly, the muscle forces crossing the hip and knee generally showed mostly negative (OA
The current study is limited in a few aspects. First, static optimization was used to estimate muscle forces in our simulations rather than dynamic optimization. We chose static optimization due to the large number of simulations needed and the low computational expense associated with this approach. However Anderson and Pandy (2001), found the results of static and dynamic optimization to be essentially equivalent for estimating in vivo quantities like muscle forces [56]. The absolute magnitude of muscle forces may change if we made different modeling assumptions, but our conclusions regarding the relative muscle force changes would likely not be different because the same assumptions were used across all cycling simulations. Second, the experimental data collection did not include EMG for our subjects. Even though we did not have EMG data, our results matched experimentally collected data from neutral cycling in elite male athletes [98]. Lastly, the data was collected with only one instrumented pedal (force plate); this caused us to only analyze one side of the participants. However, this limitation will not alter the results of the analyzed leg.
Despite these research challenges, our results are consistent with findings of others. Muscles activations for our healthy subjects were similar to EMG data from Neptune, et al. [98], with the same pedal cadence of 60 RPM but a higher work rate of 120 W compared to our study at 80 W. In addition, the hip, knee and ankle angles [39] and moments [37, 39, 100-102] compared well to previous research for the healthy subjects at various pedal cadences and work rates. The pedal reaction forces also compared well noting differences in pedal cadence and work rate [100, 103]. Because of these consistencies, we are confident in our simulation results, without EMG data and with the use of static optimization results for our muscle force estimation.
What has not been answered, in previous literature until now is the question of whether individuals with and without knee OA have different muscle forces during cycling in addition to different pedal modifications. Previous literature focused on elite or competitive athletes [35, 37, 38, 98, 103, 104], while cycling at different pedal cadences and work rates. For example, Neptune, et al. [98], examined elite male athletes and the effects of pedaling rate on muscle activation. Our study investigated and compared both subjects with and without knee; OA and none of the subjects were competitive cyclists. Previous literature, has not examined the muscle force changes of knee OA and healthy cohorts in the same study with different pedal modifications (lateral wedge and toe-in). These pedal modifications alter the joint kinematics and kinetics, which may have in turn changed joint moment arms generated by muscle forces. Furthermore, these cycling pedal modifications, the muscle lengths, velocities and moment arms, affect muscle force contributions to joint contact loads.
Subjects with OA showed to have been affected more by the pedal modifications and had a greater overall ability to increase their muscle forces in their mean and peak muscle forces compared to neutral condition. Subjects with knee OA had more statistically significant differences, in the mean and peak muscle forces in the toe-in conditions. Subjects without knee OA were not as able to increase their muscle forces in all pedal modifications, compared to neutral; conversely, they were more affected by the lateral wedge condition compared to neutral. This is opposite what was seen in the OA cohort. This may be impart due to the altered kinematic adaptations from a normal more neutral joint positioning allowing them to maintain stability in their kinematics to decrease the associated overall knee pain. Overall, the mean and peak muscle forces were very similar some were not seen as to be statistically significant in the peak muscle forces but were in the overall mean muscle forces. This may be due to the mean muscle forces minimum force increased or decreased while maintaining the same overall peak muscular force. This newly found information on muscle forces will aid in improving rehabilitation strategies targeting muscle strength and improving joint function. Since knee, OA has been linked to a reduction in muscular strength, endurance and speed, which in return may alter joint kinematics and may worsen the joint space narrowing in the medial compartment. One study [105], has shown that muscle rehabilitation enhanced activities of daily living and muscle function, which in return reduced difficulty carrying out activities and overall pain in knee OA subjects. Where the subjects with OA, were able to increase their muscular force in order to improve the knee joint kinematics. If a subject can maintain their physical activity while increasing, their muscular strength that will help improves their knee joint function and mobility; while, decreasing associated pain and possible slowing the progression of OA.
In conclusion, this study showed exercise rehabilitation involving cycling with different pedal modifications has a greater effect on the mean and peak muscle forces of subjects with knee OA compared to healthy subjects without knee OA. More specifically, toe-in pedal modifications had a greater effect than lateral wedge modifications on subjects with knee OA. The effects on muscle forces causing either an increase or decrease depended on which joint(s) each muscle spanned. Regardless of pedal condition, the subjects with knee OA showed significant muscle force differences compared to healthy subjects, which also depended on the joint(s) each muscle spanned. We have shown how cycling modifications change muscle forces for improving muscle strength during exercise rehabilitation in subjects with knee OA, but further research is needed to determine the effects of muscle forces on the joint contact loads using cycling modifications. Joint loading plays a crucial role in the progression of joint degeneration and severity of pain during exercise and activities of daily living; therefore, a better understanding of muscle forces and their effects on the joint contact loads is necessary for designing safe and effective exercise rehabilitation therapies.
Chapter III
Lower-Limb Joint Contact Loads Adapt Differently in Individuals with and without Knee Osteoarthritis during Cycling with Pedal Modifications
Abstract
Osteoarthritis (OA) is a clinical problem affecting an estimated 27 million adults in the United States with the only clear treatment options being pain management. Cycling is an integral component of exercise for individuals with knee OA, while the joint contact loads during cycling remain unknown. Thirteen subjects with medial compartment knee OA and eleven healthy subjects performed a cycling protocol with a neutral pedal and four pedal modifications. Six hundred muscle-actuated inverse dynamic simulations (24 subjects, 5 trials in each of 5 conditions) were performed to estimate joint contact load differences between conditions. Subjects with knee OA had 11 (lateral wedge) and 12 (toe-in) significant changes. Healthy subjects had 22 (lateral wedge) and 14 (toe-in) significant changes. When comparing the cohorts the neutral pedal condition revealed 7 significant differences, the lateral wedge conditions revealed 14 significant differences and the toe-in conditions revealed 16 significant differences. This study showed, while cycling in different pedal modifications the knee OA subjects had more beneficial changes in their knee adduction-abduction moment compared to subjects without knee OA. Lateral wedge modification had a greater impact on the subjects with knee OA. Subjects with and without OA had greater contact forces at the hip and ankle across pedal modifications compared to neutral. Subjects with OA mostly decreased their knee contact forces but healthy subjects without OA mostly increased these forces across all joints and with all pedal modifications.
3.1 Introduction
Osteoarthritis (OA) is a joint disease altering the quality of life [5] for an estimated 27 million people in the United States [6]. OA causes a breakdown and wear on joint cartilage triggering disabling pain and disability [79]. Knee OA usually affects the aging population, 60 years of age and older, 37.4% are affected by OA [6, 9]. Of those with OA, one-fourth reported problems with activities of daily living (ADL) [8, 10]. Treatment of OA is a $128 billion industry [7, 10] creating a disturbing clinical burden with 36 million ambulatory care visits and 750,000 hospitalizations per year [7] creating high socioeconomic expense [3]. OA is a clinical problem with few solutions beyond pain management until the disease progresses far enough needing invasive, expensive surgery [12].
One common treatment used in current rehabilitation for knee OA is the prescription of exercise (i.e., walking and cycling) [86, 106], though effects of exercise modifications on joint contact loads are not well understood. Subjects with knee OA, low-intensity cycling is as effective as high-intensity cycling for improving joint function, decreasing pain and increasing aerobic capacity [33]. Cycling’s health benefits include potentially reduced OA-related joint contact loads; however, there is little scientific evidence of magnitude and location of these contact loads during cycling. Loading plays a crucial role in progression of joint degeneration and severity of pain during exercise and ADL [5, 34]; thus, a better understanding of how different cycling modifications affect joint contact loads is necessary for improving knee OA related treatments. The external knee adduction moment and the internal knee abduction moment is used as an indirect measurement for the medial loading in the knee and has been linked to the progression and severity of OA [42]. However, these moments, estimated from inverse dynamics, does not account for muscle force contributions to the joint contact loads and is not always a proper representation of the medial load at the knee [107]. Cycling modifications have not been studied as often as walking [30-32]. One study [51] found a 22% decrease in peak internal knee abduction moment ( 10° lateral wedge) and another study [52] found a decrease in the peak knee adduction angle for subjects with (61% for 5°, 73% for 10°) and without knee OA (77% for 5°, 109% for 10°). No studies have shed light on OA-related joint contact loads during cycling with pedal modifications.
This study used muscle-actuated inverse dynamic simulations, in combination with experimental data, to identify differences pedal modifications have on knee joint contact loads for subjects with and without knee OA during cycling. First, we hypothesized pedal modifications will cause a change (increase or decrease) in peak joint contact loads compared to neutral pedal condition for each cohort. Secondly, we hypothesized that peak joint contact loads for subjects with knee OA will be different (higher or lower) compared to healthy subjects during cycling in each pedal condition. Identifying effects of modified cycling exercise on joint contact loads and differences between the cohorts will contribute to a better understanding for improving the treatment of knee OA with cycling exercise, ultimately allowing for patient-specific rehabilitation.
3.2 Methods
3.2.1 Subject Demographics
Twenty-four subjects participated voluntarily for this study: 13 subjects with medial compartment tibiofemoral (knee) OA (7 female, 6 males | 56.85.2 yrs. | 83.222.3 kg | 1.75 0.14 m | BMI:25.63.6
kgm2) and 11 subjects without knee OA (5 female, 6 males | 50.09.7 yrs. | 80.223.1 kg | 1.75 0.12 m | BMI:25.95.4
kgm2) [52]. To be included in this study, all subjects were between 50 and 66 years of age and had a BMI of no more than 35 kg/m2. A rheumatologist had diagnosed subjects with medial compartment knee OA. They were excluded, if they had a previous lower extremity joint replacement, had OA in hip or ankle, had a previous arthroscopic surgery or intra-articular injections prior to testing and/or had a type of systemic arthritis. Two days prior to testing all subjects were asked to stop pain medication. For more criteria on the inclusion and exclusion, please refer to Gardner, et al. [51, 52].
3.2.2 Experimental Motion Capture Data Collection
Participants performed an experimental protocol including warm-up, cycle fitting, cycling trials and rest periods. Participants performed 3 minutes of treadmill walking at a self-selected pace, which served as an initial warm-up. Participants were asked to sit on a cycle ergometer and researchers fit it to each subject accordingly by adjusting: seat height, seat depth and trunk angle. The participants were asked to warm-up on the cycle ergometer for 3 minutes to asses proper positioning and observation of pain levels. Each trial of cycling was performed for 2 minutes at a pedal cadence of 60 RPM and an 80W work rate. Creating 600 trials, where 24 subjects each performed five trials in five pedal conditions. For more information on the data collection process, please refer to Gardner, et al.[51, 52].
3.2.3 Development and Analysis of Muscle-Actuated Inverse Dynamic Simulations
We created subject-specific models using the generic, full-body musculoskeletal model [91] in OpenSim that contains 10 body segments, 23 degrees of freedom and 92 muscle–tendon actuators (Figure 5). The pelvis is defined as 6 degrees of freedom relative to the ground. Another segment was the head, arms and torso which were represented as one segment that connected to the pelvis through representation as a ball-and-socket joint [95]. Another ball-and-socket joint was used to model the hip joint [96]. The knee was modeled as a planar joint with tibiofemoral and patellofemoral translational constraints as a function of knee flexion [96]. The ankle and subtalar joints were designed as separate revolute joints [97]. We enhanced our musculoskeletal model further by adding a patellar ligament constraint to the model so that it articulated with the femur and allowed the muscles to constrain and wrap around the patella [76]. This allowed the patella to function as a pulley that forces the quadriceps forces to act in the corresponding direction of the patellar ligament. This refined the muscle force estimates for generating the measured knee kinematics and it allowed for a more accurate estimation of the joint contact loads.
The procedure for creating and analyzing muscle-actuated inverse dynamic simulation of each individual’s movement consisted of a four-step procedure. Step one, we modified an existing, generic model of the musculoskeletal system and scaled it to account for differences in the subject’s height, mass properties and muscle moment-generating capacity due to the muscles moment arms [57]. The model was scaled (Figure 5) by using the musculoskeletal geometry of the generic model and the marker data collected from experimental motion analysis. Step two, we used inverse kinematics to solve for the model’s joint angles to minimize errors between experimental and model markers. Step three, we used inverse dynamics along with kinematics from the previous step and experimental pedal reaction forces to determine the net joint moments. Step four, we used static optimization [56] to determine the muscle activations and corresponding muscle forces that generate the net joint moments. We chose to use the inverse dynamics approach for our large number of simulations (600 total simulations= 24 subjects x 5 conditions x 5 trials per condition) created because this approach is a well-established and computationally efficient [56].
3.2.4 Estimation of Joint Contact Loads
To determine the effects of the pedal modifications on the joint contact loads, the contact forces and moments (i.e., contact loads) were estimated using the joint reaction analysis in OpenSim [108]. This analysis is used to estimate joint contact forces and moments that are transferred between two adjacent bodies accounting for the models motion and all forces acting on the model including muscle-tendon actuators [58, 92-94]. This tool takes into account the muscle forces that were calculated in step four, the kinematic movement in step two and the pedal reaction forces from the experimental data collection. The joint reaction analysis results in six different outputs, including three contact forces in the anterior-posterior shear, compression and medial-lateral shear as well as three contact moments in the adduction-abduction, internal-external rotation and flexion-extension all of which are expressed in the child reference frame (tibia). This analysis is used to extract information from the subject-specific model and cycling simulations to better understand the experimentally measured motion capture data [108]. All joint contact forces were normalized to body weight and joint contact moments to body weight time’s height.
3.2.5 Tests of the Hypothesis
We evaluated our first hypothesis regarding the differences in peak joint contact loads (forces and moments) within each cohort by conducting a repeated measure one-way analysis of variance (ANOVA) in Matlab 2015 at a significance level set to 0.05. An ANOVA tells us if our results are significant though it does not tell you where the actual significance may be. We then decided to apply a post-hoc pairwise comparison using the Tukey-honest significant differences (Tukey-hsd) in order to determine where the significances may be located. This was done for all four pedal modification compared to neutral to detect significant differences between joint contact loads. Peak joint contact loads were chosen for the first peak in the joint contact loads during the power phase of the cycle (0°-180°). We evaluated our second hypothesis regarding the differences in peak joint contact loads between cohorts (knee OA and healthy) by conducting an ANOVA of unequal sample size with a level of significance at 0.05. Then again, the Tukey-hsd post-hoc correction was applied in order to find the associated significant differences. After these tests were carried out we created identity grids in order to analyze and summarize the significant change joint contact loads between pedal modifications compared to neutral pedal condition as well as when comparing the two cohorts of subjects with and without knee OA in the same corresponding pedal condition.
3.2.6 Validation of the Model and Simulation Results
Experimental data was used to validate the 24 inverse dynamics subject-specific models and 600 simulations. Every subject’s model markers were placed in precise locations to match experimentally collected data. For scaling the models marker location were inspected and the positions were moved and adjusted in order to match experimental positions and reduce error from static markers. They were then compared to see if they were underneath the acceptable threshold (≤ 2 cm). Then after inverse kinematics was carried out, the root mean square (RMS) and peak marker errors were examined, as well as the weighting for tracking of the markers was adjusted to reduce these errors under a suitable threshold (RMS error ≤ 2 cm and maximum error ≤ 3 cm). After inverse kinematics was completed, it was then compared to the experimental kinematics. Specifically the resulting joint angles (hip, knee and ankle) were examined visually and compared to those from Visual 3D motion capture analysis software [51, 52]. After inverse dynamics was carried out, the resulting net joint moments were compared the experimental data [51, 52] for the hip, knee and ankle. After static optimization the muscle activations for the healthy subjects in neutral cycling were compared to published EMG data [98] with the understanding that there are similarities in the pedal cadence (60 RPM) and a difference in the work rate (80 W) (Figure 7). These steps were done according to literature and previous research in order to make the model “good enough” [99].
3.3 Results
3.3.1 Lateral Wedge Peak Joint Contact Load Differences across Conditions
There were many changes in the peak joint contact loads for the subjects with and without knee OA in the lateral wedge condition compared to the neutral pedal condition and this confirmed our hypothesis that these modification would lead to changes in peak joint contact loads (Table 4). All of the peak percent changes were compared to the neutral pedal condition. Subjects with knee OA had 11 significant (p
Healthy subjects had 22 significant (p
3.3.2 Toe-in Peak Joint Contact Load Differences across Conditions
There were many changes in the peak joint contact loads for the subjects with and without knee OA in the toe-in modification compared to the neutral pedal condition, which confirmed our hypothesis that modifications would lead to changes in peak joint contact loads (Table 4). Subjects with knee OA had 12 significant (p
Healthy subjects had 14 significant (p
3.3.3 Peak Joint Contact Loads Differences between Cohorts
There were many differences in the peak joint contact loads for the subjects with knee OA in each pedal condition compared to healthy subjects and this confirmed our hypothesis that cohort differences would lead to differences in peak joint contact loads (Table 5). The neutral pedal condition revealed 7 significant (phealthy) difference by 76%; while, there was no obvious negative (OA
3.4 Discussion
We examined the effects of five different pedal modifications on the joint contact loads during cycling. We confirmed that pedal modifications cause changes in the peak joint contact loads compared to the neutral pedal condition for each cohort. Moreover, we confirmed that the peak joint contact loads for subjects with knee OA are different compared to subjects without knee OA while cycling in each pedal condition. Our findings suggest that subjects with knee OA have a greater potential than subjects without knee OA to significantly improve their important joint contact loads (knee adduction-abduction moment) overall by using pedal modifications. Conversely, the healthy subjects were affected overall more harshly (by increasing joint contact loads) using the pedal modifications compared to the neutral condition, more than the OA cohort did. Furthermore, these changes for the subjects with knee OA were mostly increases in the hip and ankle contact loads, but mostly decreases at the knee when compared to the neutral pedal condition. The changes for healthy subjects without knee OA were mostly increases in the hip, knee and ankle contact loads compared to the neutral condition.
The current study has a few research challenges and results should be interpreted in the context of these limitations. First, the use of static optimization to estimate muscle forces instead of dynamic optimization. Static optimization was chosen due to the large amount of simulations created (600) and the low computational expense associated with this approach. Additionally, it has been previously determined by Anderson and Pandy (2001) that static and dynamic optimization is essentially equivalent for estimating in vivo quantities such as muscle forces and joint contact forces for gait [56]. Therefore, the results from static optimization should not affect our conclusions drawn from this study. Second, electromyography (EMG) data was not obtained from the data collection process for our subjects. Even with not having EMG, our results of healthy subjects matched well with experimentally collected data from elite male athletes in neutral pedal condition [98]. Third, the load values reported in the current study represent the whole knee joint contact loads, rather than narrowing in on the medial compartment where subjects exhibit radiographic evidence of knee OA. The adduction-abduction moment relates to the unbalanced forces compressing the medial and lateral compartments and contribution to the bone-on-bone contact characteristics leading to knee OA, thus finding a reduction in this contact load shows an improvement for subjects with medial knee OA. Fourth, the data was obtained with only one instrumented force plate (in the pedal); this restriction only allowed us to only analyze one side of the participants, though this will not alter the results of the analyzed leg. Although the magnitude of joint contact loads may change if we made different modeling assumptions, our conclusions regarding the relative changes in these loads during modified cycling compared to neutral cycling would unlikely change significantly since the same assumptions would be used across all 600 simulations.
Despite these research challenges, our results are consistent with findings of others. For example, our hip, knee and ankle joint angles during cycling with a neutral pedal condition were consistent with previous literature using similar conditions [51, 52]. The muscles activations from the subjects without knee OA were similar to EMG data from another study carried out Neptune, et al. [98], with the same pedal cadence (60 RPM) and a higher work rate of 120 W versus our 80 W. We also visually compared the joint angles of the hip, knee and ankle [39] as well as the moments [37, 39, 100-102] and both compared well to previous studies for the subjects without knee OA at a different work rate. The pedal reaction forces compared well noting differences in pedal cadences and work rates [100, 103]. In addition, our results agreed with the experimental study carried out by Gardner, et al. [51, 52], where they found a decrease in the peak internal knee abduction moment (related to medial compartment load) for lateral pedal wedge conditions rather than toe-in pedal conditions.
What has not been discovered in previous research until now is the question of whether subjects with and without knee OA have different joint contact loads during cycling that may change with different cycling modifications. Previous literature focused on calculating the magnitude of the inverse dynamic moments, which does not take into account the muscle forces contributing to joint contact loads while cycling at different pedal modifications, cadences and work rates [36, 38, 102, 109]. For example, Mornieux, et al. [102], examined male cyclists while testing the utility and robustness of the estimated joint moments during cycle. Our study investigated and compared both subjects with and without knee OA and all subjects were not competitive cyclists. Previous literature has not examined the effects of cycling modifications (lateral wedge and toe-in) on the joint contact loads in subjects with and without knee OA. These pedal modifications alter the kinematics, which in turn changes the joint moments generated by the muscle forces [41]. This newly found information on the joint contact loads will aid in improving rehabilitation prescription.
Though one previously published paper that examined the lateral pedal wedge condition found that for the peak internal knee abduction moment which is used by scientists as a measurement for the medial compartment loading of the knee. There was an effective decrease in the lateral wedge condition by reducing the internal knee abduction moment by over half compared to previous literature on walking. Our use of the joint contact loads which takes into account the estimated muscle forces and there effects on the joints overall motion also found a decrease for the subjects with knee OA in the lateral wedge conditions, where the 5° wedge decreased by just about half compared to the neutral condition. The 10° decreased by over three-fourths compared to the neutral condition. Subjects without knee OA did not show any statistically significant difference in the lateral wedge condition. Another previously published toe-in study [52] which also examines the internal knee abduction moment found that this pedal was not effective in reducing the internal knee abduction moment across all participants. In contradiction to these findings, we found in subjects with knee OA they were able to reduce their adduction-abduction moment compared to neutral in both the 5° and 10° toe-in condition. Where the 5° toe-in was able to reduce the adduction-abduction moment by over half and 10° toe-in was able to decrease the same moment by almost sixty percent compared to neutral. Conversely, for the subjects without knee OA they were able to only show a significant reduction in the 10° toe-in by about one-fifth compared to neutral. These different numbers may be because joint contact loads take into account the estimated muscle forces as opposed to the knee abduction moments calculation without input from the associated muscle forces that span or innervate the joint. These numbers might also be significant within individual subjects but not across the cohort as a whole which may be skewing the results. Since individual muscles’ contribution to dynamic movements branches from the context in which a muscle accelerates and affect joints in which they do not span and segments in which they do not innervate. Hence, in order to examine the knee joint contact loads, the critical input from the muscles is necessary in order to examine the influence cycling movement has on the joint contact loads.
In conclusion, this study showed different pedal modifications have a greater effect on the knee adduction-abduction moments for subjects with knee OA compared to subjects without knee OA. More specifically, lateral wedge pedal modifications had a greater effect than toe-in modifications on subjects with knee OA, which allowed them to decrease their knee adduction-abduction moment when compared to the neutral condition. Regardless of pedal condition, the healthy subjects had more significant differences (mostly increases) when compared to subject with OA. This study has shown how cycling modifications change joint contact loads for improving exercise in subjects with knee OA, but further research is needed to optimize cycling in order to see what kinematics and coordination patterns minimize the knee’s adduction-abduction moment and the compressive load related to OA progression. Joint loading plays a crucial role in the progression of joint degeneration and severity of pain during exercise and ADL; therefore, a better understanding of optimizing (i.e., minimizing) joint contact loads is necessary for designing and adapting exercise prescription for subjects with knee OA.
Chapter IV Investigating Changes in tibiofemoral joint contact loads due to variations in muscle activity during stationary cycling
Abstract
During activities of daily living, the tibiofemoral experiences a large mechanical loading. Cycling remains a crucial exercise component for subjects with OA; it decreases the subject’s joint contact loads, while increasing their aerobic capacity. Cycling’s health benefits include potentially reduced joint loads on the diseased joint tissues, by increasing the aerobic capacity. The purpose of this study was to determine the changes in the tibiofemoral joint contact loads triggered by variation in muscle activation patterns to minimize the tibiofemoral joint contact loads. Thirteen subjects with knee OA and eleven healthy subjects performed a cycling protocol in neutral pedal condition. Then we modified a static optimization framework to carryout joint load optimizations. We simulated 10,560 trials of neutral cycling with different muscle coordination pattern s and examining the effects on the minimized joint contact loads. The tibiofemoral forces were sensitive to altering the muscle activations of the lower limb muscles. In order to determine the potential for subjects with and without OA to decrease tibiofemoral joint contact loads during cycling by allowing the lower limb to adopt a muscle activation pattern that would achieve the desirable (i.e. minimized) joint contact loads. We determined the maximum muscle activation patterns while minimizing the joint contact loads for subjects with and without OA. Subjects with OA were able to decrease adduction-abduction contact moment more so than subjects without OA. Subjects without OA were not as affected by varied muscle activation patterns. Our model achieved tibiofemoral forces that were lower in both cohorts than the joint contact loads that were previously estimated in joint reaction analysis in both step two and three by adopting an altered muscle activation patterns. This work provides a range of feasible muscle activations that is different than previously calculated or observed in normal cycling kinematics.
4.1 Introduction
During activities of daily living, the tibiofemoral (knee) joint experiences a large mechanical loading. During walking, forces have been shown to be as large as three times the bodyweight about the tibiofemoral (knee) joint [62, 110]. These loads affect the development, maintenance and health of the joint tissues [111]. The progression of OA can be associated with large mechanical loading experienced at the tibiofemoral joint [112, 113] and increased tibiofemoral loads have been linked to pain and progression in subjects with knee OA [114]. Since joint contact loads during walking are produced primarily by muscle forces [115, 116], muscle activation plays a pivotal role in determining tibiofemoral joint contact loads. A previous study [76] examined the use of an optimization framework with a range of muscle activation patterns that varied, in order to calculate the joint forces resulting from muscle activations that reproduced the subject’s walking dynamics. Cycling, rather than other exercise modalities, is considered an alternative exercise for subjects with OA. The goal of cycling is to improve a subject’s overall health (i.e. weight management) and increase their aerobic capacity. Low-intensity cycling was shown to be as effective as high-intensity cycling for improving joint function, decreasing pain and increasing aerobic capacity [33, 88].
Cycling remains a crucial exercise component for subjects with OA; it decreases the subject’s joint contact loads, while increasing their aerobic capacity. Cycling’s health benefits include potentially reducing joint contact loads on the diseased joint tissues, by increasing the aerobic capacity. Cycling has a wide range of muscle activation patterns, but the effect of varied muscle activations on tibiofemoral contact forces remains unclear. This loading plays a crucial role in the progression of joint degeneration and severity of pain during exercise and activities of daily living [5, 34]; thus, a better understanding of how different muscle activation patterns affect the joint contact loads is necessary for the improvement of treatment with movement modifications.
Studying the effects of altered muscle coordination on tibiofemoral loads is challenging because a direct measurement of tibiofemoral loads during cycling requires an instrumented prosthetic knee to be implanted. This technique provides important information, but it is highly invasive, making measurement of tibiofemoral loads unreasonable in healthy subjects and limiting the data set to the number of OA subjects willing to receive this type of replacement taking away the OA aspect of the knee. An alternative to direct measurement is calculating the tibiofemoral contact loads using musculoskeletal modeling. Model-based studies have calculated tibiofemoral (knee) loads using a diverse assortment of muscle coordination strategies, like minimizing muscle activations or activity [117, 118], muscle stress [119] and energy consumption [120]. A previous study has determined a single set of muscle activation pattern forces during walking and the resulting tibiofemoral loads [76].
The purpose of this study was to determine the changes in the tibiofemoral joint contact loads triggered by variation in muscle activation patterns to minimize the tibiofemoral joint contact loads. First we evaluated the effect (increase or decrease) specific muscles have on the tibiofemoral joint contact loads during cycling by altering the muscle activation weightings to alter the tibiofemoral joint contact loads. Secondly, we determined a set of muscle activation patterns that sought to minimize the joint contact loads, by adapting to a varied muscle activation pattern. Finally, we wanted to identify the muscles with the greatest potential to alter tibiofemoral joint loading by determining the changes in tibiofemoral joint contact loads due to a muscle activation framework that wanted to maximize muscle activation patterns of the lower limb.
4.2 Methods
4.2.1 Subject Demographics
Cycling data was used from 13 subjects with tibiofemoral OA (7 female, 6 males | 56.85.2 yrs. | 83.222.3 kg | 1.75 0.14 m | BMI: 25.63.6
kgm2) and 11 healthy subjects without tibiofemoral OA (5 female, 6 males | 50.09.7 yrs. | 80.223.1 kg | 1.75 0.12 m | BMI: 25.95.4
kgm2BMI), all of whom voluntarily participated in this study. This data was previously collected and published by Gardner, et al. [51, 52]. The subject’s data include three-dimensional kinematics, marker positions, pedal reaction forces measured during cycling at a pedal cadence of 60 RPM and a workload of 80W. For more information on the human subjects and the data collection process, please refer to Gardner, et al. [51, 52].
4.2.2 Development and Analysis of Muscle-Actuated Inverse Dynamic Simulations
We created subject-specific models using the generic, full-body musculoskeletal model [91] in OpenSim that contains 10 segments, 23 degrees of freedom and 92 muscle–tendon actuators [58, 92-94] (Figure 1). The pelvis position and orientations were defined as 6 degrees of freedom relative to the ground. The head, arms and torso were represented as one segment and connected to the pelvis by a ball-and-socket joint [95]. The hip was modeled as a ball-and-socket joint [96]. The tibiofemoral joint was modeled as a planar joint with tibiofemoral and patellofemoral translational constraints as a function of tibiofemoral flexion [96]. The ankle and subtalar joints were modeled as separate revolute joints [97]. We improved our model further by adding a patellar constraint to model so that it articulated with the femur and in order to allow the muscles to wrap around the patella [76]. The patella functioned as a pulley that allows quadriceps forces to act in the line of action of the patellar ligament. This refined the muscle force estimates for generating the measured tibiofemoral kinematics and it allowed a more accurate estimation of joint contact loads.
We used the full-body model to simulate 3D cycling dynamics of the subjects with tibiofemoral OA. The procedure for creating and analyzing muscle-actuated inverse dynamic simulation of each individual’s movement took place in three-steps. Step one, we modified an existing, generic model of the musculoskeletal system and scaled it to account for differences in the subject’s height, mass properties and muscle moment-generating capacity due to the muscles’ moment arms (Figure 5) [57]. The model was scaled by using the musculoskeletal geometry of the generic model and the marker data collected from experimental motion analysis. Step two, we used inverse kinematics to solve for the model’s joint angles to minimize errors between experimental and model markers. Step three, we used inverse dynamics along with kinematics from the previous step and experimental pedal reaction forces to determine the net joint moments. After step three, we went on to modify the static optimization framework that was developed [76].
4.2.3 Static Optimization and Joint Reaction Analysis
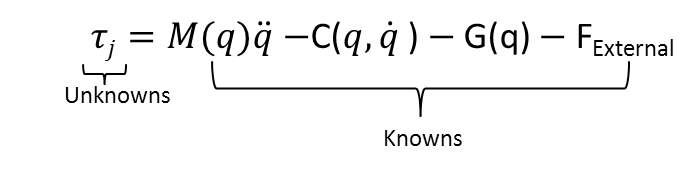
(1)
Before we can modify and determine an optimization framework, we must first run standard static optimization to determine the normal muscle forces and joint contact loads with the recorded and given kinematics. This tool steps through each time frame of a motion and computes the muscle activations in the model that generate the experimental kinematics. The equations of motion relate the model’s accelerations to the muscle moments and the musculoskeletal geometry relates those muscle moments to the muscle forces. The muscle contraction dynamics relate the muscle forces to the muscle activations. The kinetics (Equation 1) necessary to accelerate the model are determined using Newton’s Second Law by treating the skeleton as a set of interconnected rigid-bodies with inertial properties. Where
τj is the set of joint torques,
q,
q̇ and q̈ are the generalized coordinates, their velocities and accelerations, respectively. The M(q)
q̈ is the mass matrix which depends on the coordinates and inertial properties of the model. The C(q, q̇) is the combination of Coriolis and centrifugal forces, which depends on the coordinates and their velocities. The G(q) is the gravitational force, which depends on the coordinates and FExternal is pedal reaction forces and moments applied to the model that were collected in the data collection process.
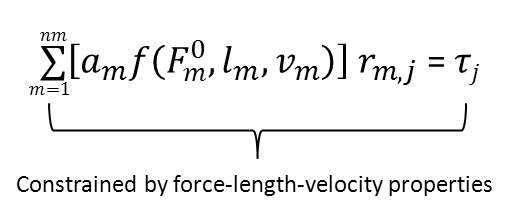
(2)
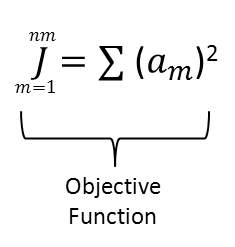
(3)
The motion of the model is completely defined by the coordinates, velocities and accelerations. Consequently, all of the terms on the right-hand side of the equation of motion are known. The remaining set of joint torques (τj) on the left-hand side is unknown. The static optimization tool uses the known motion of the model to solve the equations of motion for the unknown joint torques and is subject to a constraint (Equation 2) while minimizing the objective function (Equation 3). Where
nm is the number of muscles in the model,
am is the activation level of muscle m at a discrete time step,
Fm0 is its maximum isometric force,
lm is its muscle length and
vm is it is shortening velocity.
The fFm0, lm, vm, is a function of its force-length-velocity surface, rm, j is its moment arm about the jth joint axis and τj is the generalized force acting about thejth joint axis.
After this procedure, we ran joint reaction analysis in order to determine the effects of the pedal modifications on the joint contact loads. The contact forces and moments were estimated in OpenSim using the joint reaction analysis tool [108]. This analysis is used to estimate joint contact forces and moments that are transferred between two consecutive bodies because of all motions and forces acting on and within the model including the muscle-tendon actuators [58, 92-94]. This tool takes into account the outputs from static optimizations (i.e., muscle forces), the derived inverse kinematic motion and the pedal reaction forces from the experimental data collection process. The joint reaction analysis results in six outputs, including three contact forces in the anterior-posterior shear, compression and medial-lateral shear as well as three moments in the adduction-abduction, internal-external rotation and flexion-extension all of which are expressed in the child reference frame (or as experienced by the tibia).
4.2.4 Creating the Objective Function
From this point we went on to create a joint load optimization [76] framework that was previously modified from the static optimization framework stated above. This was done in order to calculate individual muscle forces and resulting net joint contact loads of interest for each trial of cycling. This modified optimization strategy minimized the sum of the muscle activations and joint contact loads by combining them in a single objective function:
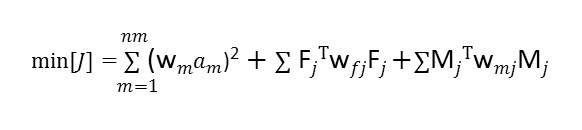
(4)
The objective function was subjected to the same extent and constraints as static optimization such that the estimated muscle forces and measured pedal reaction forces balanced all inertial forces to carry out the measured cycling dynamic motion (Equation 1-3). In the objective function shown in Equation 4,
a m,was the activation of the mth muscle, which could vary anywhere between an activation of 0 to an activation of 1. The
wm was a muscle weighting constant set to penalize a specific muscles, in order to constrain the muscle in order to activate or not to activate. The joint contact force-weighting constants
wfx, wfy and wfz, were set to penalize the vector components of the jth joint reaction force, Fj. Similar to the joint forces, the joint contact moment weighting constants, wmx, wmy and wmz, were set to penalize the vector components of the jth joint reaction moment, Mj. The joint reaction moments and forces represent the connecting joint contact loads experienced in the connecting joint structures and were calculated using the joint reaction analysis in OpenSim.
4.2.5 Exploring the Muscle Activation and their Effects on the Joint Contact Loads
We then went on to simulate 10,320 trials (24 subjects x 5 Trials x 43 muscles x 2 different muscle weightings) of neutral cycling with a muscle coordination pattern that aimed to minimize muscle activations without regard to the joint contact loads then we will examine the effects on the associated joint contact loads. In this case, the generalized objective function Equation 4 was reduced to:
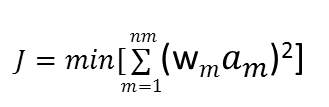
(5)
This was done in order to determine the sensitivity of the tibiofemoral joint contact loads due to an adopted variation in muscle activation patterns of individual muscles in the lower limb by carrying out optimizations that altered the muscle activation weighting constants, wm, for each individual lower extremity muscle. In OpenSim, normal static optimization has a default muscle weighting (wm) of 1 for all associated lower limb muscles. We decided to alter the weighting of the muscle activations that is shown in Equation 5 with a weighting of 0 or 100 for one specific muscle at a time while keeping all other wm=1. Where a wm= 0 represents no penalty and allowing the muscle to activate or be used as much as it wants or needs, conversely, with a wm = 100 turns the muscle completely off and penalizes, prohibits activation or use of a specific muscle.
This was done in order to investigate the variation in tibiofemoral joint contact loads due to different muscle activation weighting by adopting or altering the activations of lower extremity muscles, then examining a specific muscle’s influence on the joint contact loads. To do this we performed two modified optimizations for each muscle and trial of neutral cycling. First, before we altered the muscle activations of specific individual muscles, we ran normal static optimization, which has a muscle weighting of 1 in order to observe normal muscle activation in neutral cycling. Second, we altered the muscle weightings by taking a specific muscle and altering the
wm to exclude activation of a specific muscles, we executed a modified optimization with changing previously established muscle weighting from wm = 1, to wm = 100 for a specific muscle while wm for all other muscles was kept at 1. Third, in order to encourage muscle activation, we then performed a modified static optimization procedure again with wm= 0 for a specific muscle while all others kept it at 1. Performing these two optimizations for each muscle of the lower limb determined the range of tibiofemoral joint contact loads caused by a variation in muscle activations. We determined the change in peak tibiofemoral joint contact loads due to activation of a muscle by computing the change between peak tibiofemoral joint contact loads obtained from the static optimizations with wm = 0 and wm= 100 and the normal static optimization carried out with all wm = 1.
4.2.6 A Muscle Coordination Pattern Minimizing Compressive Tibiofemoral Force
We simulated the 120 trials of neutral cycling with a muscle coordination pattern that minimized the important contact loads of the tibiofemoral joint. In this case, the generalized objective function (Equation 4) simplified to:
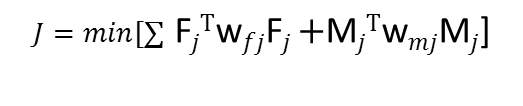
(6)
The weighting of the muscle activations, wm, was set to zero for all muscles in order to not penalize any of the muscles and allow the muscles to adopt a muscle activation pattern that would minimize the joint contact loads of the tibiofemoral joint. We only examined the joint contact loads that have been linked to OA progression, which are the compressive contact load and the adduction-abduction moment. The compressive tibiofemoral contact load was penalized by setting its weighting constant, wfy=1Peak(Fy2) and the adduction-abduction moment was penalized by setting the weight constant, wmx=1Peak(Mx2), while all other joint contact forces and moments were set to zero. This strategy determined a muscle coordination pattern that would minimize the important tibiofemoral joint contact loads that would aid in slowing the progression of medial compartment tibiofemoral OA.
4.2.5 Change in Tibiofemoral Forces Due to Maximized Muscle Activations
We went on to determine the change in tibiofemoral forces due to adopting a maximized muscle activation pattern of the lower limb by performing optimizations with muscle activation weighting constants, wm=-1, for each muscle. The objective function in equation 4 is simplified to:
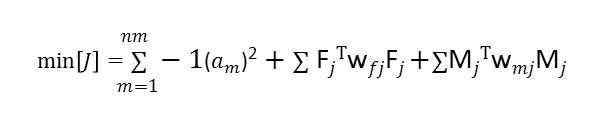
(7)
In Equation 7, wm=-1, this represents no penalty to optimize or maximize the muscle during cycling while minimizing the important tibiofemoral contact loads. The compressive tibiofemoral contact load was penalized by setting its weighting constant, wfy=1Peak(Fy2) and the adduction-abduction moment was penalized by setting the weight constant, wmx=1Peak(Mx2) while all other joint contact forces and moments were set to zero. This strategy identified a muscle coordination pattern that would minimize the important tibiofemoral joint contact loads, while promoting a maximized muscle coordination strategy. This would aid in rehabilitation for the OA subjects by improving the overall muscular strength to have a better overall impact on the joint kinematics to aid in slowing the progression of medial compartment tibiofemoral OA while promoting exercise.
4.3 Results
4.3.1Minimize Muscle Activations
A muscle coordination strategy that minimized muscle activations produced a wide range of tibiofemoral contact loads that were greater than or less than those previously estimated with a normal wm=1, which is from the unmodified normal static optimization procedure with the joint reaction analysis (Table 6). Subjects with knee OA showed an increase for the Medial Gastrocnemius, knee adduction-abduction moment of 255% with wm=0. There was also a decrease of 90% in the knee adduction-abduction moment for the Soleus. Meanwhile, with a wm=100,Adductor Magnus showed an increase by 196% in the knee adduction-abduction moment. The Semitendinosus, showed a decrease of 93% in the adduction-abduction moment. The Sartorius showed an increase of 119% for the knee compressive load with wm=0. As well as Semitendinosus showed a decrease of 70% for the knee compressive load. With a wm=100, the Pectineus showed an increase of 287% for the knee compressive load. The Tibialis Anterior decreased by 20% for the knee compressive load. Consequently, subjects with knee OA mostly increased their knee adduction-abduction moment and compressive load with a wm=1 and a wm=100. While there was some success in decreasing the loads, they were mostly seen in biarticular muscles.
Just like in the OA cohort, the tibiofemoral contact load depended on which muscle was altered (Table 6). Subjects without knee OA showed an evident increase in the Vastus Medialis’s knee adduction-abduction moment of 109% with wm=0. There was also an evident decrease in the Tensor Fascia Lata, knee adduction-abduction moment of 33%. While for a wm=100,Semimembranosus showed an increase of 104% for the knee adduction-abduction moment. The Medial Gastrocnemius, showed a decrease of 21% in the adduction-abduction moment. There were no evident increases in the compressive load with wm=0. While the Vastus Medialis showed a decrease of 18% for the knee compressive load. While the Semimembranosus with a wm=100,showed an increase of 51% for the knee compressive load. While Adductor Brevis showed a decrease of 3% for the knee compressive load. There were many other muscles that showed to have a noticeable effect on the joint contact loads (Figure 8, Table 6). Consequently subjects without knee OA mostly increased their knee adduction-abduction moment with a wm=1 and wm=100. Though their compressive load varied differently in both weightings depending on the muscle and its specific function being altered.
4.3.2 Minimize Joint Contact Forces
A muscle coordination strategy that minimized tibiofemoral joint contact loads produced lower contact loads than the forces that were previously estimated in normal joint reaction analysis (Figure 9). Subjects with knee OA showed a significant decrease of 73% for the knee adduction-abduction moment. There was a noticeable decrease in the compressive load of 96%. For the subjects without knee OA there was a noticeable decrease in the for the knee adduction-abduction moment of 72%. There was also an evident a decrease of 95% for the compressive load. In general subject without knee OA with the given kinematics were affected more than subjects without knee OA. All subjects were able to significantly lower their joint contact loads as shown in Figure 9.
4.3.3 Maximizing the muscle force to minimize the joint loads
A muscle coordination strategy that maximized muscle activations while minimizing the tibiofemoral forces, produced results that further decreased the peak and overall knee joint contact loads while maximizing the muscle forces (Figure 9). Subjects with knee OA portrayed an evident decrease in the knee adduction-abduction moment of 25%. While the compressive load showed a decrease of 19%. For the subjects without knee OA there was also a noticeable decrease in the knee adduction-abduction moment of 81%. However, for the compressive load there was a decrease of 17% for the compressive load. Overall subject without knee OA with the given kinematics were affected more than subjects without knee OA. Just like in the minimized joint contact loads, all subjects reacted differently to the optimization protocol some lowered their joint contact loads more while other did not as shown in Figure 9.
4.4 Discussion
Our results demonstrate that varying muscle activation patterns during stationary cycling can induce large variations in the joint contact loads at the tibiofemoral joint. Evaluating a varied set of different in muscle activation weightings was used in order to evaluate whether a strategy minimizing activations patterns by either turning the muscles on without penalty or turning the muscles off. This was done in order to examine the effects a specific muscles has on the tibiofemoral contact loads to see if they was consistent or different (increase or decrease) from previously estimated joint contact loads. Subjects with knee OA seemed to have achieved more intense results in the knee adduction-abduction moment and compressive load with both muscle variation weightings as they mostly increased; although, this largely depended on the specific muscle function during the cycling motion as well as the joint it spanned. Like the OA cohort subjects, subjects without OA also experienced mostly increases in contact loads compared to normal except with a muscle weighting of zero for the compressive load. These results suggest that treatment interventions meant for retraining muscle coordination should target the specific muscles in the OA cohort that showed a reduction in the adduction-abduction moment and the compressive load in order to reduce tibiofemoral contact loads. Alternatively, avoid the use of these specific muscles, which increase the tibiofemoral loading with an wm=0 in order to lower the mechanical loading at the knee.
We determined the potential for subjects with and without OA to decrease tibiofemoral joint contact loads during cycling by allowing the lower limb to adopt a muscle activation pattern that would achieve the desirable (i.e. minimized) joint contact loads. Our model achieved tibiofemoral forces that were lower than previously estimated in joint reaction analysis with a normal static optimization wm=1 by adopting different muscle coordination patterns that achieved the desired results. Subjects with knee OA were able to reduce their contact loads more than the healthy subjects. We determined a maximized muscle activation pattern while minimizing the joint contact loads for subjects with and without OA. Our model achieved tibiofemoral forces that were lower in all cases than the normal joint reaction analysis and higher than the results of the minimized contact loads, which was to be expected since we wanted to increase the lower extremities muscular strength. This provides a range of feasible joint contact loads that is different from previously calculated or observed in normal cycling kinematics, which had a huge effect overall in both cohorts and on the important joint contact loads.
This current study has a few research challenges and our results should be interpreted in the context of these limitations. First, a modified static optimization framework was used to estimate muscle forces that were used in simulations over the use of a dynamic optimization framework. Static optimization was selected due to the large amount of simulations created (10,560) and the low computational expenditure associated with this approach. In addition, it has been previously validated by Anderson and Pandy (2001) and found that static and dynamic optimization is fundamentally equivalent for predicting in vivo quantities such as muscle forces and joint contact loads for walking [56]. Consequently, the results from static optimization should not alter the conclusions that we have drawn during this study. Secondly, the data collection procedure did not include EMG for the 24 subjects. Even though we did not have EMG data, our data was previously matched with normal static optimization during neutral cycling to experimentally collected data from a study that was carried out by Neptune et al. [98] investigating elite male athletes. Additionally, the joint contact load values reported in this study characterize the whole tibiofemoral joint contact loads, rather than the specific narrowing in exhibited in the radiographic evidence of the subjects with medial compartment tibiofemoral OA. However, adduction-abduction moment relates to the uneven contact loads that compress the medial and lateral compartments and contribution to the bone-on-bone contact characteristics leading to tibiofemoral OA, thus finding any alteration from the normal contact load shows an improvement or a limitation for subjects with medial tibiofemoral OA. Lastly, the pedal reaction force data was collected only from one instrument pedal; this constraint will not alter the results of the analyzed leg would just be used as a comparison within subjects. Although the joint contact loads may vary if we made different modeling assumptions; however, our conclusions regarding the relative changes in references to the estimated joint contact loads in cycling would not be likely to change drastically. This is due to the face that the same modeling assumptions remained consistent throughout all the simulations and across all subjects.
What has not been answered in previous literature until now is the question of whether or not subjects with and without knee OA have can further reduce their tibiofemoral contact loads by adopting varied set of muscle activations patterns. Previous literature focused on walking with an instrumented knee implant in a healthy subject rather than cycling [76]. For example DeMers et al, [76] used this modified optimization framework in walking. Our study investigated both subjects with and without knee OA and none of the subjects were competitive cyclists. Additionally, previous literature has not examined the use of an optimization framework in knee OA and healthy cohorts during neutral cycling. This newly found information on muscle activations may help improve rehabilitation strategies targeting muscle strength and improving joint function.
This study identified specific muscles that substantially affect tibiofemoral forces during stationary cycling as well as adopted a muscle activation pattern that minimized the joint contact loads. This research has provided a range of feasible muscle activations and now the question is to examine these muscle activations that were obtained in minimized knee joint loading and maximized muscle activations, to figure out how these activations can be obtained as well as what are different in these muscle activations than actual cycling kinematics. Future work needs to discover how to target a specific muscle in order to activate or deactivate it in order to obtain the associated decrease in the contact loads. It should also focus on if and in what pedal modifications can obtain the desired and calculated minimized joint contact loads and the maximized muscle activation while minimizing the important joint contact loads (i.e., compressive and adduction-abduction moment). Further testing is also needed with modeling and simulation, in conjunction with biofeedback. To figure out how to activate and deactivate a specific muscle group to achieve lower tibiofemoral contact loads as well as test the utility of different pedal modifications in order to be able to achieve or target different muscle groups that’s have the greatest affect in lowering the joint contact loads.
Chapter V Conclusion
5.1 Significance of Research
Effective OA rehabilitation training and a proper subject-specific treatment would have enormous potential to significantly reduce the progression of OA degeneration in not only the subjects with OA but would also help the subjects without OA as a preventative measure. However, improving the effectiveness of rehabilitation and treatment of OA programs are challenging because the cause-effect relationship between muscle function and joint biomechanics with respect to OA progression is not well understood. Dynamic modeling and simulations provide the capability to determine the meaningful role of muscles contributions, which is essential to understanding their effect and relationship with the joint contact loads. Developing dynamic simulations helps to define the biomechanical aspects that influence knee motion in order to provide a better treatment protocol. We anticipated that the insights gained from this work would aid in providing new guidelines for designing subject-specific OA treatment programs resulting in a significant drop in OA progression and possible pain.
The biomechanical models and computational tools developed, will find broad application. Numerous studies have been performed to record neuromuscular excitation patterns, understand muscles contraction dynamics, characterize musculoskeletal geometry and quantified multijoint movements and kinematics. However, linking the detailed knowledge of these elements of the neuro-musculoskeletal system to create an integrated understanding of normal and pathological movement remains a major challenge in the application of biomechanics to a wide range of clinical problems and basic science research. The long-term goal of this research was to provide a scientific framework for planning, evaluation and improvement of subject-specific rehabilitation for individuals with knee OA.
This work developed methodologies for interpreting the dynamic functions of muscles during cycling movement, generated novel biomechanical optimization approaches to undercover the muscles forces and joint contact loads during movement and a general computational framework for pursuing further research. The simulations we developed were implemented in freely available musculoskeletal modeling and simulation software [57] which enables us to share the results with other research groups. Over the past few years, there have been more than 165,103 downloads of models, simulations and software from the project’s website (see simtk.org/home/OpenSim) with over 28,096 active users of these freely available biomechanics tools. This project will continue this development. Thus, the aims of this proposed research program were achieved; this work will have a broad impact on the basic studies of neuromuscular biomechanics and for clinical treatment and prevention for the subjects with and without knee OA.
5.2 Research Innovation
The main beliefs that direct the relationships between muscles contributions and dynamic repetitive movement in individuals during stationary cycling have not been discovered until now. For decades, experimental research and approaches have driven our understanding of biomechanics of the human body. However, the lack of ability to experimentally measure muscle forces and ascertain the cause-effect relationships between muscle forces contribution to joint biomechanics has limited research progress over the years. The trouble in defining an individual muscles’ contribution to dynamics movement stems from the circumstance that muscles accelerate and affect joints that they do not span and body segments in which they do not innervate. Thus in order to determine a muscles influence on the movement required a new approach motivated by the use of an instrumental set of tools to accomplish this task.
Muscle-actuated inverse dynamic simulations provide a scientific framework, in combination with complementing experimental approaches for estimating important variables and identifying cause-and-effect relationships.In this study, muscle-actuated inverse dynamic simulations, optimization and motion capture analysis were used to bridge the gap in providing a better understanding of dynamic human movement.
This work advanced rudimentary knowledge and understanding in this arena. It engaged the experimental capabilities of physicians, physical therapist and rehabilitation scientists, engineers’ use of computer simulations, mathematicians and computer scientist to address the biomechanical importance of the research questions. An additional advantage of this research was that the creation of subject-specific musculoskeletal models from a freely available open source software (OpenSim). The results that this dissertation discovered will advance research and accelerate the understanding of the underlying fundamental mechanisms behind biomechanical injuries and treatment of OA.
5.3 Fundamental Contributions
The objective of this research was to determine individual muscle force contributions during cycling in different pedal modifications, in order to assess how individual muscles, affect the tibiofemoral joint contact loads. As well as adapting a varied muscle activation patterns designed to minimize the important joint contact loads. The work presented here was able to accomplish all of these objectives.
Static and biomechanical optimizations are techniques that have been previously used to study muscle contribution to walking, running and other treatment modifications. However, the vital importance was in the development of the subject-specific models and application of these tools to investigate their effect on subjects with and without OA in order to examine and identify the fundamental differences between and within the cohorts. Subject-specific models, were customized and tailored based on the subject parameters from data collection and our specific research question that we posed. Since, OA treatment is often based upon the clinicians, clinical experience to treatment plans. The models that are commonly used by physicians and constructed using data from other patients, these clinical outcomes are unreliable. We developed an approach using computational models based on the engineering mechanics and optimization to predict subject-specific outcomes. We simulated how cycling modifications that were used as a possible treatment option for knee OA, which may properly strengthen the lower extremity muscles while decreasing the subjects important joint contact loads (compressive force and the knee adduction-abduction contact moment) for specific subjects.
5.4 Future Work
This study identified specific muscles forces and joint contact loads in subjects with and with tibiofemoral OA during stationary cycling. As well as found a range of feasible muscle activations that are possible to reduce the joint contact loads. However, the question is to examine the muscle activations that were obtained in study three, to figure out how these activations can be obtained through different cycling modifications, see how to effectively achieve the desired activations and muscle forces mentally and physically receiving biofeedback. Future work should test other cycling modifications in order to discover how to obtain the desired muscle activations, the minimized joint contact loads and the maximized muscle activation. Since our model is lacking biofeedback and does not experience any pain these activations that were achieved through optimization may not be feasible for all subjects. Research also needs to obtain a possible way to receive and give biofeedback through simulated human movement.
List of References
1. C.D.C, Prevalence and most common causes of disability among adults–United States, 2005. MMWR Morb Mortal Wkly Rep, 2009. 58(16): p. 421-6.
2. Hootman, J.M., Helmick, C.G., Brady, T.J., A public health approach to addressing arthritis in older adults: the most common cause of disability. Am J Public Health, 2012. 102(3): p. 426-33.
3. Gerbrands, T.A., Pisters, M.F., Vanwanseele, B., Individual selection of gait retraining strategies is essential to optimally reduce medial knee load during gait. Clin Biomech (Bristol, Avon), 2014. 29(7): p. 828-34.
4. Lawrence, R.C., et al., Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Arthritis Rheum, 2008. 58: p. 26-35.
5. Mundermann, A., C.O. Dyrby, and T.P. Andriacchi, Secondary gait changes in patients with medial compartment knee osteoarthritis: increased load at the ankle, knee, and hip during walking. ARTHRITIS & RHEUMATISM, 2005. 59(9): p. 2835-44.
6. Lawrence, R.C., et al., Estimates of the Prevalence of Arthritis and Other Rheumatic Conditions in the United States. Arthritis & Rheumatism, 2008. 58(1): p. 26-35.
7. Hootman, J.M. and C.G. Helmick, Projections of US prevalence of arthritis and associated activity limitations. ARTHRITIS & RHEUMATISM, 2006. 54(1): p. 226-229.
8. Semanik, P., R.W. Chang, and D.D. Dunlop, Aerobic Activity in Prevention & Symptom Control of Osteoarthritis. PM & R: the journal of injury, function, and rehabilitation, 2012. 4(5 0): p. S37-S44.
9. Dillon, C.F., et al., Prevalence of knee osteoarthritis in the United States: arthritis data from the Third National Health and Nutrition Examination Survey 1991–1994. Journal of Rheumatology, 2006. 33(11): p. 2271-2279.
10. Cheng, Y.J., et al., Prevalence of Doctor-Diagnosed Arthritis and Arthritis-Attributable Activity Limitation — United States, 2007–2009. 2010. 59(39): p. 2238-2239.
11. Rozen, D.N. Biomechanical aspects of knee osteoarthritis and AposTherapy. Scientific Approach 2013; Available from: http://apostherapy.com/en/healthcare-professionals/treatment-packages/biomechanical-aspects.
12. Shull, P.B., et al., Muscle force modification strategies are not consistent for gait retraining to reduce the knee adduction moment in individuals with knee osteoarthritis. Journal of Biomechanics, 2015. 48: p. 3163-69.
13. Felson, D.T., et al., Osteoarthritis: new insights. Part 1: the disease and its risk factors. Ann Intern Med, 2000. 133(8): p. 635-46.
14. Felson, D.T., et al., Evidence for a Mendelian gene in a segregation analysis of generalized radiographic osteoarthritis: the Framingham Study. Arthritis & Rheumatism, 1998. 41(6): p. 1064-71.
15. Felson, D.T., The Epidemiology of Knee Osteoarthritis -Results from the Framingham Osteoarthritis Study. Semin Arthritis Rheum, 1990. 20: p. 42-50.
16. Nevitt, M.C. and D.T. Felson, Sex hormones and the risk of osteoarthritis in women: Epidemiological evidence. Ann Rheum Dis, 1996. 55: p. 673-6.
17. Srikanth, V.K., et al., A meta-analysis of sex differences prevalence, incidence and severity of osteoarthritis. Osteoarthritis and Cartilage, 2005. 13(9): p. 769-81.
18. Theis, K.A., C.G. Helmick, and J.M. Hootman, Arthritis burden and impact are greater among U.S. women than men: intervention opportunities. Journal of Womens Health, 2007. 16: p. 441-53.
19. Baker, K., et al., Quadriceps weakness and its relationship to tibiofemoral and patellofemoral knee osteoarthritis in Chinese: the Beijing osteoarthritis study. Arthritis Rheum, 2004. 50(6): p. 1815-21.
20. Slemenda, C., et al., Reduced quadriceps strength relative to body weight: a risk factor for knee osteoarthritis in women? Arthritis & Rheumatism, 1998. 41(11): p. 1951-9.
21. Slemenda, C., et al., Quadriceps weakness and osteoarthritis of the knee. Ann Intern Med, 1997. 127: p. 97-104.
22. Lohmander, L.S., et al., High prevalence of knee osteoarthritis, pain, and functional limitations in female soccer players twelve years after anterior cruciate ligament injury. Arthritis & Rheumatism, 2004. 50: p. 3145-52.
23. Felson, D.T., Occupational physical demands, knee bending, and knee osteoarthritis: results from the Framingham Study. Journal of Rheumatology, 1991. 18(10): p. 1587-92.
24. Coggon, D., et al., Occupational activities and osteoarthritis of the knee. Arthritis & Rheumatism, 2000. 43(7): p. 1449-9.
25. Focht, B.C., et al., Exercise, self-efficacy, and mobility performance in overweight and obese older adults with knee osteoarthritis. Arthritis & Rheumatism, 2005. 53: p. 659-65.
26. Messier, S.P., et al., Do older adults with knee osteoarthritis place greater loads on the knee during gait? A preliminary study. Arch Phys Med Rehabil, 2005. 86(4): p. 703-9.
27. Messier, S.P., Obesity and osteoarthritis: disease genesis and nonpharmacologic weight management. Rheum Dis Clin North Am, 2008. 34(3): p. 713-29.
28. Zhang, W., et al., OARSI recommendations for the management of hip and knee osteoarthritis, Part I: Critical appraisal of existing treatment guidelines and systematic review of current research evidence. OsteoArthritis and Cartilage, 2007. 15: p. 981-1000.
29. Zhang, W., et al., OARSI recommendations for the management of hip and knee osteoarthritis, Part II: OARSI evidence-based, expert consensus guidelines. 2008.
30. Hinman, R.S., et al., Lateral Wedges in Knee Osteoarthritis: What Are Their Immediate Clinical and Biomechanical Effects and Can These Predict a Three-Month Clinical Outcome? Arthritis & Rheumatism, 2008. 59(3): p. 408–415.
31. Shull, P.B., Silder, A., Shultz, R., Dragoo, J.L., Besier, T.F., Delp, S.L., and Cutkosky, M.R., Six-Week Gait Retraining Program reduces Knee Adduction Moment, Reduces Pain, and Improves Function for Individuals with Medial Compartment Knee Osteoarthritis. Journal of Orthopaedic Resarch, 2013. 31: p. 1020-1025.
32. Shull, P.B., Shultz, R., Silder, A., Dragoo, J.L., Besier, T.F., Cutkosky, M.R., Delp, S.L., Toe-in gait reduces the first peak knee adduction moment in patients with medial compartment knee osteoarthritis. Journal of Biomechanics, 2013. 46: p. 122-128.
33. Mangione, K.K., et al., The effects of high-intensity and low-intensity cycle ergometry in older adults with knee osteoarthritis. J Gerontol A Biol Sci Med Sci., 1999. 54(4): p. 184-90.
34. Akbarshahi, M., et al., Subject-specific evaluation of patellofemoral joint biomechanics during functional activity. Medical Engineering Physics, 2014. 36(9): p. 1122-1133.
35. Ericson, M., On the biomechanics of cycling. A study of joint and muscle load during exercise on the bicycle ergometer. Scand J Rehabil Med Suppl., 1986. 16: p. 1-43.
36. Ericson, M.O., Mechanical muscular power output and work during ergometer cycling at different work loads and speeds. European Journal of Applied Physiology, 1988. 57: p. 382-7.
37. Ericson, M.O., et al., Power output and work in different muscle groups during ergometer cycling. European Journal of Applied Physiology, 1986. 55: p. 229-235.
38. Ericson, M.O. and R. Nissell, Tibiofemoral joint forces during ergometer cycling. The American Journal of Sports Medicine, 1986. 14(4): p. 285-90.
39. Sanderson, D.J. and A. Black, The effect of prolonged cycling on pedal forces. Journal of Sports Sciences, 2003. 21(3): p. 191-99.
40. Bailey, M.P., F.J. Maillardet, and N. Messenger, Kinematics of cycling in relation to anterior knee pain and patellar tendinitis. J Sport Sci, 2003. 21: p. 649-657.
41. Too, D., Biomechanics of Cycling and Factors Affecting Performance. Sports Medicine, 2012. 10(5): p. 286-302.
42. Schipplein, O.D. and T.P. Andriacchi, Interaction between Active and Passive Knee Stabilizers during Levelwalking. J Orthopaed Res, 1991. 9: p. 113-9.
43. Miyazaki, T., et al., Dynamic load at baseline can predict radiographic disease progression in medial compartment knee osteoarthritis. Ann Rheum Dis, 2002. 61: p. 617-22.
44. Chakravarty, E.F., et al., Long distance running and knee osteoarthritis – A prospective study. Am J Prev Med, 2008. 35: p. 133-8.
45. Ahlbäck, S., Osteoarthrosis of the knee. A radiographic investigation. Acta Radioligica Diagnosis Supplement, 1968. 277.
46. Cerejo, R., et al., The influence of alignment on risk of knee osteoarthritis progression according to baseline stage of disease. Arthritis & Rheumatism, 2002. 46: p. 2332-36.
47. Hurley, M.V., THE ROLE OF MUSCLE WEAKNESS IN THE PATHOGENESIS OF OSTEOARTHRITIS. Rheumatic Disease Clinics in north America, 2005. 25(2): p. 283-298.
48. Hurley, M.V., Muscle Dysfunction and Effective Rehabilitation of Knee Osteoarthritis: What We Know and What We Need To Find Out. Arthritis & Rheumatism, 2003. 49(3): p. 444-52.
49. Walter, J.P., Korkmaz, N, Fregly, B.J., Pandy, M.G., Contribution of Tibiofemoral Joint Contact to Net Loads at the Knee in Gait. Journal of Orthopaedic Resarch, 2015. 33(7): p. 1054-60.
50. Baliunas, A.J., Hurwitz, D.E., Ryals, A.B., Karrar, A., Case, J.P., Block, J.A., Andriacchi, T.P., Increased knee joint loads during walking are present in subjects with knee osteoarthritis. Osteoarthritis Cartilage, 2002. 10(7): p. 573-9.
51. Gardner, J.K., et al., Acute effects of lateral shoe wedges on joint biomechanics of patients with medial compartment knee osteoarthritis during stationary cycling. Journal of Biomechanics, 2016. 49: p. 2817-23.
52. Gardner, J.K., et al., Effects of Toe-in Angles on Knee Biomechanics in Cycling of Patients with Medial Knee Osteoarthritis. Clin Biomechanics, 2015. 30: p. 276–282.
53. Kautz, S.A., M.L. Hull, and R.R. Neptune, A Comparison of Muscular Mechanical Energy-Expenditure and Internal Work in Cycling. Journal of Biomechanics, 1994. 27(12): p. 1459-1467.
54. MacIntosh, B.R., R.R. Neptune, and J.F. Horton, Cadence, power, and muscle activation in cycle ergometry. Medicine and Science in Sports and Exercise, 2000. 32(7): p. 1281-1287.
55. Anderson, F.C. and M.G. Pandy, Dynamic optimization of human walking. J Biomech Eng, 2001. 123(5): p. 381-90.
56. Anderson, F.C. and M.G. Pandy, Static and dynamic optimization solutions for gait are practically equivalent. Journal of Biomechanics, 2001. 34(2): p. 153-61.
57. Delp, S.L., et al., OpenSim: open-source software to create and analyze dynamic simulations of movement. IEEE Trans Biomed Eng, 2007. 54(11): p. 1940-50.
58. Thelen, D.G., Adjustment of muscle mechanics model parameters to simulate dynamic contractions in older adults. J Biomech Eng, 2003. 125(1): p. 70-7.
59. Thelen, D.G. and F.C. Anderson, Using computed muscle control to generate forward dynamic simulations of human walking from experimental data. Journal of Biomechanics, 2006. 39(6): p. 1107-15.
60. Thelen, D.G., F.C. Anderson, and S.L. Delp, Generating dynamic simulations of movement using computed muscle control. Journal of Biomechanics, 2003. 36(3): p. 321-8.
61. Hamner, S.R., A. Seth, and S.L. Delp, Muscle contributions to propulsion and support during running. Journal of Biomechanics, 2010. 43(14): p. 2709-16.
62. Fregly, B.J., et al., Grand challenge competition to predict in vivo knee loads. J Orthop Res, 2012. 30(4): p. 503-13.
63. Morrison, J.B., The mechanics of the knee joint in relation to normal walking. Journal of Biomechanics, 1970. 3: p. 51-61.
64. Bei, Y. and B.J. Fregly, Multibody dynamic simulation of knee contact mechanics. Medical Engineering & Physics, 2004. 26(9): p. 777-89.
65. Lin, Y.C., et al., Simultaneous prediction of muscle and contact forces in the knee during gait. Journal of Biomechanics, 2010. 43(5): p. 945-52.
66. Hast, M.W. and S.J. Piazza, Dual-joint modeling for estimation of total knee replacement contact forces during locomotion. Journal of Biomechanical Engineering, 2013. 135(2): p. 021013.
67. Thelen, D.G., K.W. Choi, and A.M. Schmitz, Co-simulation of neuromuscular dynamics and knee mechanics during human walking. Journal of Biomechanical Engineering, 2014. 136(2): p. 021033.
68. Yang, N.H., et al., Effect of frontal plane tibiofemoral angle on the stress and strain at the knee cartilage during the stance phase of gait. Journal of Orthopaedic Research, 2010. 28(12).
69. Gerus, P., et al., Subject-specific knee joint geometry improves predictions of medial tibiofemoral contact forces. Journal of Biomechanics, 2013. 46: p. 2778-2786.
70. Hsu, R.W., et al., Normal axial alignment of the lower extremity and load-bearing distribution at the knee. Clinical Orthopaedics and Related Research, 1990. 225: p. 215-227.
71. Hurwitz, D.E., et al., The knee adduction moment during gait in subjects with knee osteoarthritis is more closely correlated with static alignment than radiographic disease severity, toe out angle and pain. Journal of Orthopaedic Research, 2002. 20(1): p. 101-107.
72. Halder, A., et al., Influence of limb alignment on mediolateral loading in total knee replacement: in vivo measurements in five patients. Journal of Bone and Joint Surgery: American Volume, 2012. 94: p. 1023-29.
73. Moreland, J.R., L.W. Bassett, and G.J. Hanker, Radiographic analysis of the axial alignment of the lower extremity. Journal of Bone and Joint Surgery: American Volume, 1987. 69(5): p. 745-9.
74. Winby, C.G., et al., Muscle and external load contribution to knee joint contact loads during normal gait. Journal of Biomechanics, 2009. 42(14): p. 2294-2300.
75. Kumar, D., K.S. Rudolph, and K.T. Manal, EMG-driven modeling approach to muscle force and joint load estimations: Case study in knee osteoarthritis. Journal of Orthopaedic Research, 2011. 30(3): p. 377-83.
76. DeMers, M.S., S. Pal, and S.L. Delp, Changes in Tibiofemoral Forces due to Variations in Muscle Activity during Walking. Ortho Research Society, 2014: p. 1-8.
77. Bergmann, G., et al., Hip contact forces and gait patterns from routine activities. J Biomech, 2001. 34(7): p. 859-71.
78. Richards, C. and J.S. Higginson, Knee contact force in subjects with symmetrical OA grades: differences between OA severities. J Biomech, 2010. 43(13): p. 2595-600.
79. Peat, G., R. McCarney, and P. Croft, Knee pain and osteoarthritis in older adults: a review of community burden and current use of primary health care. Ann Rheum Dis, 2001. 60: p. 91-7.
80. Aglietti, P. and P.P. Menchetti, Distal femoral varus osteotomy in the valgus osteoarthritic knee. American Journal of Knee Surgery, 2000. 13(2): p. 89-95.
81. Coventry, M.B., D.M. Ilstrup, and S.L. Wallrichs, Proximal tibial osteotomy. A critical long-term study of eighty-seven cases. Journal of Bone and Joint Surgery: American Volume, 1993. 75(2): p. 196-201.
82. Edgerton, B.C., E.M. Mariani, and B.F. Morrey, Distal femoral varus osteotomy for painful genu valgum. A five-to-11 year follow-up study. Clinical Orthopaedics and Related Research, 1993. 288(263-9).
83. Naudie, D., et al., The Insall award. Survivorship of the high tibial valgus osteotomy. A 10- to 22-year follow-up study. Clinical Orthopaedics and Related Research, 1999. 367: p. 18-27.
84. Losina, E., et al., Cost-effectiveness of Total Knee Arthroplasty in the United States: Patient Risk and Hospital Volume. Archives Internal Medicine, 2009. 169(12): p. 1113-22.
85. Bachmeier, C.J., et al., A comparison of outcomes in osteoarthritis patients undergoing total hip and knee replacement surgery. Osteoarthritis and Cartilage, 2001. 9: p. 137-146.
86. Roddy, E., W. Zhang, and M. Doherty, Is gout associated with reduced quality of life? A case-control study. Rheumatology (Oxford), 2007. 46(9): p. 1441-1444.
87. Hurley, M.V. and N.E. Walsh, Effectiveness and clinical applicability of integrated rehabilitation programs for knee osteoarthritis. Current Opinion in Rheumatology, 2009. 21: p. 171-6.
88. Bakker, M.F., et al., Tight control in treatment of rheumatoid arthritis efficacy and feasibility. Ann. Rheum. Dis., 2007. 67: p. 190-195.
89. Fregly, B.J., J.A. Reinbolt, and T.L. Chmielewski, Evaluation of a patient-specific cost function to predict the influence of foot path on the knee adduction torque during gait. Computer Methods in Biomechanics and Biomedical Engineering, 2008. 11(1): p. 63-71.
90. Fregly, B.J., et al., Design of patient-specific gait modifications for knee osteoarthritis rehabilitation. Ieee Transactions on Biomedical Engineering, 2007. 54(9): p. 1687-1695.
91. Rajagopal, A., et al., Full-Body Musculoskeletal Model for Muscle-Driven Simulation of Human Gait. IEEE TRANSACTIONS ON BIOMEDICAL ENGINEERING,, 2016. 63(10).
92. Millard, M., et al., Flexing computational muscle: modeling and simulation of musculotendon dynamics. J Biomech Eng, 2013. 135(2): p. 021005.
93. Winters, J.M., An improved muscle-reflex actuator for use in large-scale neuromusculoskeletal models. Annals of Biomedical Engineering, 1995. 23(4): p. 359-74.
94. Zajac, F.E., Muscle and tendon: properties, models, scaling, and application to biomechanics and motor control. Crit Rev Biomed Eng, 1989. 17(4): p. 359-411.
95. Anderson, F.C. and M.G. Pandy, A Dynamic Optimization Solution for Vertical Jumping in Three Dimensions. Comput Methods Biomech Biomed Engin, 1999. 2(3): p. 201-231.
96. Delp, S.L., et al., An interactive graphics-based model of the lower extremity to study orthopaedic surgical procedures. IEEE Trans Biomed Eng, 1990. 37(8): p. 757-67.
97. Hoy, M.G., F.E. Zajac, and M.E. Gordon, A musculoskeletal model of the human lower extremity: the effect of muscle, tendon, and moment arm on the moment-angle relationship of musculotendon actuators at the hip, knee, and ankle. Journal of Biomechanics, 1990. 23(2): p. 157-69.
98. Neptune, R.R., S.A. Kautz, and M.L. Hull, The effect of pedaling rate on coordination in cycling. Journal of Biomechanics, 1997. 30(10): p. 1051-1058.
99. Hicks, J.L., et al., Is My Model Good Enough? Best Practices for Verification and Validation of Musculoskeletal Models and Simulations of Movement. Journal of Biomechanical Engineering, 2015. 137(2): p. 1-24.
100. Neptune, R.R. and M.L. Hull, Evaluation of performance criteria for simulation of submaximal steady-state cycling using a forward dynamic model. Journal of Biomechanical Engineering-Transactions of the Asme, 1998. 120(3): p. 334-341.
101. Smak, W., R.R. Neptune, and M.L. Hull, The influence of pedaling rate on bilateral asymmetry in cycling. Journal of Biomechanics, 1999. 32(9): p. 899-906.
102. Mornieux, G., et al., Influence of cadence, power output and hypoxia on the joint moment distribution during cycling. Eur J Appl Physiol, 2007. 102: p. 11-18.
103. Neptune, R.R. and M.L. Hull, A theoretical analysis of preferred pedaling rate selection in endurance cycling. Journal of Biomechanics, 1999. 32(4): p. 409-415.
104. Neptune, R.R. and M.L. Hull, Methods for determining hip movement in seated cycling and their effect on kinematics and kinetics. Journal of Applied Biomechanics, 1996. 12(4): p. 493-507.
105. Leddy, J.J., et al., Effects of Exercise Rehabilitation on Muscle Function, Activity and Joint Spaces in Patients with Osteoarthritis. Med Sci Sports Exer, 1994. 26(Suppl): p. S142.
106. Kutzner, I., et al., Loading of the Knee Joint During Ergometer Cycling: Telemetric In Vivo Data. Journal of Orthopaedic & Sports Physical Therapy, 2012. 42(12): p. 1032-38.
107. Walter, J.P., et al., Decreased knee adduction moment does not guarantee decreased medial contact force during gait. J Orthop Res, 2010. 28(10): p. 1348-54.
108. Seth, A., et al., OpenSim: a musculoskeletal modeling and simulation framework for in silico investigations and exchange. Iutam Symposium on Human Body Dynamics, 2011. 2: p. 212-232.
109. Neptune, R.R. and S.A. Kautz, Knee joint loading in forward versus backward pedaling: implications for rehabilitation strategies. Clin Biomech (Bristol, Avon), 2000. 15(7): p. 528-35.
110. Kutzner, I., et al., Loading of the knee joint during activities of daily living measured in vivo in five subjects. J Biomechanics, 2010. 43: p. 2164–2173.
111. Carter, D.R. and M. Wong, The role of mechanical loading histories in the development of diarthrodial joints. J Orthop Res, 1988. 6(6): p. 804-16.
112. Baliunas, A.J., et al., Increased knee joint loads during walking are present in subjects with knee osteoarthritis. Osteoarthritis and Cartilage, 2002. 10(7): p. 573-579.
113. Sharma, L., et al., Knee adduction moment, serum hyaluronan level, and disease severity in medial tibiofemoral osteoarthritis. Arthritis & Rheumatism, 1998. 41(7): p. 1233-40.
114. Schnitzer, T.J., et al., Effect of piroxicam on gait in patients with osteoarthritis of the knee. Arthritis & Rheumatism, 1993. 36(9): p. 1207-1213.
115. Sasaki, K. and R.R. Neptune, Individual muscle contributions to the axial knee joint contact force during normal walking. Journal of Biomechanics, 2010. 43(14): p. 2780-4.
116. Shelburne, K.B., M.R. Torry, and M.G. Pandy, Contributions of muscles, ligaments, and the ground-reaction force to tibiofemoral joint loading during normal gait. J Orthop Res, 2006. 24(10): p. 1983-90.
117. Steele, K.M., et al., Compressive tibiofemoral force during crouch gait. Gait & Posture, 2012. 35(4): p. 556-60.
118. Sritharan, P., Y.C. Lin, and M.G. Pandy, Muscles that do not cross the knee contribute to the knee adduction moment and tibiofemoral compartment loading during gait. Journal of Orthopaedic Research, 2012. 30(10): p. 1586-95.
119. Glitsch, U. and W. Baumann, The three-dimensional determination of internal loads in the lower extremity. Journal of Biomechanics, 1997. 30(11-12): p. 1123-1131.
120. Shelburne, K.B., M.R. Torry, and M.G. Pandy, Muscle, ligament, and joint-contact forces at the knee during walking. Med Sci Sports Exerc, 2005. 37(11): p. 1948-56.
Appendix
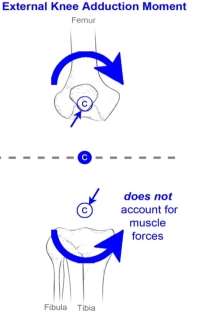
Figure 1: Illustrations showing the direction of the net external knee adduction moment (KAM) represented by the blue curved arrows toward the midline or medial aspect of the body or corresponding knee joint this is a result from a traditional inverse dynamics analysis determining the net forces responsible for the movement. The important fact in the difference between the adduction-abduction contact moments for this research is the net KAM is determined without taking into account for the internal muscle forces.
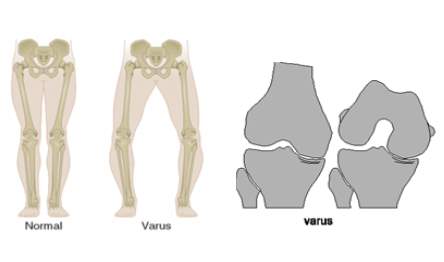
Anterior View
Posterior View
Figure 2: Showing what the a) lower limb looks like when there is a change in alignments, b) shows a closer look as to what happens at the knee joint when the joint changes from normal alignment to a more varus or adducted alignment.
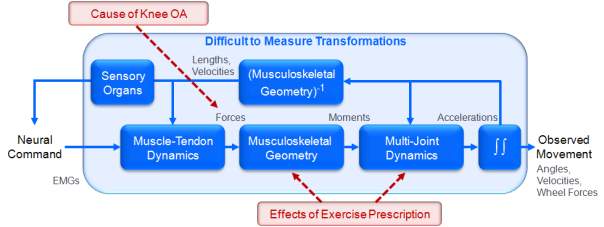
Figure 3: Many factors contribute to knee joint contact loads associated with OA disease progression and disability during movements. The transformations between experimental EMG patterns and coordinated multi-joint movement (shaded region) are complicated. Furthermore, to make exercise prescription decisions, clinicians must predict joint load changes after equipment adjustments. Exercise prescription alters musculoskeletal geometry and multi-joint dynamics and these changes are not easily measured. The long-term goal of our work is to provide a scientific basis for exercise prescription that reduces harmful knee joint contact loads.
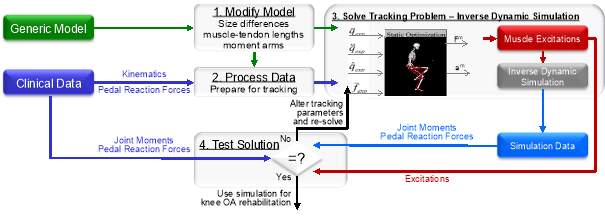
Figure 4: Four-step procedure to generate a subject-specific inverse dynamic simulation that reproduces experimental data.
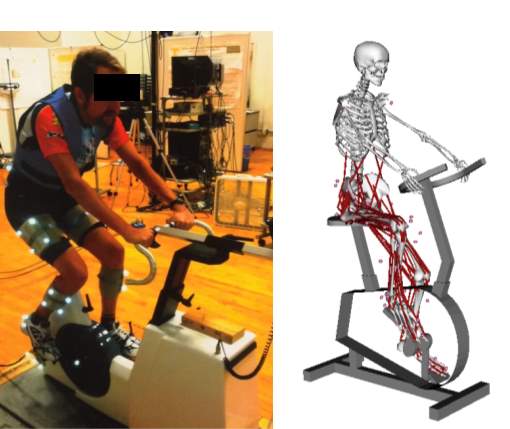
Figure 5: Example subject during a) data collection and example b) subject-specific musculoskeletal model for muscle force estimation with the added patella.
Table 1: A muscle force change identity grid showing the significant differences within each cohort in all lower extremity muscles compared to neutral pedal condition, for the mean muscle forces. Red indicates an increase, while blue indicates a decrease compared to neutral. The darker the color the greater the increase or decrease.
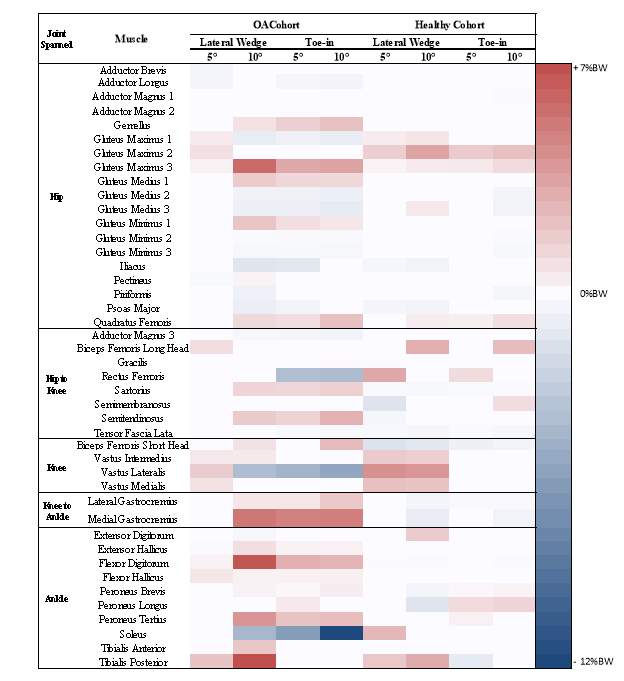
Figure 6: Some of the muscles that showed the most change across the conditions (at least 3 conditions compared to neutral) with a threshold set at greater than 10%BW, observed for a) mean and b) peak muscle forces normalized by %BW.
Table 2: A muscle force change identity grid showing the significant differences within each cohort in all muscles compared to neutral, for the peak muscle forces. Red indicates an increase, while blue indicates a decrease compared to neutral; while, the darker the color the greater the increase or decrease.
Table 3: Mean and peak muscle force significant differences between OA (+) and Healthy (-) in all conditions. Red indicates knee OA cohort was greater within that conditions, while blue indicates healthy cohort was higher within that conditions; the darker the color the greater the difference.
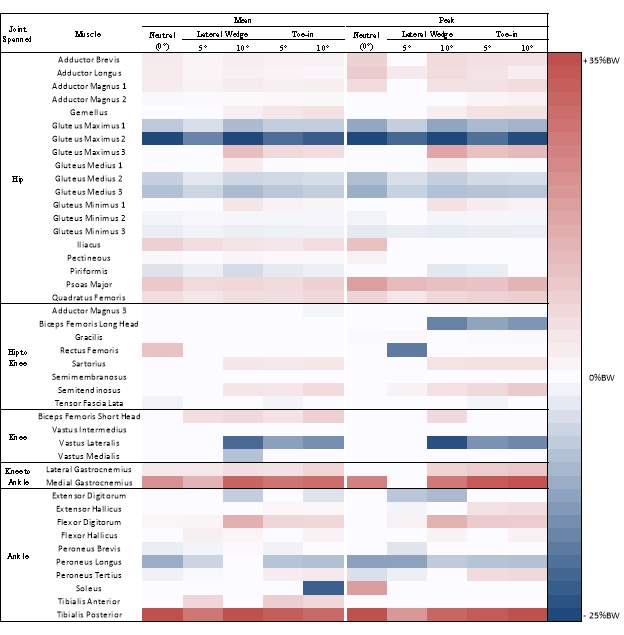
Figure 7: Comparison of muscle activations during cycling with the neutral pedal condition (at 80W and 60 RPM) and EMG from Neptune, et al., 1997 from elite male cyclists (at 127W and 60 RPM).
Table 4: The joint contact loads change identity grid showing the significant differences within each cohort in all contact loads compared to neutral, for the peak contact loads. Red indicates an increase compared to neutral, while blue indicates a decrease compared to neutral; while, the darker the color the greater the increase or decrease.
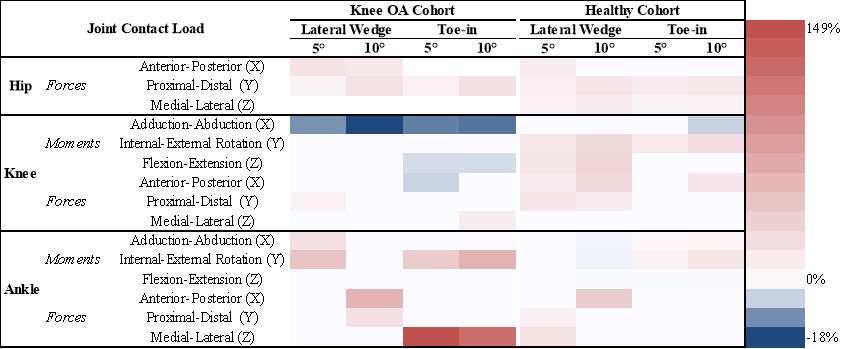
Table 5: The peak joint contact loads significant differences between OA (+) and Healthy (-) across all conditions. Red indicates knee OA cohort was greater within the corresponding condition, while blue indicates healthy cohort was higher within that condition. The darker the color the greater the difference.
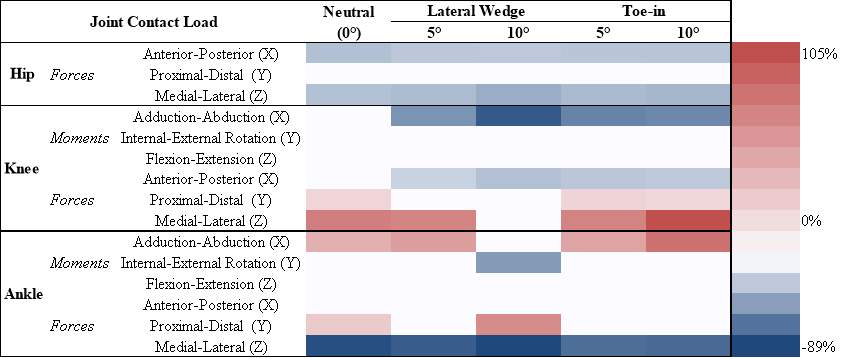
Table 6: A change identity grid showing the differences in the joint contact loads for the specific muscles that changed from the normal joint reaction analysis. Results are shown for each cohort for step 1’s muscle variation, for the subjects with and without knee OA in all muscles used stands for a muscle weighting on 1, while don’t stands for. Red indicates an increase compared to joint reaction analysis, while blue indicates a decrease compared to joint reaction analysis. The darker the color the greater the increase or decrease.
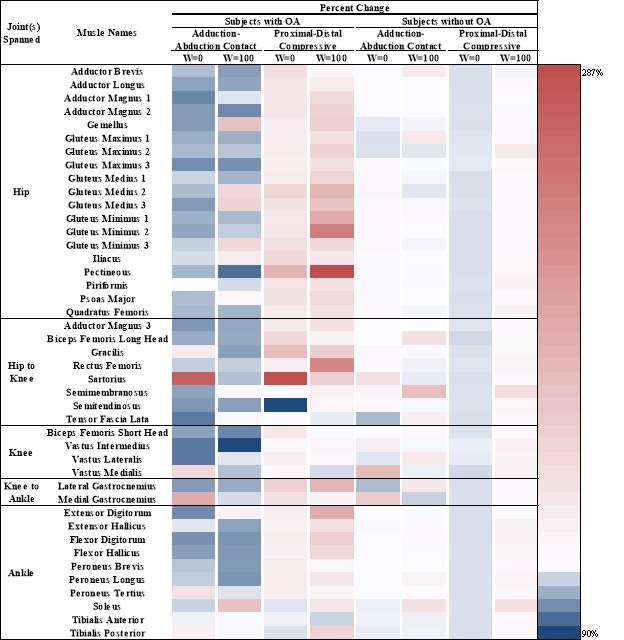
Figure 8: An example of a few muscle activations for a) subjects with OA and b) Subjects without OA. This tested the sensitivity of the joint contact loads by altering the muscle activations weightings from normal static optimization (weighting of 1) shown in blue. The red line is a weighting of 0 indicating being able to use the muscle as much as it wants to carry out the cycling movement. The green line is with a weighting of 100 penalizing the muscle with a and not allowing it to be used. Lastly, the black line is for maximizing the muscle activations.
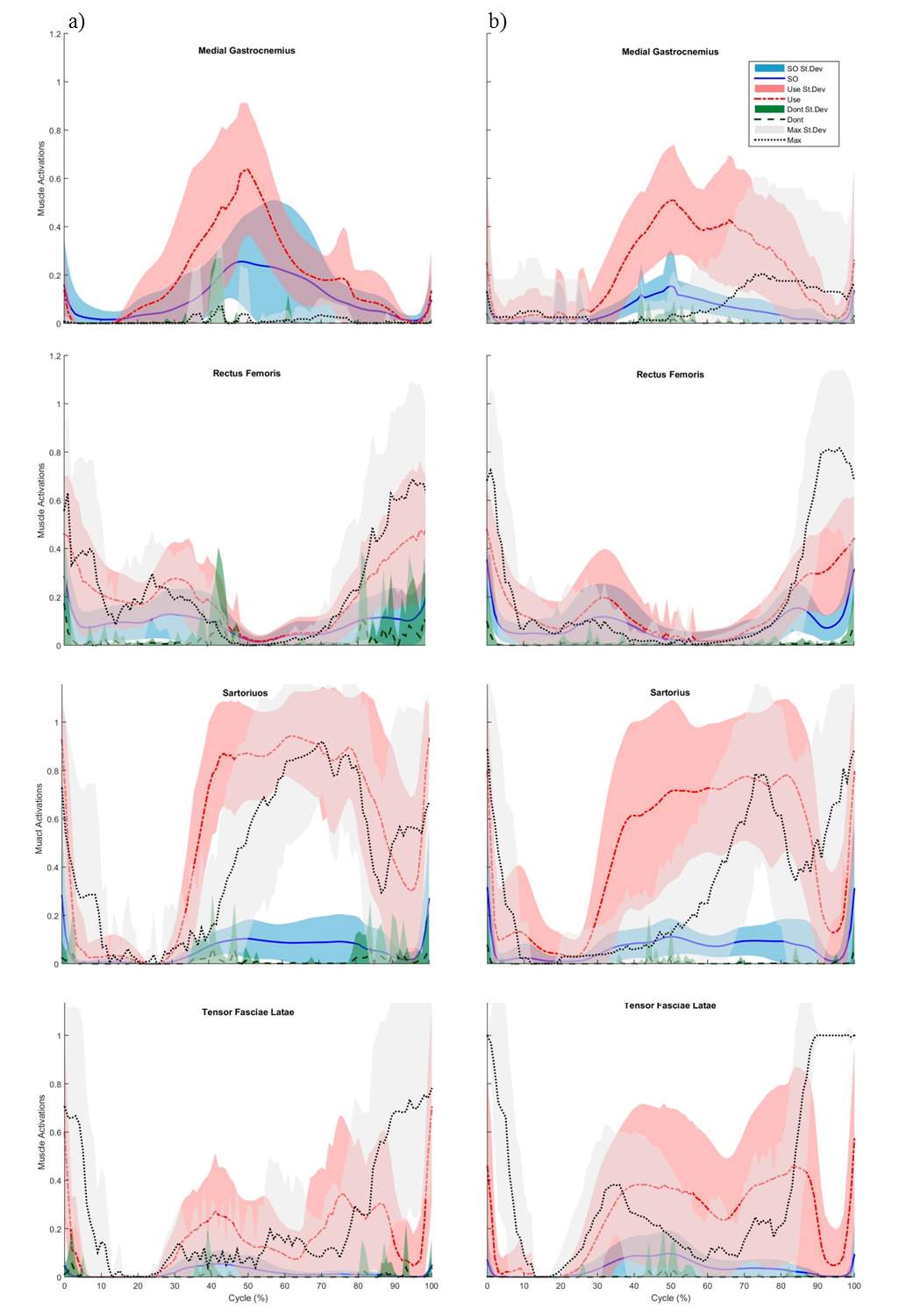
Figure 8 Continued
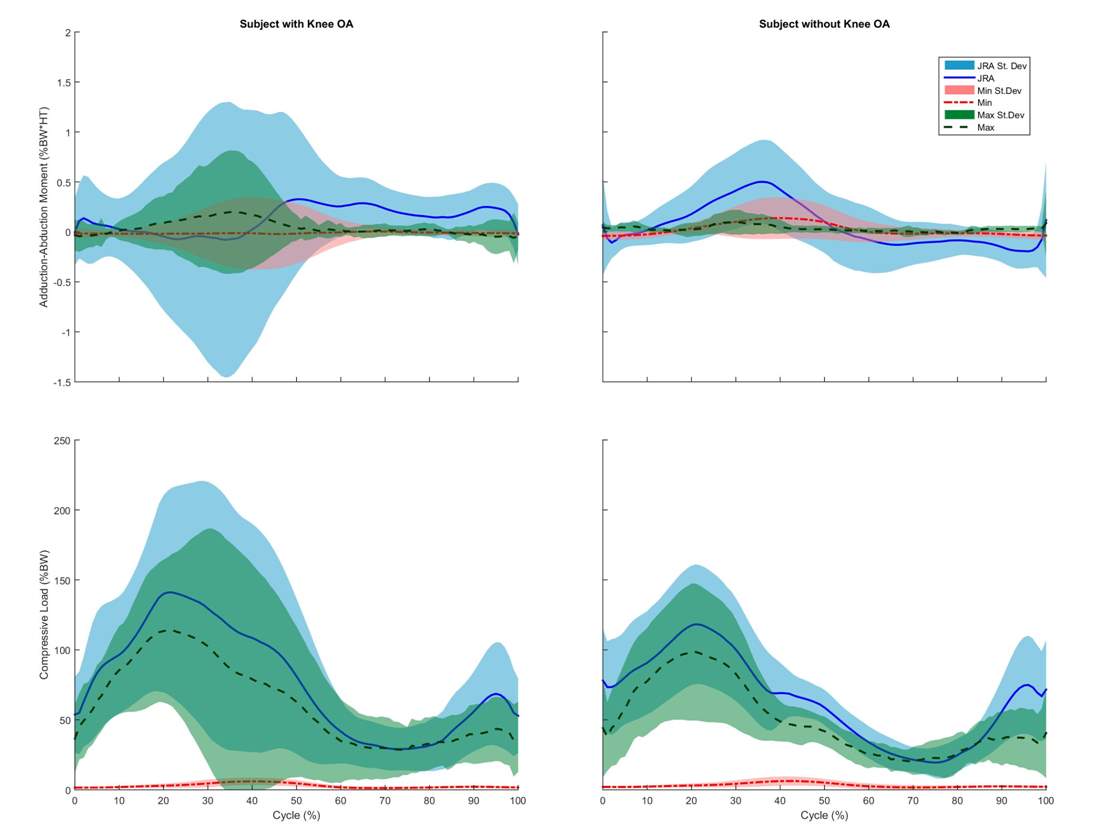
Figure 9: Subjects with (left) and without (right) knee OA’s optimized adduction-abduction moment (top) and the compressive load(bottom) results shown, from the minimized knee joint contact loads and the maximized muscle forces in order to minimize joint contact loads. Minimum knee contact loads shown in red and maximized muscle forces shown in green were plotted against previously calculated joint contact loads from joint reaction analysis shown in blue.
Vita
Rachel L. Thompson was born in Rochester, NY, to the parents of Ann and Brian Thompson. She is the third child of three: Matthew Thompson and Nicole Ray. She grew up and attended elementary through high school in Rochester, NY. After her graduation, Rachel went on to receive her Bachelors of Science degree in 2012 from Slippery Rock University in Health and Environmental Science with a minor in Adapted Physical Activity. After Graduation, Rachel went on to an internship at the Canandaigua Veteran Affairs (VA) Medical Center, where she was introduced to the field of Biomedical Engineering. Then she went on to work for a year at the University of Rochester Medical Center in the operating room, as a project assistant and then ultimately a cardiac and vascular implant coordinator. Where her job was to order and receive medical implants for specific surgeries from here, her passion for implants, prosthetics and medical devices grew and she ultimately decided to pursue an advanced degree in Biomedical Engineering. In her quest for knowledge and desire to learn in this field of Biomedical Engineering, she attended the University of Tennessee where she received her Master’s degree in 2016. Finally, Rachel went on to pursue and ultimately complete a PhD in Biomedical Engineering at the University of Tennessee. She graduated in May 2017. She is planning on working in the medical research field.
Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allRelated Content
All TagsContent relating to: "Sports"
Sports are a combination of skill and physical activity, and can be done as either an individual or as part of a team. Sports can help you to keep fit and provide you or others with entertainment.
Related Articles
DMCA / Removal Request
If you are the original writer of this dissertation and no longer wish to have your work published on the UKDiss.com website then please: