Changes for Implementation of a Closed Loop Infusion Therapy System into an Existing Working Paediatric Hospital
Info: 10785 words (43 pages) Dissertation
Published: 22nd Feb 2022
Introduction
Hospitals are increasingly combining medical devices and ICT into complex Medical IT Systems. The intent is to manage patient data better and to configure devices into systems that as a whole provide more functionality and safety than the individual equipment can do when used as standalone devices. Medication error in hospitals is a significant problem worldwide. Hospitals are developing closed loop medication systems in an attempt to reduce this problem. A complete closed loop infusion medication system (CCLIMS) brings together technical medical equipment previously used in isolation and includes Infusion Pumps, Drug Libraries, Drug Storage, Logistics and monitoring systems and Electronic Health Care Records (EHR). Such systems support the use of engineering controls and decision support software which adds a layer of safety (Miller et al., 2017, Ohashi et al., 2014).
The current development of the New Children’s Hospital (NCH) on the St James Campus in Dublin’s city centre where the design brief to open this 400-bed world class hospital as a Healthcare Information and Management Systems Society (HIMSS Level 7) establishment. (The grading of a hospital in relation to the electronic healthcare record is gauged in the HIMSS scale, level 7 is the highest award possible). To open a large complex ‘state of the art’ paediatric Hospital at level 7 will require a detailed level of planning that will be a greater challenge than the architectural plans for the buildings themselves. The vast amount of integrated services needed to create this digital environment will only be successful if the right approach to their implementation is followed. The NCH is being designed to ‘be born’ as a digital hospital (see figure 1-1), to support the ‘hub & spoke’ delivery approach towards technology will be at the forefront of the venture. This technology will allow seamless efficiency when sharing patient information to ensure patient safety throughout the care model.

Figure 1‑1:- ‘Born’ Digital (eHealth Ireland, 2018)
If the NCH is to be ‘born’ digital, the HSE need to take notice of the lessons learnt across the globe and within Ireland. This will ensure its success as the governance of such a change is critical and is a true opportunity for the HSE to make an impact with the exciting innovation of a truly paperless/digital new children’s hospital for the children of Ireland (eHealth Ireland, 2018).
1.3 Motivation
The researcher is employed as a clinical engineer in a large Dublin Paediatric hospital since 2000 (OLCHC). The advances in Infusion pump technology especially toward accuracy and safety has spiralled over the last two decades. A complete closed loop infusion medication system (CCLIMS) is the end of this journey for medication errors. If correctly implemented with the correct interoperability between the different systems involved the goal of safe, error free infusion pump administrated medication is here.
These commercially available Closed Loop Systems (CLS) will utilise Auto-Programming (APG), Smart infusion Pumps (SIPs), Drug Error Reduction Software (DERS), Computerised Physician Order Entry (CPOE), Barcode Medication Administration (BCMA) with an Electronic Healthcare Record (EHR) which throughout all the different departments which will make the process possible. This study will look at both the system’s technology and infrastructure requirements (within the existing PICU environment in OLCHC), how the introduction and merging of SIP infusions and medical charting system functions, simplify or complicate the working practices of the staff who make up and administer prescribed drug infusions to patients.
1.4 Study Domain
OLCHC is a national 243 bed paediatric children’s hospital solely for the care of children in Ireland since 1956. OLCHC employs nearly 1200 staff and has two paediatric intensive care units (PICU 1 & PICU 2). PICU 1 is a 17-bed state of the art modular build facility on the 1st floor and while PICU 2 is an 8-bed unit with 6 open bed bays and 2 isolation room which resides within the original 1956 hospital footprint. PICU 1 has been re-developed and updated many times for the needs of the patients over the last couple of decades but carries an ageing ICT infrastructure which can limit technological advancements when trying to improve the communication and interoperability of any new system such as the one considered in this work.
Presently, both PICUs in OLCHC use the Phillips IntelliSpace Critical Care and anaesthesia (ICCA) electronic patient record (EPR) system which includes an e-prescribing module. In OLCHC, within the PICUs, smart infusion pump technology (SIPs) are already in use with advanced and internally developed Drug Error Reduction Software (DERS) (i.e. Paediatric/Neonatal drug library). This comprehensive drug library is already shared with Temple Street’s Children’s Hospital and is to be rolled out to the 19 maternity hospitals as part of a national initiative. Standardising practices across sites is laying important preparatory work prior to the opening of the NCH. (Howlett, 2018).
1.5 Research Question
After discovering where to address the focus of this research within the hospital and coupled with the motivation of reducing medication errors to children by using the combination of available technologies which combined together make a CCLIMS, the research question is;
What are the changes to work practices through the proposed implementation of a closed loop infusion therapy system into an existing working paediatric hospital?
1.6 Study Aims and Objectives
The aim of this study is to ascertain what are the changes needed to work practices in order to effectively integrate a complete closed loop medication system into an existing paediatric intensive care.
The information gathered will be invaluable when the integration of a complete closed loop infusion medication system (CCLIMS) is to be installed into a existing hospital or quite possibly the construction NCH when it is delivered for the children of Ireland in 2022.
1.7 Outline of the Research
This research set out to identify what are the changes to working practices when a CCLIMS is introduced into a functioning well-advanced highly technical environment such as a working paediatric ICU. This research should clearly highlight the barriers, limitations and advantages to the acquirement and implementation of a CCLIMS. Medication safety is first and foremost, one target for this research is to highlight how a well-integrated system will benefit the sick children of Ireland delivering safer drug infusions within the PICU in either OLCHC or the NCH. The researcher wants to establish the shortcomings if the socio-technical part of the integration process of a multi departmental system is not approached correctly, or the changes to working practices aren’t thought-out entirely. If these shortcomings are not addressed in a rigorous manner this complicated CCLIMS which is proposed for the NCH won’t be able to be implemented fully, this will unfortunately leave the most vulnerable of patients, children at risk from medication error.
A literature review was conducted which highlighted the difficulties faced by other hospitals around the globe and the solutions that they adopted. Hospitals in the United States have been developing closed loop systems for many years and they are ahead in this field of technology to-date. Functioning systems in the United States include modules for billing the patient or an insurance company at the end of the hospital stay (automatic billing of the infused medications used etc.), (interestingly the hospitals that have developed these systems are mostly privately-run institutions). The literature is narrow regarding implementing a CCLIMS into a children’s hospital ICU (the most noteworthy referenced papers have a strong linkage to medical device manufacturers who has sponsored the publication, the conflict of interest is disclosed and the content is proven to be sound (in relation to what is known about the subject of CCLIMS). To date it is believed a CCLIMS has not been implemented in Ireland or the UK. The difficultly with a children’s hospital is the weight range of the patients (ie from 500g to 100kg) using standard concentrations of drugs throughout the needed weight bands becomes a complex task (Irwin et al., 2008).
After interviewing the experts from all the departments which need to be involved when implementing a CCLIMS, comparisons of work and information flows and practices were developed on activity UML diagrams. These proposed, and now discussed system changes to the CCLIMS work practices (and information flows) as outputs of this work will be used to guide the organisation in culminating suggestions to be considered before a CCLIMS is tendered for and implemented into our national children’s hospital. Figure 2 shows a flow diagram of the process undergone to complete this dissertation.
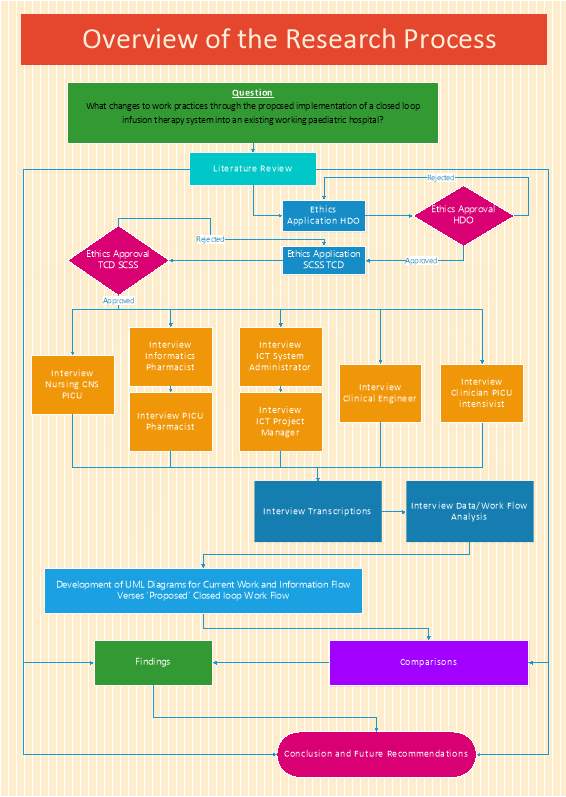
Figure 1‑2:- Overview of the Research Process
1.8 Overview of the Dissertation
This chapter has presented the motivation for the research, the study domain, the research question, objectives and an outline of the research.
Chapter 2: – Provides the literature review. It first addresses infusion devices and the recent technological advancements with smart infusion pumps (SIP) and software based safety systems associated with them, re: – Drug Error Reduction Software (DERS). Secondly the literature review explores the use of these smart infusion pumps (SIP) combined with Electronic Health/Medical Records systems (EHR, EMR) to form a combined interoperable system to aid medical staff and allied healthcare professionals.
Chapter 3: – Presents the design of the research study and outlines the methodologies used and the rational for choosing the qualitative method of purposeful specialist sampling based on the expert’s knowledge from each specialty field/department and any ethical considerations.
Chapter 4: – Presents the detailed findings of the study, identifying and quantifying the differences and changes to the work and information flow which this new technology introduces and what effects these have on the working practices of the doctors, nurses, pharmacists, ICT and Clinical Engineering Professionals in the proposed system studied.
Chapter 5: – Discusses the findings of the comparisons in the different working practices. How these findings address the research question, the significance of the findings, and the impact of the changes in work practices from one discipline and how it effects the other departments work and information flow. This chapter also addresses the limitations of the study.
Chapter 6: – Concludes the dissertation, makes recommendations for paediatric hospitals planning to implement a complete closed loop medication system and identifies possible future research work in this area.
Literature Review – State-of-the-art
2.1 Introduction
Before proceeding to describe the research study in full detail, it is necessary to describe the state of the art in this field. Medication safety is a widely discussed topic in medicine and many studies prove that a carefully designed support system to address difficulties in medication compounding add extra value to the essential safety net. The most vulnerable patients to a medication error are the patients in a paediatric intensive care unit. In the landmark study ‘to err is human’ this book states frank truth about errors in the health system and how medicine is not as safe as it should be (Linda T. Kohn, 2000). ‘Death by decimal’ is a article calling for a more aggressive approach to stopping another preventable death of a paediatric patient from occurring (Broselow and Schuman, 2008).This chapter provides the literature review. It first addresses the current, published scientific evidence of the development of infusion devices including the recent technological advancements of these devices into smart infusion pumps (SIPs) and the software based safety systems associated with them, re: – Drug Error Reduction Software (DERS). Secondly the literature review explores the use of these smart infusion pumps (SIPs) combined with Electronic Health/Medical Records systems (EHR, EMR) to form a combined interoperable system to aid, guide and offer assistance to the medical staff and the allied healthcare professionals involved in the delivery of IV infusions. The review will also explore the socio-technical aspect of a complicated hospital wide implementation of a CCLIMS system (or similar equivalent).
2.2 Background
Many hospitals who aspire to implement a closed loop medication system view it as the introduction of a technical system and either don’t understand the need for a corresponding change in the socio systems that support drug delivery, or underestimate the level of investment needed in the socio side of the socio-technical system.
The medical system suppliers make it sound so straightforward to implement their systems into hospitals. If these systems are carelessly designed, integrated or poorly
resourced they’ll impact on working practices which can decrease the benefits to patients (Burgin et al., 2014). In reality, there are many failed projects in the domain of health Informatics, this can be due to the complex interdisciplinary tasks needing to be performed. Multidisciplinary teams need to assess various different integrations across many departments in hospitals (Samaranayake et al., 2014).
For example, a complete closed loop paediatric medication system will involve at least five different departments having to work to a common goal. The professions involved will be from Pharmacy, Medical, Nursing, Clinical Engineering and Information Technology (ICT). These disciplines will have to input into the closed loop medication system (and change their working practices) to ensure it can be integrated into a Hospitals existing infrastructure. The Medical vendors also have a difficult role to action as they need to progress all integrations and ensure the interoperability with the other vendors systems (i.e. EMR/EHR) until the customer / Health-Care Delivery Organisations (HDO) is satisfied. A phased team approach with a large training regime will be needed to get a CCLIMS operational (Sims and Schneider, 2012).
2.3 Search Strategy
An extensive, systematic search was conducted using subject matter from several scientific databases, namely TCD Library Stellar Search, Scopus, Pubmed, Science Direct, Google Scholar (these are shown in the table below). When in the first phase of searching, a suitable article was found (full text version wasn’t available) either from a subscription requirement or a restricted journal article, the researcher found that a second input into the TCD stellar search would locate a full text PDF version within the TCD library (including TCD’s external library alliances sourcing the required article/paper/journal access with an approximate 85% success rate) so the article could be fully accessed and read for appropriateness for inclusion or exclusion.
Table 2‑1:- Electronic Database Information Search Details
| Electronic Database Information Search Details | |
| Databases | TCD Library Stellar Search, Scopus, Pubmed, Science Direct, Google Scholar, Wiley Online Library. |
| Type | Academic Papers, Journals, Websites, Government Reports, |
| Keywords ‘Terms’ Searched | Infusion Pump, Syringe Driver, Smart Infusion Pump, Drug Error Reduction Software, Drug Library, Electronic Health Record, Auto-Programming, Automation, Closed Loop Medication Systems, Medication Errors/prevention & Control Dispensing Cabinets. |
| Criteria | English Language, Years 2002 to 2018 for CCLIMS, SIP, DERS, BCMA, etc.
All years for Infusion pump history timeline |
2.4 Infusion Pumps from the beginning
There are mostly two types of infusion pumps used in medicine. Large-volume (Volumetric) infusion pumps and small-volume syringe pumps. Volumetric pumps either use a peristaltic pump head or piston driven drive head which delivers nutrient solutions large enough to feed a patient. These peristaltic/piston type pumps generally use a disposable dedicated giving set which normally comprises of either a silicone section (for the peristalitic type device) or a piston cassette type (which usually comprises of a piston receiving vessel with a stretched silicone membrane inside it. Small-volume syringe pumps infuse more accurately by using a computer controlled stepper motor attached to a lead screw which in turn activates the plunger on a disposable syringe. Syringe pumps and are generally used for medicines such as opiates and inotropes (Lee, 2015).
Infusion pumps are found in every ward in every hospital today, this was not always the case, the first commercial syringe driver was developed in 1951 and had only a fixed rate of 1ml/hr using a glass syringe (see figure 2-1 below).

Figure 2‑1:- A 1951 Mechanical Syringe pump (B. Braun, 2018b)
Infusion pumps first emerged in UK and Ireland between 1970’s and 1980’s these were very simple limited mechanical devices with basic functions and no safety features and flawed user control designs. Interestingly before 1970 in the UK medications (because of their high risk) were only allowed to be administered by an anaesthetist or medical doctor. In 1976 the Breckenridge report recommended for the first time that a nurse can be trained up with a new skill to give a drug with an IV fluids infusion (Lee, 2015).
In today’s world infusion pumps use micro-supercomputers with 40 years of technological developments behind them, (examples of these pumps from two different manufacturers are shown in figure 2-2). This technology aids and governs their use but most importantly adds a layer of safety and helps protects patients from medication errors (which unfortunately still is a common occurrence with these complicated medical devices) (Ohashi et al., 2014).
Figure 2‑2:- Today’s infusion pump designs (BD, 2018, B. Braun, 2018a)
2.5 Smart Infusion Pumps with Drug Error Reduction Software (DERS)
‘Smart’ infusion pumps SIP’s (with DERS) were the first wave of defence against the standard ‘dumb’ infusion pump which could be programmed to infuse drugs/fluids at any setting allowed within the range of that device. These ‘dumb’ pumps gave no alerts to the user if a high rate was inputted in error or a litre of fluid was set to be infused instead of 100ml (to a child for example). A safety net was needed, hence the birth of the smart infusion pump. A standard pump (without software to assistance the user) could accept an infusion rate from 0.1ml/hr to over 999ml/hr. Mistakes in interpreting the infusion rate for the volume to be infused (VTBI) could cause a fatal administration dose (Proctor, 2014). For over 30 years errors with infusion pumps and pump programming still remain to be a critical issue in infusion therapy. The ECRI Institute’ in 2017 had infusion pumps in the number one place on the Health Devices Groups list of the ‘Top 10 Health Technology Hazards’ (ECRI, 2018).
Computer chip technology advancements made it possible for pump manufacturers to make customisable (by the users) drug safety management software by employing electronically erasable programmable read-only memory (EEPROM). This hardware coupled with the required software allowed the clinicians and pharmacists to develop a hospital wide drug library with specific hard and soft limits (Vanderveen, 2018). Now armed with standard concentration infusions within specific areas of the hospital for each drug and patients weight bands this was a great development specially for Paediatric and Neonatal ICU patients. The implementation of SIP’s significantly improved IV medication safety (Howlett, 2018). Since the 14 years that SIP’s have been in development their introduction has promoted standardisation and greatly reduced programming medication errors. Programming a IV infusion pump is one small piece in the complex process of delivering error free infusion therapy. In a study, 20 months after the introduction of SIP’s pumps by Pang et al, a repeat audit was supported for 27 days. The results were a reduction of errors from 18% before the installation of the SIP’s to a reduction of errors by 47% from 18% to 9.4% afterwards. What is more significant is where the drug library (DERS) was used the error rate dropped by 79% from 18% to 3.6%, See Figure 2-3 (not all drugs are in the library and in some cases the clinicians choose to go outside the drug library and use the manual function)(Pang et al., 2011)
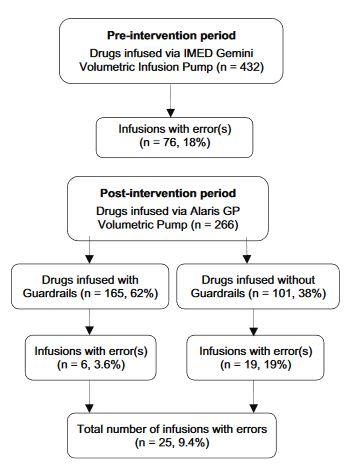
Figure 2‑3:- Distribution of errors during the pre- and post- intervention periods (Pang et al., 2011).
Although the results from the introduction and use of SIPs are proved to reduce medication errors they is still room for improvements as the study by Trbovich et al. shows, that even with the introduction of soft drug dosage limits harm could still get through to the smallest of patients, children. Trbovich et al. proved that hard limits work but unfortunately the soft limit warnings were mostly dismissed as nuisance alarms. Armed with is knowledge the call for more safety focused mechanisms like bar-coded medications linking the patients weight and size from an electronic patient record (EPR) to an electronic medication order (CPOE) thus ensuring accurate safety limits for the weight of the patient are in place (Trbovich et al., 2010).
2.6 Electronic Health Record / Electronic Medical Record (EHR / EMR)
An electronic / digital assembly of a person’s personal medical information. An EHR consists of a complete medical history for that individual, comprising of medication information, treatment records and diagnoses. The advantages to capturing information digitally and having a complete picture of an individual’s health status (which is easily accessible by clinicians), and is sharable from one institution to another all from different sources. This interoperability allows for much easier diagnoses and by getting actuate patient history means better services and outcomes for the patients receiving treatment (Stacy, 2013).
Safran et.al. (2010) developed a system that allowed the EMR to monitor the SIP’s considering previous dosage levels and patient demographics (weight, age, gender etc). This study continued for 22.5 months in a 24 bed ICU, monitoring 23 high-risk infusions, this system averaged 1.4 alerts a day which 14% were found to have prevented harm. This advanced integration between an EMR in 2010 showed the benefits of what errors can be addressed with the elimination of manually programming SIP’s (Safran et al., 2010)(Pettus and Vanderveen, 2013).
2.7 Computerised Physician Order Entry (CPOE)
CPOE is normally an extension to a clinical information system or EMR. These electronic order systems allow the medical practitioner to enter instructions (like prescribing medicines, ordering an x-ray or getting a lab result) for the treatment of patients under their care. CPOE systems reduce transcription errors and increase efficiency by reducing the time to write and distribute orders (Linda T. Kohn, 2000). HDO’s implement CPOE to help prevent adverse drug errors and this increased access to patient data improves clinical standard adherence (Baysari et al., 2018). It has been shown by published studies that CPOE systems have reduced adverse drug events up to 81% (Koppel et al., 2005). Similarly research carried out in a paediatric hospital’s wards and ICU over 6 months, returned results of a 44% reduction in preventable medication errors and 63% in potential medication errors in a comparison study before and after the implementation of a CPOE system (Holdsworth et al., 2008). Another study which highlighted the benefits of a CPOE system across 3 HDO’s (systemwide reduction of medication errors by 75%), recommended that the optimization of workflow needs to be engaged by the multidisciplinary team before the implementation of a CPOE system to ensure inter-professional collaboration (Chung et al., 2018). HDO’s looking to implement a CPOE are recommended to implement as many verified clinical decision support systems (CDSS) that are available in order to catch medication errors before they reach the patient. This report suggests that if a clinician is subject to an alert, that when the alert is resolved the user interface places the operator back into workflow that they had been interrupted from (before the Alert occurred). By following this method, this process can protect the user from Alert fatigue (Kuperman et al., 2007). Alert fatigue can govern how well a new system is accepted into existing work flows, if clinicians regularly have to ignore and override alerts, the alerts become invaluable (Page et al., 2017).
2.8 Barcode Medication Labelling (BCMA)
In healthcare, Barcode Medication Labelling was developed to help prevent medication errors, improve quality, safety and create electronic records of the medication administration process. In the United States BCMA’s became mandated by the federal government on all prescription medications (Cescon and Ethchells, 2008). In a dissertation undertaken by Holecek (2011) on medication errors, before, during and after the implementation of a BCMA in a hospital setting, The administration errors decreased steadily in all times periods throughout the study (Holecek, 2011)(Trbovich et al., 2010).
In 2006 a time-motion study conducted by Poon et.al. (2006) on the changes of workflow for nurses (in a 739-bed hospital) who recently implemented a BCMA system. The study showed that if a HIT system was correctly implemented and the staff were adequately trained, (BCMA systems require a change to workflow and this could lead to a reduced amount of time the nurse was at a patient’s bedside), that the introduction of a BCMA would influence the nurse’s workflow but not add to the time needed to make up (or in this case print) a drug label. This study consisted of 232 observation sessions over 10 months each lasting 2 hours. The results showed that before the introduction of the BCMA’s 26.5% of the nurses time was spent on medication administration, and after it actually reduced to 24.5% (Poon et al., 2006). A similar study by Samaranayake et al. (2014) came up with a similar conclusion as Poon et.al (2006), with the nursing staff involved stating that the introduction of a BCMA helped them with the accuracy of medication administration (Samaranayake et al., 2014, Poon et al., 2006).
BCMA has been proven to reduced medication error in some cases by large percentages (Patterson et al., 2002). In research carried out by Morriss et al. (2009), a 47% reduction in the risk of adverse drug events were prevented in a 92,398 dose administrated study (Morriss et al., 2009). Similarly Helmons et al. (2009) found BCMA reduced medication errors by 58% in the medical-surgical ward but oddly not in the ICU, (this was believed to be down to time delays in getting medications to patients in ICU) (Helmons et al., 2009).
2.9 Overview of Complete Closed Loop Infusion Management Systems
Complete Closed Loop infusion Management Systems bring together all the individual devices with their advancements in hardware and in software design becoming one ‘truly intelligent’ system. SIP, DERS, BCMA, CPOE and most importantly the EHR/EMR. As mentioned in the introduction, the intent is to manage patient data better and to configure devices into systems that as a whole provide more functionality and safety then the individual equipment can do when used as standalone devices (PSQH, 2016).
2.9.1 Work/Process Flow & Auto-programming
In a CCLIMS the Physician enters a prescription using CPOE, the Pharmacy (or ICU nursing) staff access the EHR, make up the patient’s prescription. The EHR system will then print a barcoded label (BCMA) with all the necessary information on it (for an example of a medication bar-code printer see figure 2-4). The bar-code printer shown in figure 2-4 has a scanner built into it where a quick check of the labels content is needed for checking purposes.

Figure 2‑4:- Barcode Label Printer in Pharmacy (Melsungen1, 2017)
In the next step, the nurse receives the medication from pharmacy, or make it up so the next steps are to scan the patient, pump and themselves (the caregiver) (Pettus and Vanderveen, 2013). Now this is the clever part, only if there is a valid prescription in the EMR from the CPOE program the system will auto-programme the SIP with the correct data i.e. the drug name, volume and concentration, infusion rate and VTBI. The caregiver must then confirm this ‘automatically populated information’ before the infusion can start (the caregiver is ultimately responsible for the medication administration by law, not the system). What should be noted is that the DERS within the SIP will continue to work independently from the auto-programming technology (and all the usual safety features on SIP’s are still active as they are standalone even when the network might be unavailable). Remember, communication and pump setup information sharing is not possible if the patient/pump association has not been verified (Proctor, 2014). Figure 2-5, shows graphically a good flow to the order entry and infusion data integration (Pettus and Vanderveen, 2013).
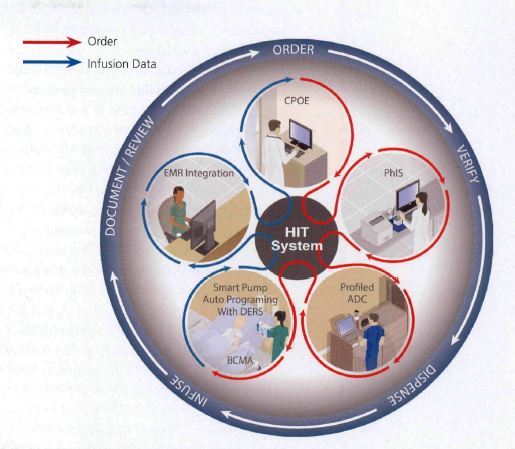
Figure 2‑5:- Integrated closed loop medication safety system (A system of Systems) (Pettus and Vanderveen, 2013)
2.9.2 Auto-Documentation
Auto-documentation delivers considerable rewards in productivity gains for nurses when charting multiple IV infusions on critical ICU patients. Prior to auto-documentation this task was very time consuming for the nurse’s taking away clinical nursing time from their patients to do extensive paperwork. Pettus et. al (2017) discussed the substantial amount of time needed to manually chart IV infusions (see figure 2-6 for an example of systems components needed), this charting can distract the nurse and may introduce serious errors. It was discovered that the seamless electronic transfer of the SIP’s data to the EMR gives the nurse back much needed time for direct patient care. The caregivers role has changed (from the previous practice of medication administration calculations ) and is now to review and confirm all the infusion data going from the SIP’s to the EMR is correct and complete (Pettus et al., 2017). This automated action is welcomed by the nursing staff and was included in a technology ‘wish list’ for intelligent infusion systems (Proctor, 2014).
A PHD study by J. Melius (2012) looked at the relationship between mathematics anxiety and nurses working environment, her study subject ranged from nurses not liking mathematics, to medication administration stress (when the nurses were exhausted at the end of a 12-hour shift). The number of hours a nurse worked was in a direct relationship with the mathematics anxiety score (Melius, 2012). Basically, nurses don’t like mathematics, so rather than force an issue (where there is a lack of natural ability) and look for unreliable solutions, why not use the technology available in a CCLIMS to remove this burden on nurses and improve medication safety.
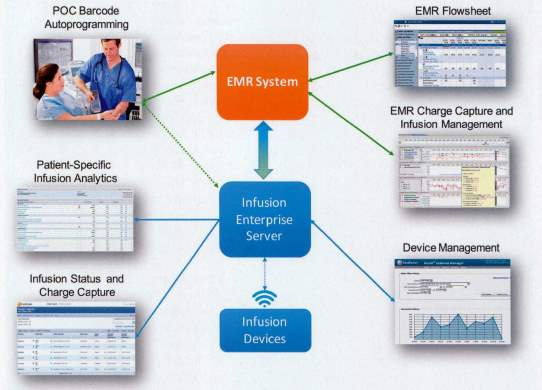
Figure 2‑6:- Graphical overview of system components (Pettus et al., 2017)
In this dissertation some of the steps involved will be simplified to allow for a clearer overview on how a complete CCLIMS sits within a HDO. The advanced pharmacy systems element will primarily be the most ‘just excepted’ example of a simplification of process and work flows (as to deeply evaluate it is outside the scope of this study). The Researcher will discuss the pharmacy element of this research further in the coming chapters. The main reason for the pharmacy to be treated differently is the way the pharmacy services in Europe and the work practices differ to the pharmacy services in the United States (more about these differences later).
2.10 Will a CCLIMS Help Prevent Medication Errors?
Over the last 10 years CCLIMS development certainly takes the biggest step towards this challenge and has been proven to be successful within private hospitals in the United States. The complexity of a CCLIMS installation has to date always relied on an external equipment vendor and presents too complex for individual hospitals to manage only with internal departments expertise (Pettus and Vanderveen, 2013). Fig 10 shows where the individual systems stop and smart pumps take over, AP fills this GAP but only if the interoperability between the systems is 100%.
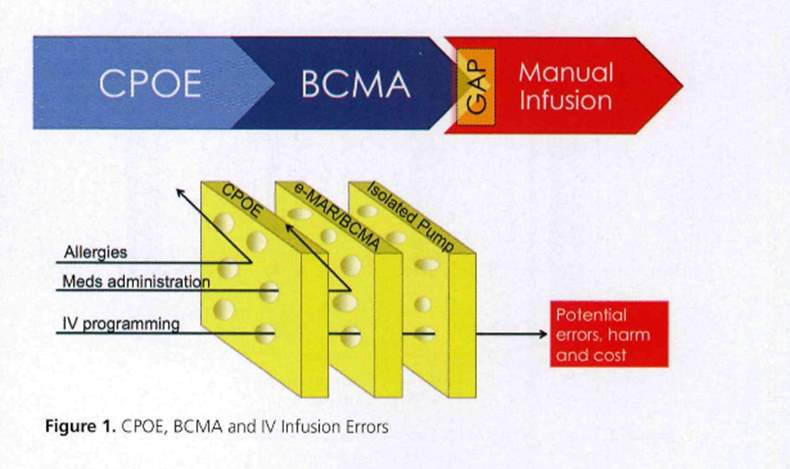
Figure 2‑7:- The GAP Auto programming fills (Pettus and Vanderveen, 2013)
It appears to be a clear and concise message to the HDO’s that if all the hurdles with interoperability can be overcome then some real strides will be made in eradicating prescribed medications differing from the administered amount by closing the loop in infusion and medication safety (Pettus et al., 2017). The challenges don’t stop with the technology’s integration. The many users of a CCLIMS need to adapt from their current work practices to the demands of the new system. In many cases the technical advancements (once the advances are adapted to) will actually relieve the users (nurses) stress in drug admixture and administration (Brady, 2010). A study on paediatric nurses’ knowledge of pharmacology resulted in an outcome of 61.5% showed insufficient knowledge when administering infusions to paediatric patients. Evidence-based results also state that 9.2% of the reported incorrect doses monitored has serious significances for the children involved (Lan et al., 2014). These studies and many more, place the evidence front and centre to why medication safety through implementing a CCLIMS in our paediatric hospital should be a priory in the battle against adverse drug events (Chung et al., 2018, Pham et al., 2016, Pettus and Vanderveen, 2013, Prusch et al., 2011, Russell et al., 2010, Hennings et al., 2010) .
2.11 Improved work flow for SIP & Drug Library updating using CCLIMS
Biomedical engineering departments have a difficult job in maintaining hundreds and in some hospitals thousands of infusion devices. Not only do these infusion devices need maintenance and calibrations carried out there has been the added task of rolling out the latest version of the hospital pharmacies drug library (PSQH, 2016). When these pumps were single smart pumps without being networked, this task involved locating and upgrading every pump singly or in small batches, this procedure takes up valuable manpower hours and is often fault ridden due to devices not being located correctly. The advantages of having all the pumps networked for interoperability gives the biomedical engineer a powerful new engineering software tool. Drug library updates and real-time pump location history (not to mention the usefulness of real time battery status for replacement purposes etc). Drug library changes need to be carefully coordinated with the pharmacy and CPOE systems to ensure correct connections between standard drug concentrations and weight banding are preserved. Incidences of mismatched prescriptions will be high if the drug library does not mirror the ordering system (AAMI, 2014). Each pump should be able to be traceable on a Biomedical/Clinical Engineering dashboard (similar to a pharmacy dashboard) and each SIP that is infusing in the hospital will be able to be monitored from this infusion portal regardless of the SIP’s location (Pettus et al., 2017, Pettus and Vanderveen, 2013). These system tools will change the current pump maintenance practices but will increase efficiencies it the process.
2.11.1 Faults and a Complex System who’s problem is it?
The traditional repair route in any HDO would be to send the individual item/pump etc. to the Clinical Engineering (CE) department for repair. Now considering the fact that with interoperability all these medical devices are now connected and with the technologies involved hospital staff will need to be aware that if a pump doesn’t work or if the drug library won’t upload is it the SIP or the network? Who can determine what is wrong IT or CE? When the process needs all the systems to work together to achieve the goal (i.e. EMR’s, SIP’s, CPOE, BCMA & IT Networks) the problem becomes far more challenging and complex to identify. A very high level of the interdisciplinary interdepartmental understanding will be required to find a solution and rectify the issues and stop it happening again (Pettus et al., 2017). The working practices within and between the ICT department and CE department will change dramatically as the informatics roles increase in the hospital. The introduction of these complex systems will have a dramatic effect on the socio-technical dynamics within the hospital and the HDO will need to give considerable thought on employing an informatics trained medical device engineer can understand the complete system and has the skills to deal with issues and maintain these complex systems. The current literature on this subject is sparse and any reports found indicates that the equipment vendors are currently supporting and controlling the systems as a whole. These are mainly in private hospitals within the United States.
2.12 Summary
The introduction of a CCLIMS at any level within a HDO will lead to a change of clinical working practices and can shift certain responsibilities from one discipline to another (e.g. drug admixture could move from the ICU nurses’ role into the Pharmacy department). It is imperative to appraise these working practices before implementing any computerised clinical system so that the HDO can make an informed decision on deployment before any adverse event or risks of error can affect patient safety.
2.13 Conclusion
The literature review confirms the advancements in medication infusion systems in reducing medication error and increasing patient safety. Regardless of the cost of interoperability every HDO should exhaust every means to deliver this safety driven technology for the wellbeing of their patients. The changes to working practices in the literature reviewed signals a comparable work load for most of the staff involved in medication administration, e.g. BCMA label printing and affixture are offset by the removal of other tasks (like handwriting out drug labels etc.). After staff get familiar with the operation of the system the clinicians’ will benefit from the increased information available to care for their patients when and where the need arises.
The next chapter describes the methods used for the research methodology used to provide the discoveries and findings to answer the research question.
Research Methodology
3.1 Introduction
The literature review in the previous chapter established that with the advances in infusion pump technology and the associated prescribing, labelling and electronic charting systems can (if implemented correctly) help to prevent medication errors. One noticeable gap in the information reviewed is, what work practice’s and information flow changes does a HDO have to consider in order to successfully implement a system as complex as a CCLIMS across all the affected departments and different disciplines that combine together to provide a medication service to the PICU. Another consideration (as mentioned in chapter 1) is that the installation of a CCLIMS will need to completely integrate with the existing systems which are functioning already and in clinical use within the HDO.
3.1.1 Review of the Research Question
Remembering the motivation of reducing medication errors associated with the intensive care of children using the combination of available technologies which combined together form a CCLIMS, the research question is: –
What are the changes to work practices through the proposed implementation of a closed loop infusion therapy system into an existing working paediatric hospital?
3.2 Reminder of the Study aims and objectives
The aims of this research are to: –
1) Determine the conditions under which a completely closed loop infusion therapy system can be safely integrated into a working paediatric hospital.
2) Investigate and document what are the changes to work practices for the following stakeholder’s: –
- Pharmacy professionals
- Nursing professionals
- Clinicians
- Clinical Engineers
- ICT professionals
It is anticipated that the improved overall understanding of a CCLIMS will help to guide and prepare the healthcare institution towards the correct approach for purchasing and implementing a CCLIMS. It is also expected that the sociotechnical element of the installation of a CCLIMS can be partly prepared for from the outcomes of this research study which should help the many different professionals who will be required to operate and maintain it.
3.3 Study design
This study will use mainly qualitative and applied research methodologies. The information gathered from semi-structured individual interviews with selected staff on the existing procedural work practices (when delivering a medication with an infusion pump to a patient) in their departments. The information gathered will then be compared against the new perceived work practices which are associated with the proposed system (these new work and information flows shall be described by the researcher, with the utilisation of a digital media source coupled with an in-depth discussion of the technical detail of the perceived use of the proposed closed loop system). It is proposed that this approach will add to the knowledge and awareness towards the changes involved in implementing such an intricate system across many departments and disciplines.
3.4 Participant Recruitment and Rationale for selection
Participants are work colleagues from OLCHC and are known to the researcher. They will be purposefully selected for their expert knowledge in each of their specialities, this recruitment procedure will be purely on a voluntary basis.
It is proposed to interview five to seven individuals in Pharmacy, Medical, Nursing, Clinical Engineering and ICT backgrounds. These purposefully selected experts (in their professions) coupled with their experience and their in-depth knowledge of the current working practices (within a paediatric hospital’s ICU) will guide the researcher with the knowledge to critique the required work practices of the proposed CCLIMS. These five to seven individuals represent the total number needed for the research to be carried out. The rationale behind only interviewing one or two professionals from each department is backed up by the researcher’s outlining knowledge of the procedure’s and systems used to deliver medications with infusion pumps to patients within a PICU. The Researcher coupled with the knowledge of knowing the selected interviewees and their skills and comparing this information with the knowledge gained from a wider range of subjects (who don’t have as deep a knowledge of the systems in use) will only dilute the accuracy of the findings.
The researcher is a working colleague with no influence over the selected participants with regards to participation in this study. Their participation will be completely on a voluntary basis. Each participant will be invited to take part in writing by the researcher (after an informal conversation to ascertain if it is something they would be willing and interested to take part in).
3.5 Study methodology
3.5.1 Semi structured interviews
The researcher proposes to carry out semi structured interviews with selected staff (using purposeful sampling) from: – (See Table 3-1 Below),(Rogers, 2018, Tuckett, 2004).
Table 3‑1 Purposefully Selected Participants
| Department | Title | Role |
| Clinical Care | Clinician /ICU Intensivist | Cares for critically ill patients mostly in the ICU and Theatre environments. |
| Pharmacy | Informatics Pharmacist | To ensure medication guidelines are supported within the HDO while utilising technology |
| Nursing | Clinical Nurse Specialist (CNS) | Trainer on smart pump technology and application |
| Clinical Engineering | Clinical Engineering Technician | Medical Device management and support |
| ICT | System administrator | Clinical Data Gatekeeper for PICU’s EPR system. |
The structure for all the interviews will be similar (the direction and content will differ greatly with the variation of participants and their disciplines) and should produce a good overview of the methods under review: –
3.5.2 Step 1 (Interview explaining existing practices)
Firstly, the participant will be asked to describe and step though their current procedures in detail with regards to delivering an infusion to a patient. (this information will be recorded on a digital data device and later transcribed and returned to the participant for an accuracy check and amendment (if deemed necessary).
3.5.3 Step 2 (Digital media presentation of a CCLIMS)
Secondly, the researcher will display a tutorial/informative short video (less than 5 minutes long) on a closed loop infusion therapy system in operation http://www.space.bbraun.com/cps/rde/xchg/hc-space-en-int/hs.xsl/7743.html. This video shows how a CCLIMS can ensure the 5 rights of medication delivery stepping through all the phases quickly and safely. This commercially available video simplifies the complex integrations between the user, the information systems and the physical product including the patient which gives a good awareness of the new work practices to be introduced when a system like this is to be implemented.
3.5.4 Step 3 (Critiquing of the proposed new work flow)
Thirdly, the participant will be asked to discuss the new therapy delivery techniques and critique the finer details with the researcher who has studied the new system in depth. Again, this information will be recorded on a digital data device and later transcribed and returned to the participant for an accuracy check and amendment (if deemed necessary).
The Comparisons in the work and information flow practices will be deliberated the main differences written out and where applicable highlighted using unified modelling language (UML) activity diagrams. The varying roles between the different disciplines will be prevalent, but this will help develop a broad base of knowledge with interesting interoperability comparisons.
3.6 Ethical Considerations
Before any participant recruitment could take place in the HDO the researcher had to apply for ethics approval from both the HDO (OLCHC) and Trinity College Dublin through the School of Computer Science and Statistics (SCSS) Ethics committee. The application to the HDO was a long process delayed further by the rescheduling of meeting dates etc.
- The full HDO (OLCHC) application is in Appendix B.
- The HDO (OLCHC) Approval Letter was received on the 28th of February and is in Appendix A.
Once the HDO had approved the ethics application for this research an application to Trinity College Dublin could be submitted. The application to the Ethics committee of the SCSS was made online through the Trinity Ethics WebApp (see Appendix C).
- The SCSS Ethics Trinity application (Appendix C) was approved on the 27th April
- This application involved submitting a Research Project Proposal (Appendix D)
- An Information Sheet for Prospective Participants (Appendix E)
- An Informed Consent Form (Appendix F)
3.6.1 Potential conflict of interest
As the researcher is a work colleague and known to all the purposed participants there is a potential conflict of interest in relation to this research study. However, it is the researcher’s intention to adhere to good practice and follow the ethical code for research at all times during this study.
3.6.2 Participant debriefing arrangements
The Researcher will inform the participants of the purpose of this research. If participants request further information it will be arranged to send them the completed copy of the final dissertation in a PDF format.
3.6.3 Data Protection
As per the Data Protection acts 1988 & 2003 all data collected will be anonymized, this will be done by ensuring no participant is named or identified from their unique role in the HDO or by any information disclosed, (See 3.6.4 & 3.6.5 below). No disclosures of participants identities or any personal information will be provided, participants will be referred to by their professional title No consent is required from the data controller.
All research documents for this dissertation are stored (or will be stored) on the researcher’s personal laptop computer. This personal laptop computer is password protected and is encrypted with the safehouse explorer 448-bit encryption software program.
3.6.4 Data collected
Data will only be retained for as long as necessary by Trinity College though the researcher after which it will be destroyed using Safehouse Explorer’s shredding file facility. When digital shredding takes place, spurious data is interlaced throughout the file volumes and then deleted to leave no legible trace.
3.6.5 Confidentiality of collected data
No personal information will be collected or recorded and all data will be totally confidential and anonymous.
3.7 Conclusion
This chapter described the research methodology and rationale for the selection of participants needed to answer the research question. The ethical approval processes of the HDO and the college was explained in detail and the outcomes stated, (the full ethical application is included in Appendix A for the HDO, and in Appendix C for Trinity College Dublin, including screen grabs from the online application approval process).
The following chapter will present the findings of the investigations undertaken when preparing to answer the research question.
Results
4.1 Introduction
This chapter explores and develops the knowledge and pathways investigated during the selective interview process as outlined by the methodology in chapter 3. Each interviewee’s knowledge and experience was used to build up an accurate description of the existing work practices of the various different healthcare professionals (see table 3-1 In chapter 3) who play a role in delivering medications using smart infusion pumps to PICU patients within OLCHC.
4.2 Clinical Engineer Interview
(Full interview transcription see Appendix J)
4.2.1 Summary of the main points in step 1 (Clinical Engineer Interview)- Existing Practices
Clinical engineering (CE) is responsible for the complete management and lifecycle of medical devices within the HDO. Infusion pumps historically put a strain on CE resources as they are frequently moved for use and cleaning procedures and unfortunately see more than their fair share of rough handling lending to frequent repairs outside the manufacturer’s maintenance schedule. Due to the critical requirement for accuracy and reliability with infusion pumps, HDO nursing staff are very cautious when using these devices, and if a problem is only suspected, then the pump is returned to the CE workshop for inspection, repair and safety checks.
4.2.1.1 Smart Infusion Pumps (SIP’s)
Smart Infusion Pumps (SIP’s) are significantly more complicated than a standard infusion pump and more time consuming to maintain and service. SIP’s drug libraries (DERS) need updating whenever there is a modification needed to the medication data in the drug library; this includes parameters and default settings, hard and soft limits and drug concentrations (in any of the five weight bands) within OLCHC. The DERS update requires the CE department to individually physically find and update every SIP in the HDO with a laptop and cable using a bespoke vendor service program. The updating process is a time consuming costly and labour-intensive procedure especially when OLCHC has approximately 350 high and low volume SIP’s which all will need upgrading together. Having two different DERS software versions in one HDO is potentially dangerous for the patient as the users need training and familiarity with the software to ensure that user errors do not occur.
- It is worth noting here that Pharmacists are responsible for all the data in the drug library, they decide and set all the hard and soft limits, drug concentrations and patient weigh bands (these figures are decided on as part of a multidisciplinary team with the clinicians).
- The role of Clinical Engineering in this process is to update this information on the medical device.
4.2.1.2 The Networking of the SIP’s
The CE department is also responsible for physical networking between the SIP and the HDO’s EPR. In OLCHC an intermediate data capture module (Capsule Terminal Server by Qualcomm, this module is a serial to network bridge which has drivers to interface with most medical equipment and then output in health level 7 (HL7) standards see figure 4-1) is needed at every PICU bed space to interface the SIP data with the EPR. In OLCHC the EPR provider is the ‘IntelliSpace Critical Care and anaesthesia suite’ (ICCA) by Philips. The need for the intermediate data capture module is a requirement of the ICCA system. The SIPs utilise a serial output (RS232) interface connected to the data capture module (See figure 4-2). The CE department is responsible for the technology and maintain these wired interface connections. They are also the first point of call when a communication error between the SIP and the EPR is reported.


Figure 4‑1 Data Capture server in situate & Fig 4-1.2 SIP rack with an RS232 connection situated in PICU, OLCHC
The UML diagram below shows the manual steps needed to update the drug library on a single smart infusion pump currently in OLCHC. (Figure 4-2). This process will need to be repeated for every pump in the HDO.
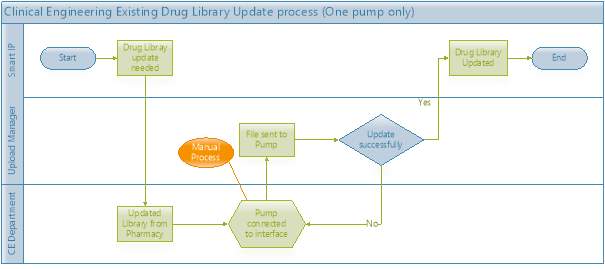
Figure 4‑2 UML Diagram; Manual steps needed to update a single pump.
4.2.2 Summary of the main points in Step 2 (Clinical Engineer Interview)
The Digital media presentation of a CCLIMS:
- The researcher and interviewee viewed the presentation video (http://www.space.bbraun.com/cps/rde/xchg/hc-space-en-int/hs.xsl/7743.html)(B.Braun, 2018).
- The SIP vendor’s maintenance software program was explored on the world wide web (OnlineSuite™, this is bespoke software from pump vendor (B.Braun))(Melsungen2, 2017). OnlineSuite™ allows the CE department to remotely monitor all networked (wired and wireless) SIP’s in the HDO. This useful software is key to assisting the CE department to monitor and update the SIP’s drug library software without having to locate and handle each SIP on a hospital-wide basis.
- More online research was carried out, and the remote upgrading and device management functionally is a feature that most of the current suppliers of auto programmable SIP’s offer to their clients (or have in development)(BD, 2018, Smiths, 2018).
4.2.3 Summary of the main points in Step 3 (Clinical Engineer Interview)
Critiquing of the proposed new workflow:
After viewing the video, the five rights of medication delivery utilising a CCLIMS was discussed at length, and the complex integrations needed between this technology and the information systems (i.e. EPR, CPOE) took place.
The SIP Device interface options available were also discussed utilising the researcher’s knowledge of a CCLIMS and the interviewee’s expertise of the existing system integrations.
One critical discovery is that when a data capture module is used through an RS232 connection data can only flow unidirectionally from the SIP. The RS232 setup does not allow the SIP to receive medication data from the EPR (See Table 4.1).
Table 4‑1 SIP Out & In-bound available connections
| Outbound SIP Data to the EPR System | In-bound EPR Data to SIP |
| Ethernet (1Gbit/s) | Ethernet (1Gbit/s) |
| Wireless LAN (Supporting 802.11b/g/n) | Wireless LAN (Supporting 802.11b/g/n) |
| RS 232 Serial Connection (9600-115kbaud) | N/A |
Another localised realisation is that the data capture module only requires one network port to function, this means up to 4 or 8 medical devices can be combined only while utilising one network point. If these SIPs need a dedicated wired connection at every PICU bed space to function reliably. Presently every network connection (at every bed icu bed space) is utilised within the PICU 1 in OLCHC. The ganged approach of using a network hub is already in use and importantly this approach is not recommended by the device manufacturers. If a connectivity issue occurs this approach to a worsening problem becomes a hard to resolve issue with the equipment vendor as the ganged approach is not recommend. The limited number of network points could be a limitation for the HDO to consider before purchasing a CCLIMS system. The installation of adding more network points to a PICU would require a full unit decant to another area of the hospital which is a complicated procedure to undertake and very disruptive to the consistency of care to the patients.
The UML diagram in Figure 4-3 shows the SIP’s Drug library update pathways when a CCLIMS is in use in a HDO. This system utilises a dedicated pump server with the ICT infrastructure to manage all the SIP’s and SIP docking stations.
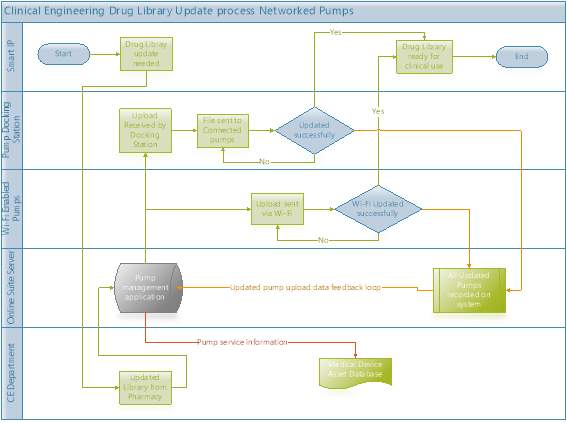
Figure 4‑3 UML Diagram; Drug Library update all networked SIP
4.3 Clinician Interview Step 1
(Full interview transcription see Appendix K)
4.3.1 Summary of the main points in Step 1 (Clinician Interview)
Existing Practices:
The Clinicians’ workflow was the most straightforward of all the disciplines involved in this study. The Clinician in OLCHC is currently provided with the use of a CPOE prescribing system which integrates with the EPR (ICCA) in both PICUs. The clinicians use order sets to prescribe multiple infusions (editing the order set’s drugs to suit each patient’s needs). The order sets consist of groups of drugs listed in standard concentrations in five weight bands. If only one or two infusions are required for a patient, the clinician will prescribe these directly not utilising the order sets (but the CPOE system still uses the drug library and limits to assist them). They currently prescribe in a range with no set starting dose. The drug library data in the CPOE system and the SIP’s drug library should be identical (this can easily not be the case), these libraries are not linked as both drug libraries require manual human input to update.
- Interoperability (or lack of it) between the two active drug libraries will be discussed further during the Pharmacists and System administrators interview summary’s.
- The SIP library updates were discussed in the Clinical Engineers interview summary.
4.3.2 Summary of the main points in Step 2 (Clinician Interview)
The Digital media presentation of a CCLIMS:
The researcher and interviewee viewed the presentation video (http://www.space.bbraun.com/cps/rde/xchg/hc-space-en-int/hs.xsl/7743.html)(B.Braun, 2018)
The Clinician was also very interested in the diagram in the video presentation of ensuring the five rights of medication safety (see figure 4-3) (Melsungen1, 2017). This diagram was of the workflow from the clinician ordering (item 1 CPOE), to drug preparation (item 2), which included the barcode labelling and onwards to the medication administration stage, including identifying the patient to the prescription through the complete process (barcode scanning item 3). The ease and efficiency of the system to match the order to the patient and the prescription (item 4) all the way to the auto-programming the infusion pump (item 5) is a significant step towards ensuring the medication safety for the patients in reducing the chances of an adverse drug event/medication error. Item 6 in the diagram is auto documentation which is currently active within the OLCHC PICU environment. The system administrator interview summary will go into the feature in depth.
Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allRelated Content
All TagsContent relating to: "Hospitals"
A hospital is a health care institution with specialised facilities and medical equipment where doctors and nurses diagnose and treat people with illnesses or injuries.
Related Articles
DMCA / Removal Request
If you are the original writer of this dissertation and no longer wish to have your work published on the UKDiss.com website then please:




