Modelling Emotion Processing in Frontotemporal Dementia
Info: 11799 words (47 pages) Dissertation
Published: 9th Dec 2019
Tagged: NeurologyAlzheimers
Literature Review
Frontotemporal Dementia
Frontotemporal Dementia (FTD) is an umbrella term to describe younger onset dementia with clinical presentations arising from progressive neurodegeneration of the frontal and temporal brain regions. Clinical presentation varies but is generally categorised as a behavioural variant of FTD (bvFTD), or two presentations where language disturbance is the primary feature: semantic dementia (SD) or progressive non-fluent aphasia (PNFA) (Mesulam, 2001; Snowden et al., 2001; Rascovsky et al., 2011; Gorno-Tempini et al., 2011) The heterogeneity of FTDs not only explained by different clinical presentation but also its diverse underlying pathologies. Frontotemporal lobar degeneration (FTLD) is the used term to describe the neuropathology of FTDs. Commonly observed groups of pathology contributed to neurodegeneration including microtubule with tau-positive (FTLD-TAU), TAR DNA-binding protein 43 (FTLD-TDP) and fused in sarcoma protein (FTLD-FUS) (Poorkaj et al., 1998; Hodges et al., 2004; Neumann et al., 2006; Neumann et al., 2009; Mackenzie et al., 2010; Mackenzie et al., 2011)
The location of predominant brain atrophy will determine the likely symptoms and behaviour changes (Devinsky, Morrell, & Vogt, 1995). Primary brain atrophies are found in the frontal and temporal area of the brain but will extend according to FTD subtypes (Rosen et al., 2002; Lam et al., 2014; Whitwell et al., 2015). The frontal and temporal regions of the brain are implicated in cognitive functions such as language, memory, and social cognition. Accordingly, inappropriate social interaction and disinhibition are often displayed by FTD patients. Impairments in language, speech, memory formation, and emotion processing, are strongly associated with temporal atrophy and its network integrity to the frontal lobe (Von Der Heide, Skipper, & Olson, 2013).
Behavioural variant of FTD
The clinical presentation of bvFTD includes inappropriate social actions and altered personality (Mendez et al., 2013; Dickerson, 2016). Six main clinical features are considered towards a diagnosis of bvFTD (Rascovsky et al., 2011). This include an inability to empathise, apathetic behaviour, disinhibition, compulsive or stereotypical behaviour, significant dietary problem, reduced executive function (Piguet et al., 2011). Brain imaging identified significant altered right lateralised fronto-temporal regions starting from the ventromedial area, which implicated in the disintegration of social cognition network (Lu et al., 2013). More specifically, anterior cingulate cortex-fronto insular network is the vulnerable pathways in bvFTD. These affected paralimbic regions can be observed since the early stage of FTD (Seeley et al., 2008). As the disease progressing to the later stage, the pattern of atrophy becomes more difuse affecting the neighbouring regions (Devenney et al., 2015). Underlying patholoogical of bvFTD are more heterogeneous compared to the language variants, 45% is associated with FTLD-tau, 32% is FTLD-TDP, and other 13% is FTLD-FUS related (Chare et al., 2014).
Language Variants of FTD
Aphasia or language impairment characterises primary progressive aphasia (PPA) (Mesulam, 2001). In general, PPA is used as a description of individuals who exhibit problem in utilise and understand words which significantly affect their the daily living, but the rest of cognition is comparatively well preserved (Mesulam, 2001). More specifically in FTD, two subtypes of PPA are considered, semantic dementia (SD) and progressive non-fluent aphasia (PNFA) (Knibb et al., 2006; Gorno-Tempini et al., 2011).
SD is characterized by the progression lost of semantic memory and progressive breakdown of single-word comprehension (Hodges et al., 1992; Gorno-Tempini et al., 2011). Not uncommonly, SD patients also report presenting with behavioural deficit that closely resembles the behavioral variants (Kumfor & Piguet, 2012). Neuroimaging studies show that SD patients have brain atrophy predominantly in the left temporal lobe (SD left), which underlies the loss of semantic knowledge. Additionally, studies have suggested that atrophy also presents in the right temporal region (SD right), particularly as disease progressing (Snowden, Goulding & Neary, 1989; Kumfor et al., 2016). Right SD has similar affected regions with bvFTD, such as anterior cingulate and oribitofrontal cortex, which makes it common to observe profound social cognition as well as semantic deficit since the early stage (Kumfor et al., 2016). FTLD-TDP were found to be correlated with at least 70% from the total cases, and it is believed as SD’s main pathology (Chare et al., 2014).
In PNFA, the clinical presentations usually begin with difficulty in speech production, agrammatism, anomia before gradually become mutism (Grossman et al., 1996; Turner et al., 1996). Most patients have preserved general cognition function in early stage (Neary et al., 1998). Moderate difficulty with emotion processing tasks is observed in a few cases of PNFA (Kumfor & Piguet, 2012). Clinical presentations of PNFA are associated with cortical thinning in the left hemisphere of inferior frontal and anterior insular regions (Gorno-Tempini et al., 2004). Post mortem study shows that 50% of the patients had tau pathology, and the rest either had AD or TDP (Chare et al., 2014).
Impairment of Social Cognition: Emotion Processing
Social cognition is a broad term to describe information processing Is used to gauge what others feel and think in a social setting (Frith & Frith, 2008; Frith, 2009). Complex social cognition is a human-specific evolutionary trait to support knowledge sharing and decision making, which essential for collaborative interactions (Bandura, 2001; Hermann et al., 2007). Social cognition includes different dimensions such as recognising emotion, understanding sarcasm, predicting what other would do in different social scenarios, and the theory of mind (Sprong et al., 2007). Considering that social cognition is a broad and sometimes an overlapping construct, this project will centralise on emotion aspect of social cognition. Why emotion tho?
The arrangement of our neuronal networks shapes the foundation of social cogniton, therefore more often than not, social cognition is considered as biological inherited that requires less refective mediation of higher mental processing. Furthermore, this nature also explains how certain types of disease are prone to social cognition problem. Social cognition network is argued as the last networks that evolutionary developed, making it susceptible to certain pathology.
Deficit in social cognition are observed in frontotemporal dementia. These deficits arise from the disintegration of neural network that are known to support social cognition processes, namely orbitofrontal cortex and temporal (Ibanez & Manes, 2012; Baez, Garcia, & Ibanez, 2017). Atrophy of this network is present to some degree in almost all types of dementia but it is particularly prominent in FTD, especially in the behavioural variant (Snowden et al., 2003). This is reflected by the more severe manifestations of social cognition deficits in bvFTD compared with other dementias including emotion recognition, the theory of mind, the ability to understand sarcasm (Eslinger et al., 2006; Kumfor & Piguet, 2012). – more papers
Emotion in SD
Both SD and bvFTD present deficit in emotion recognition. Not uncommonly, SD patients also report dificulty in recognising emotions, and more so for negative emotions (Kumfor & Piguet, 2012). SD have a problem in labelling emotion due to semantic deficit. However, SD still have a problem in non verbal emotion task, such as matching facial expression (Miller et al., 2012). These distinct impairment in labelling emotion and discriminating emotion expression reflects two dominant patterns of atrophy in SD (dominant left or right atrophy) (Snowden et al., 2017). Left atrophies is associated with disintegrity of semantic network (Kumfor et al., 2016). Emotional problem in patients with left atrophies, may be mediated by language problem (Miller et al., 2012). Emotional problem in right sided SD patients, may be due to distributed disintegrity of social-cognition network (Kumfor et al., 2016). Right-SD and bvFTD have been reported to exhibit the same magnitude of emotion deficit. Besides both variants have the overlapping impairent in social-cogntiion networks, however, right-SD have more deficit in face perception network; suggesting that deficit in face perception may be the underlying cause for emotion recognition in right-SD (Kamminga et al., 2014).
Emotion in PNFA
Limited studies have reported emotion dimension of social cognition in PNFA. One study shows that PNFA patients report poor performance in recognising emotion ollowed by profound deficit in face perception (Cuoto et al., 2013). Furthermore, the same study indicates that emotion recognition deficit has strong association with temporal region, namely bilateral insula and right temporal pole in PNFA (Cuoto et al., 2013). While the impairment of emotion in bvFTD is correlated with more frontral location of the brain, disengagement of basic perceptul networks affects maybe the main predictor of emotion deficit in PNFA (Zhang et al., 2013). Result from different study also indicates that PNFA also exhibit problem in discriminating emotional prosody that may have a basic vocal recognition (Rohrer et al., 2012). Taken together, the emotion recognition in bvFTD is more genuine manfestation of emotion processing, while in PNFA, emotion is affected by basic cognitions (Cuoto et al., 2013).
Social cognition and emotion of bvFTD versus AD
Shared psychopathological problem with AD, often leads clinicians to misdiagnose FTD (Lindau et al., 2000). Although AD patients have a hallmark of episodic memory impairment, the patients also present behavioural issues (Tariot et al., 1995). Social cognition problems that present in FTD often found in AD. A study, for example, reported impairment of social cognition in the early development of AD could indicate the progression to a full-blown AD (Bediou et al., 2009). Additionally, FTD patients also presented with cognitive problems similar to AD. Studies reported that both FTD and AD have same performance in episodic memory test (Hornberger et al., 2010: Pennington, Hodges, & Hornberger, 2011).
In comparison with AD and other types of dementias, emotion impairment is one of the most distinctive features of FTD in early stage (Bathgate et al., 2001). Latest study reported that general cognition and overall social cognition performance are not very sensitive to differentiate behavioral variant FTD from AD (Gossink et al., 2018). Narrowing to emotional aspect of social cognition, on the other hand, can distinguish between dementia groups (Gossink et al., 2018).
Although emotion problems are both present in FTD and AD, the underlying mechanisms of this impairment may be different. It is suggested the weight of cognitive funtion contribution to emotional impairment in AD and FTD are different (Lavenu et al., 1999; Lavenu & Pasquier, 2005; Miller et al., 2012). For example, impaired semantic ability causes FTD performed worse than AD in labelling emotion expression (Lavenu et al., 1999). Comorbid cognitive decline, however, is unlikely to be the main causes of emotion impairment in FTD and AD (Kumfor & Piguet, 2012). FTD patients may be able to perceive the important cues but failed in synthesising different features as a whole meaningful information (Hutchings et al., 2017). Furthermore, a study shows that lack of awarenes of deficit in emotion processing not only differentiate FTD from the healthy control but also with the AD (Scherling et al., 2017).
Emotional changes in concert with cognitive problems have different interactions in FTD and AD. It provokes several questions regarding emotion processing: How emotion and cognition inform us about the clinical diagnosis of FTD variants/ between FTD and AD/ between FTD and normal ageing?. Do emotion processing is a primary problem or secondary to cognition in different clinical population/ in different clinical stage?. To answer these questions a more comprehensive approach is needed. The complexity of emotion problem in FTD and other types of dementia requires an explicit representation using modelling. The model should incorporate data from the different level of analysis to provide comprehensive explanations of what is emotion and how emotion is generated.
Computational Modelling of Emotion
Computational modelling (CM) of cognition or computational psychology is an avenue to understanding complex systems of human behaviour (Fodor, 2001). CM is any computer models or mathematical formalisation that can derive a mechanistic explanation of a phenomenon (Busemeyer et al., 2015). Traditional statistical methods, which often ignore detailed processes, cannot capture complex data or different level of descriptions, and therefore fail to provide a realistic representation of a phenomenon (Vallacher, Read, & Nowak, 2017). CM allows the representation of variables from a different level of descriptions and distils the most relevant predictors with detailed process-based interactions (Sun, 2008). The strength of CM lies in its vast array of modelling techniques and methods (for example, model-based and data-driven approach). CM enables extraction of rich information from multidimensional data to make a better understanding of human behaviour (Adolphs et al., 2016).
As a complex construct that consists of various dimensions, which could be a generic definition problem due to different theoretical appraoches (Ekman, 2016), emotion needs to be computationally modeled to make a systematic detail (Marsella, Gratch, & Petta, 2010; Hudlicka, 2011; Hudlicka, 2014). Computational model should formalise and transform the theory of emotion to answer fundamental questions such as what are the basic components and how the component of emotions interact (Scherer, 2009; Hudlicka, 2011). There are two general operational definitions of computational modelling of emotion. First, it is related to modelling theoretical constructs of emotion (Bosse, Pontier, & Treur, 2010). Second, it is to model emotion of human and then apply it to artificial intelligent agents (Rodriguez & Ramous, 2015). The first disciplines is the main focus of psychology and cognitive neuroscience, although some overlapping works can be observed with other fields such as engeneering and computer science which more interested with the second definition (Scherer, 2009; Reisenzein et al., 2013). Futhermore, computational modelling of emotion theory can be devided into two forms of work. First, is to model emotion using data driven technique or machine learning. Second, is to formulate a mathematical model that can best explain emotion theories (Scherer, Banzinger, & Roesch, 2010; Vallacher, Read, & Nowak, 2017).
CM of emotion has not been applied to in FTD to date. A novel avenue that will enable to establish a realistic representation of emotion processing in FTD by capturing multiple predictors simultaneously (Duch, 2007; Iturria-Medina & Evans, 2015) CM can then represent these variables from different angles and find hidden correlations that cannot be observed using conventional statistical methods (Herland, Khoshgoftaar, & Wald, 2014).
CM can visualise the relationship between psychological and biological underpinning of social cognition impairment more vividly. This relation can be drawn by CM in any directions (linear or non-linear) using different mathematical formulas, and eventually test the model through statistical validation or computerised-simulations (Dayan & Williams, 2006; James et al., 2015). Therefore, CM is expected to capture every dimension of emotion processing in different variants of FTD or even bigger comparison with other diseases (Körding et al., 2007; Anticevic, Schleifer, & Youngsun, 2015; Teufel & Fletcher, 2016). Hopefully, CM render a uniform representation and a working system with broad application in the clinical setting.
In summary, the problem in understanding how cognitive decline contributes to emotion processing lies in our approach to understanding of emotion in general. It is suggested that emotional processing depends on the synchronisation of different multisensory information (Schreuder et al., 2016). Furthermore, emotion processing comprises of different cognitive appraisals modules that occur in a loop (Scherer, 2009). So to explain the contribution of cognition in emotion processing, we must look them holistically as a system not by assessing the contribution of each cognitive module in isolation (Gray, 1990). Emotion should be considered as a complex system where changes may not occur linearly. For example, a study shows that as FTD progressing in later stages, emotion performance surprisingly getting better (Lavenu & Pasquier, 2005). A traditional statistical approach often failed to explain the discrepancy of the same emotional construct between clinical population and disease stages. To quote Liza Fieldmen Barrett (2017), in studying emotion, one should assume that variability is a new standard rather than as outliers to be explained after the fact.
This study proposed computational modelling of emotion to understand emotion processing and the contribution of cognitive functions in frontotemporal dementia. In general, I divide my study into two main approaches, first is a theory-driven approach, and the second is the data-driven approach.
Proposed Approaches
- Theory-driven model
A Theory-driven or model-based approach is theoretical models that use to give a better understanding of the complex system. A model-based can be used to identify the component of the system. In this case, a model-based approach can be used to identify the component of emotion system. Different major theory of emotion which specify different emotional component. Namely,
- Basic emotion model based on the idea that emotionally salient stimuli (event) will automatically trigger specific affect programme such as joy, fear etc. A distinct physiological reaction can be observed for each type of emotion. These patterns of emotional reactions are biologically endowed (Ekman, 1992).
- Constructivism does not agree with the idea that emotion is an innate or has been biologically reprogrammed. Instead, emotion is a learned concept of our bodily response (interoception) as they respond to the external world (exteroception). When the magnitude of the bodily sensation is big enough, our brain will actively construct a concept from our core emotion (feeling pleasant or unpleasant). This feeling particular emotion such as joy, sad, etc., are something that we learn from the sociocultural environment (Averill, 1980; Russell, 2003, Barrett, 2006).
- Appraisal theory is a combination of both basic emotion and constructivism. The appraisal theory defines more of active processing the incoming emotional event. Appraisal theory emphasises that cognitive process is part of the automatic appraisal. Assessment of emotional stimuli involving validation against individual motivation as it can be observed through action tendency, physiological appraisal of bodily changes which results in a distinct motoric expression. Unlike constructivism, appraisal theory clearly defines the emotion categorisation and its associated antecedents which come from the multilevel appraisal (Lazarus, 1991; Scherer, 2009).


(b)

(c )
Figure 1. Different theories of emotion specify emotional component differently. For example (a) basic emotion, (b) constructivism, and (d) appraisal theory (Scherer, 2009).
- Data-driven model
In contrast, Data-driven models are mathematical models or technique that has not theorised a priori and are only based on the existing data that used to fit the data that we have. In turn, this approach helps generate a pattern based on the nature of the data, being agnostic to any theoretical model.
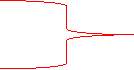
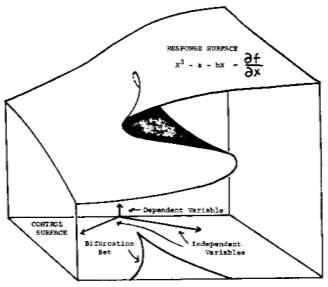
Response surface
Normal
Hysteresis
Impaired
Figure 3. Cusp catastrophe model (Zeeman, 1976)
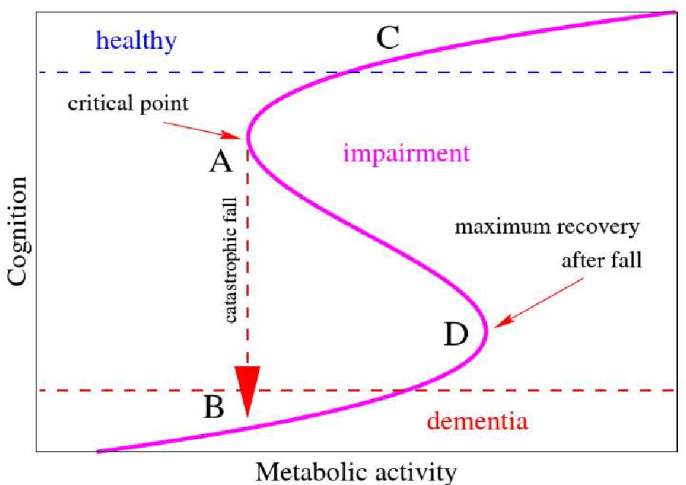
Catastrophe means a sudden change. Catastrophe model is used to explain a small continues change of variable controls that affect the dependent variable. In general, catastrophe model explains a critical coordinate where a small change is enough to cause a dramatic change in equilibrium. This small change is acting like a threshold that causes the whole system to behave differently than the normal condition (Zeeman, 1976).
Cusp catastrophe model is a good mathematical model to explain a continues phenomenon like behavioural changes in dementia. There are some properties of cusp model that make a good fit to explain this phenomenon (Flay, 1978). First is bimodality/bifurcation (e.g., normal vs abnormal behaviour). Second is abrupt change; at some point of progression rate, behavioural change is more apparent. Third is hysteresis; there is an inaccessible period.
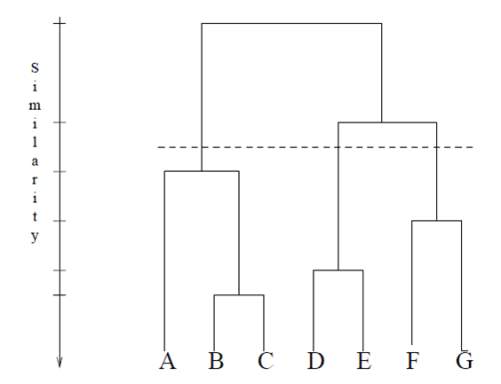
Figure 4. Hierarchical clustering model
Another data-driven model that often use in the clinical setting is clustering analysis. This technique advantages particularly for sorting cases, finding whether the case is heterogeneous or homogenous relative to certain predictors (Yim & Ramdeen, 2015).
Research Description
- Overview of Main Goals
In the first year, the primary objective is to review the relevant literature on the three main topics: (i) theories of emotion, and how this related to social cognition in general; (ii) frontotemporal dementia (FTD), and how this compared to healthy individuals; and (iii) computational modelling methods. Review in the theory of emotion is the theoretical approach and the biological basis of emotion. Understanding of FTD is acquired through meeting the patients and reviewing the previous studies arising from research conducted at FRONTIER as well as from reading the relevant papers. For computational modelling, the goal is to build technical skills such as advanced statistics, data mining, machine learning, and different types of modelling. Before developing a novel blueprint, existing model from various studies will be adapted.
- Aims and Hypothesis
- Which cognitive processes contribute to emotion processing? How do change in these modalities contribute to the reduced emotion recognition in FTD subtype
- Which physiological components contribute to emotion processing in different variants of FTD and how these compared to healthy individuals?
- Where does the proposed model of emotion stand with the traditional models of emotion? How do different models map emotion processing in FTD?
Progress Report
- Data Driven
| The contribution of emotion and cognition performance on clinical diagnostic | Hierarchical Clustering A
|
Hierarchical Clustering B
Including neuroimaging data
|
 The contributions of cognitive functions in emotion processing The contributions of cognitive functions in emotion processing |
Mathematical Models:
Cusp Catastrophe Model
|
|
Figure 5. The research roadmap of data-driven studies
Study 1
Summary of Study 1
- bvFTD is a clinical syndrome characterized by social cognition deficit, including in the ability to recognise emotion. Additionally, bvFTD patients also presented with cognitive problems. AD is the most prevalent type of dementia, on the other hand, have a hallmark of episodic memory impairment. However, AD patients also present the emotional problem. Healthy elders, likewise, often report a problem with emotion processing and cognitive function, which is due to healthy ageing.
- Having to share similar conditions, clinical diagnostic faces challenges in differentiating between bvFTD and AD at the early stage, and healthy ageing. Conventional statistical approaches rely on a priori, which often misses important features. Therefore, the more accurate diagnosis is needed to distinguish different clinical groups.
- A data-driven technique is an emerging multimodal approach. It has rarely been applied to map the diagnosis of bvFTD. A data-driven technique is agnostic to a theoretical assumption, hence present more objective measure of the phenomenon. Second, it allows us to explore different information that not has to rely on any theoretical assumption. So it gives us flexibility and a broader scope for exploring the phenomena.
- Based on the performance on the emotion and cognitive tests, can we differentiate them?
Aims
The study aims to see how emotion and cognitive process involved in different clinical diagnostic (bvFTD and AD) and compared to the healthy control.
1st Hypothesis: Clinical diagnosis of bvFTD, AD, and HC are distinguishable based on their performance on emotion and cognitive tests using a data-driven technique.
2nd Hypothesis: the interactions between emotion-cognition performance in clinical diagnosis have a hierarchical structure.
- To tests whether emotion and cognition are relevant for clinical diagnosis clustering, we conducted the clustering model separately before we include both tests. Sequentially:
- Cognitive performance only
- Emotion recognition performance only
- Both emotion and cognitive performance
Materials and Methods
Participants
The data that I use for the studies below were collected from FRONTIER research group from 2008 until 2017. For the first instance, I only use the data from the first visit of AD and bvFTD patients. Participants age is from 40 to 96 with an average of 70. The average of a total year of education is 12. 314 participants were recruited. 80 were AD, 121 were bvFTD, and 113 were healthy control; after excluding participants who did not complete or understood the tests. After only selecting cases with certain or definite diagnosis (for AD, bvFTD, and HC), it resulted in 199 participants (66 were AD, 25 were bvFTD, and 108 were HC). However, each cohort adjusted to the total bvFTD cases, so we have the same number of cases; resulting in 25 for each AD, bvFTD, and HC.
Neuropsychological assessment and Cognitive tests
These data comprise of the screening tests like Frontal Rating Scale (FRS) the Frontotemporal Dementia Functional Rating Scale (FTD-FRS; Mioshi, Hsieh, Savage, Hornberger, & Hodges, 2010) to get a general profile of the patients, such as the dementia scores, how well they function in everyday tasks, general social conduct, which important for the diagnosis.
Participants then administered with overall cognitive tests using Addenbrooke’s Cognitive Examination (ACE III; Hsieh, Mioshi, Schubert, Hoon, Hodges, 2013) to measure general cognitive function including semantic knowledge, episodic memory, and other tests to measure learning difficulties, and executive function. Sydney Language Battery (SYDBAT; Savage, Hsieh, Leslie, Foxe, Piguet, & Hodges, 2013) tests were also given to measure the participant’s cognitive ability in naming, repetition, comprehension, and semantic association.
Emotion recognition and matching tasks
Emotional battery tests: Emotion Selection Task and Face-Emotion Matching Task (Miller et al., 2012) administered to measure their ability to recognise emotion. For Emotion Selection Task, participants were asked to point different emotional face picture that matches the emotional label of different emotion category: neutral, happiness, surprise, disgust, anger, fear, sadness. While in the Face-Emotion Matching Tasks, series of emotional expression picture was presented to the participants and asked whether the same pictures are the same emotional expression. The Awareness of Social Inference Test (TASIT; McDonald, Flanagan, Rollins, & Kinch, 2003) was also given to measure different emotional constructs such as anxiety, disgust, revolted, anger, sadness, surprise, neutral, and happiness.
Analysis
R programming language is used (R Core Team, 2013). Hierarchical clustering is used as a data-driven technique to identify a cluster of cases. A feature selection was conducted to determine which cognitive tests and social cognition/ emotion tests were significantly associated with a diagnosis. Recommendation of important features were given based on boruta (Kursa & Rudnicki, 2010) and recursive feature eliminations (REF; Guyon, Weston, Barnhill & Vapnik, 2002) techniques: attention, memory, fluency, language, visuospatial of ACEIII; naming, comprehension, semantic of SYDBAT; happy, fear, angry, disgust, surprise, neutral of FAST; surprise, anxious, revolted of TASIT.
In this analysis, exploratory hierarchical cluster method with squared Euclidean distances as proximity measure with Ward’s method as the clustering algorithm. Visual observation was used to determine the main clusters. The number of the cluster then used as an input for k-means analysis. This will tell us whether the number of group in exploratory one can be confirmed.
Results
Here we test the homogeneous cluster based on three different models of tests. First model based on a cognitive test (ACE and functional analysis (FRS). The second model comprises only emotional tests (Face and Emotional Matching Tasks). The third model includes both cognitive and emotional tests following the rate of progression. Based on how well they map the homogeneous cases, the third model seems the best at predicting the diagnostic group.
This preliminary analysis showed that emotion tests or cognition tests only are not sufficient to describe the clinical diagnoses. However, using a combination of both tests performance generated better clustering into the more appropriate or accurate diagnostic. Based on the participant’s performance on cognitive and emotion recognition tests, we identified the three cluster of cases; bvFTD, AD, and healthy control.
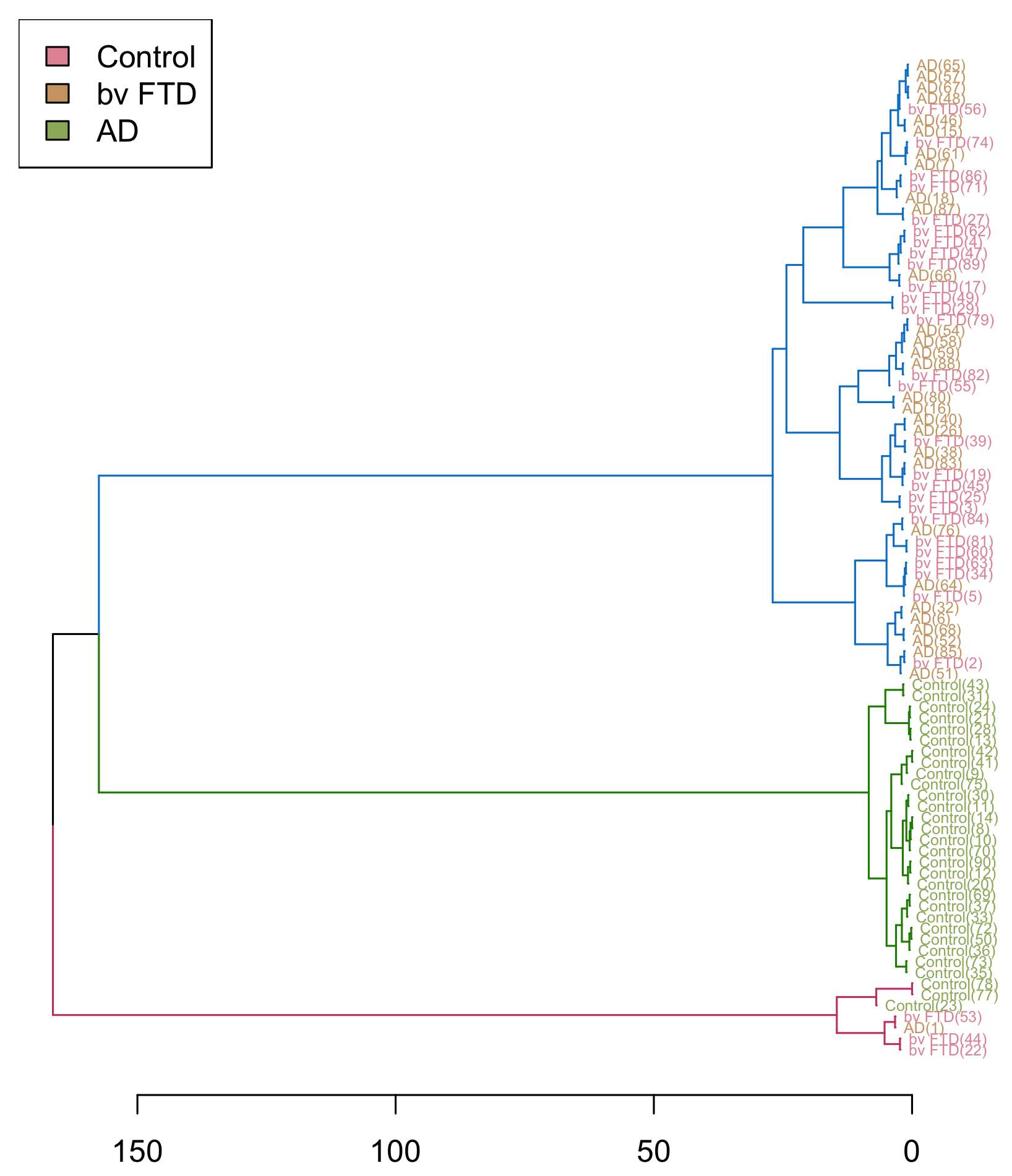
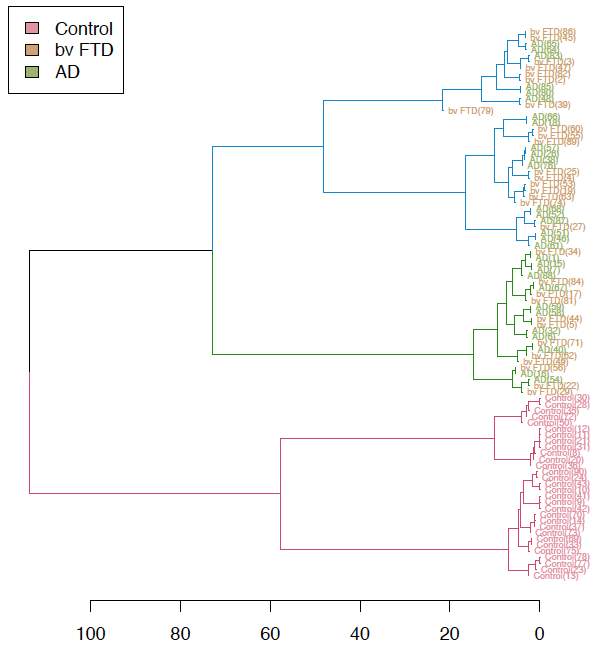
Figure 6. Hierarchical clustering of different diagnostic groups (bvFTD, AD, and control) using ACE and FRS tests. A dendrogram is used to represent the homogeneous cluster between bvFTD, AD, and control groups (n=30, N=90). Each branch in right hand represents a single case. Similar cases then merged using complete-linkage method calculating all element pairs. Less similar cases are further to the left.
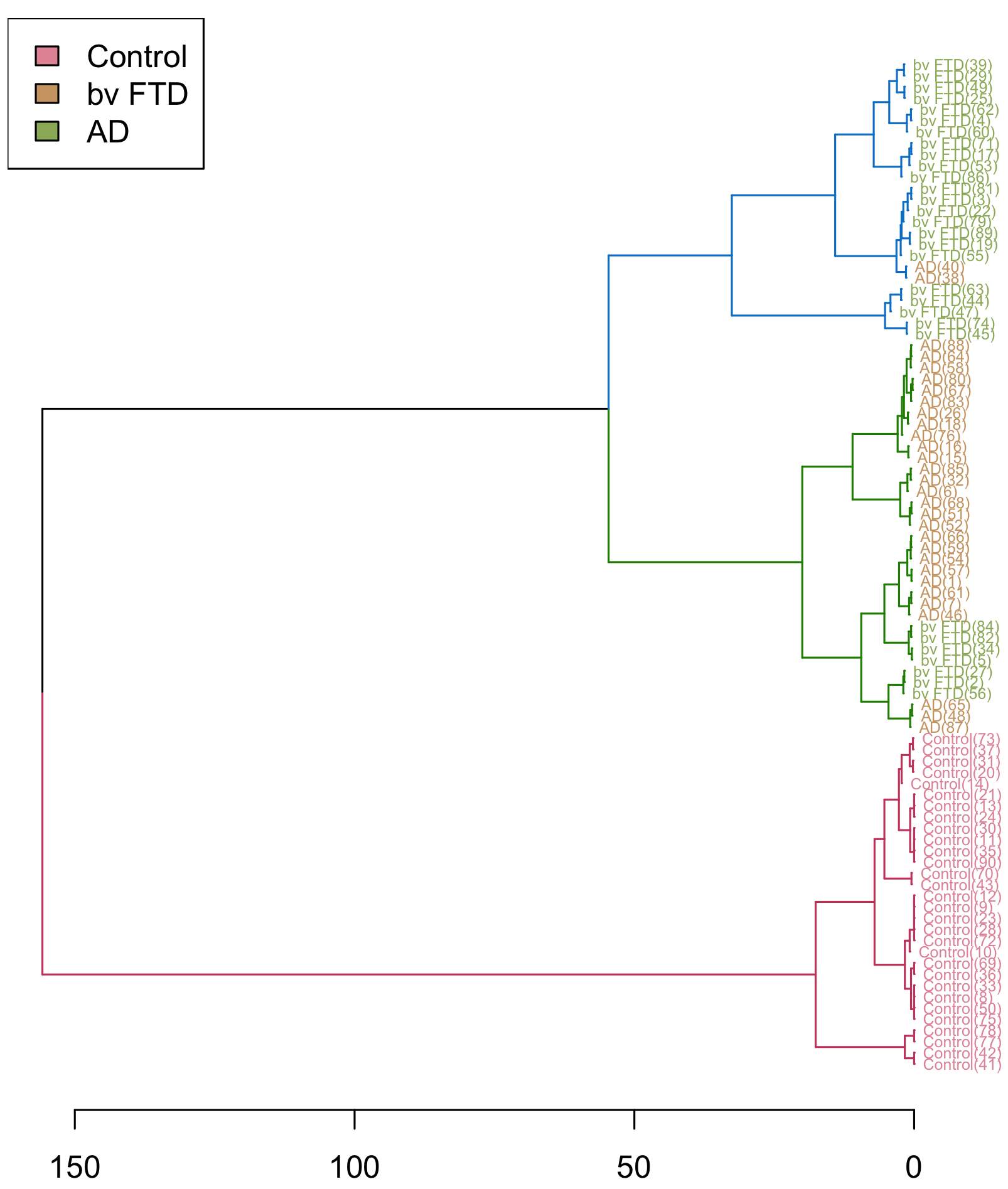
Figure 7. Hierarchical clustering of different diagnostic groups (bvFTD, AD, and control) using emotion tests only: Emotional Matching Tasks.
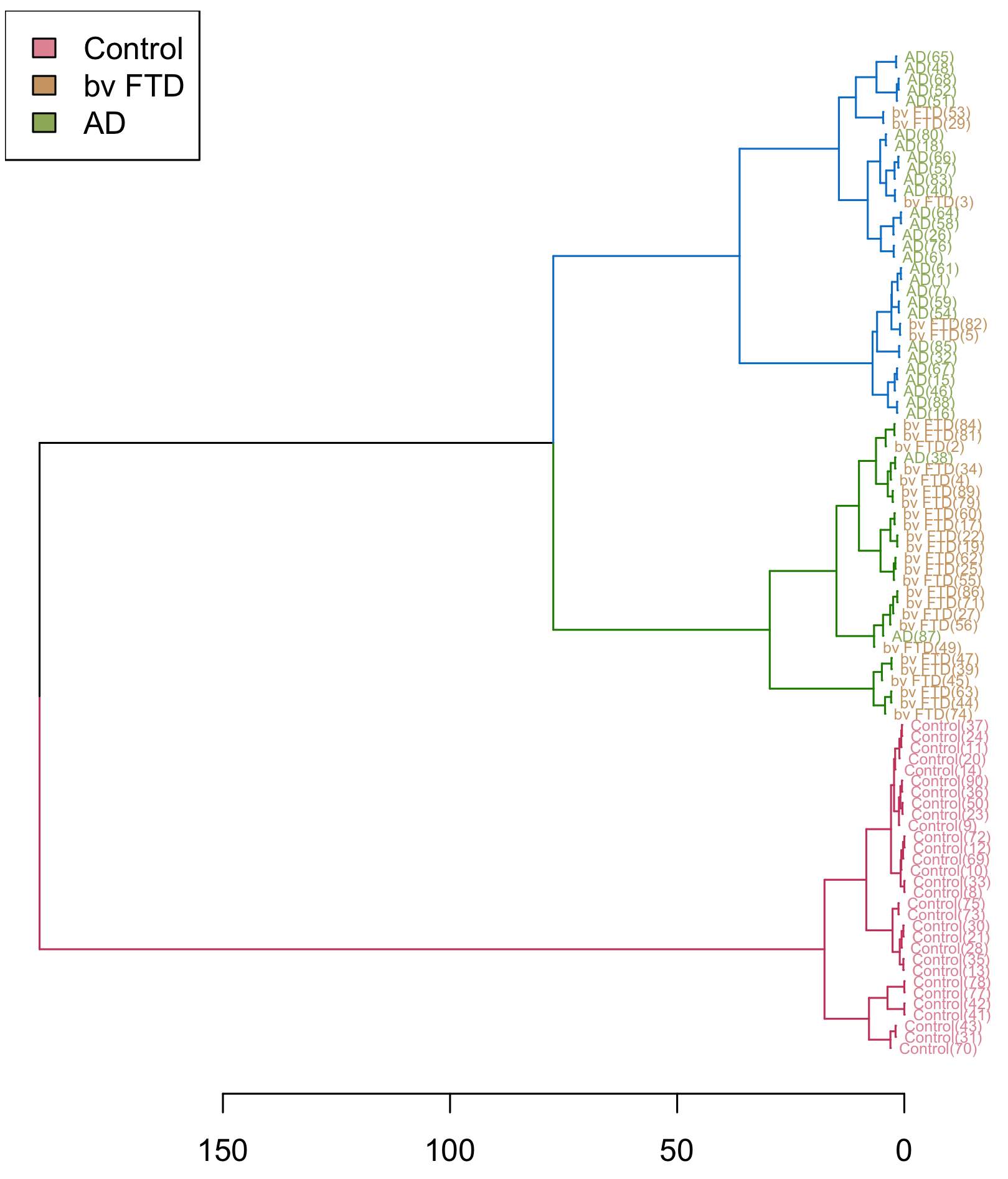
Figure 8. Hierarchical clustering of different diagnostic groups (bvFTD, AD, and control) using ACE, FRS, Emotional Matching Tasks.
Implication
- Both emotion and cognitive tests are important for characterising the clinical diagnostic between bvFTD, AD, and healthy elders.
- It illustrates that the effect of emotion may depend on the level of cognition or vice versa.
Recommendation
- Overlapping cases of bvFTD and AD based on the dendrogram indicates shared clinical syndromes. Further investigation will use neuroimaging data to find out the shared mechanisms of AD and FTD in emotion and cognitive performance.
Study 1.2
The hierarchical clustering study will include structural imaging data to enlighten us about the mechanism of emotion processing. Furthermore, it will inform us about the anatomical differences of each cluster. As noted from the result, some FTD cases are under the homogenous AD clusters, and vice versa. Understanding these variations might inform us about the overlapping mechanism of emotion processing in these particular group.
To measure differences between resultants cluster, I will deploy imaging statistical analysis. Multiple comparison will be conducted between the thickness of different brain regions.
Study 2
Background
- Although based on the works of literature that emotion performance is different between FTD and AD, some overlapping is observed based on the clustering.
Aims
- This study is aimed to understand each clustering of cases that generated using emotional test.
- 1st hypothesis: FTD cases that included in the same cluster as AD may share the same underlying cognitive problems, and vice versa.
- 2nd hypothesis: as disease progression, the configuration of a cluster will change as well as having different cognitive causes.
Analysis
- One-way analysis of variance will be conducted to evaluate differences between resultant clusters. This will also conduct separately to the data from the second and third visit. This will inform us how changes in clustering exist and the underlying causes. Different regression test will also conduct to estimate contributions of cognitive modalities to emotional performance in each cluster for each visit.
?
?
Figure 9. Hierarchical clustering of different diagnostic groups (bvFTD, AD, and control) using emotion tests only: Emotional Matching Tasks.
Study 3
Background
- We want to know how emotion impairment is relative to degradation of cognitive function following the severity rate of behavioural, temporal dementia.
- The linear model cannot explain these changes although clinical observation shows this trend.
- It is assumes using that emotion processing is steady even after the diagnosis until at some point or a threshold (of cognitive deterioration) is surpassed resulting in sudden emotional changes.
Aims
- To see how emotion processing problem in FTD and AD is predicted using catastrophe model.
- To see how emotion processing is affected by cognitive functions as the clinical stage progressing.
1st Hypothesis: The cusp catastrophe model can explain the emotional processing as predicted by dementia progression and cognitive decline.
Confirming the first hypothesis which lead us to the second hypothesis.
2nd Hypothesis: There is an intermediate non linear phenomenon in emotion processing that explains the variability of bvFTD.
Analysis
The cusp model will be compared to other models such as regression model. To see which one is better at explaining the data, I will test different information criteria such as Bayesian Information Criterion, Akaike Information Criterion, and pseudo-R-squared.
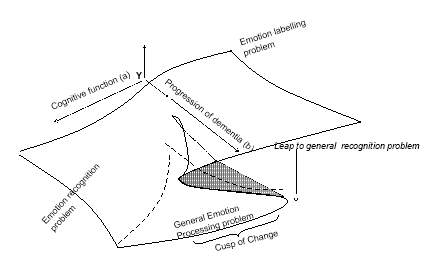
Figure 10. Proposed cusp catastrophe model to explain emotion processing impairment FTD
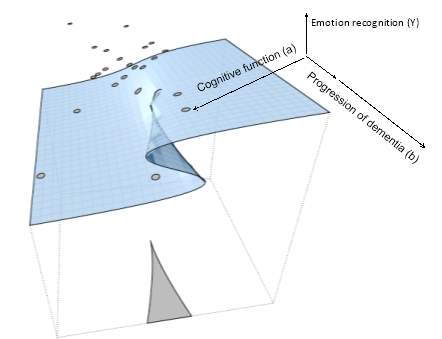
Figure 11. A cusp catastrophe modelling emotion, cognition, and dementia progression .
Emotion Processing
Cognition
Figure 12. An example of the expected result to explain the emotion processing problem in FTD following the cognitive decline. It highlights that in dementia cognitive decline must be low enough (to reach some threshold) to observe a beginning of the irreversible decline of emotion processing. This shows that the rate of the change of emotion processing changes following cognitive impairment does not happen incrementally or smoothly. There is a point where we cannot observe the changes, and suddenly there is an abrupt change to a very significant decline. This cusp catastrophe model is useful to point out non-linear changes where the intermediate process of dementia is not observed.
- Future Developments
The preliminary models have many weaknesses based on several considerations. First, the clustering of cases into three main clinical diagnoses still needs to be confirmed with brain imaging data to identify distinct brain atrophy. Second, clustering method will be tested on a different clinical variant of FTD, for example, bvFTD compared with SD or PNFA, to see how well they fit. Third, I need to compare these models using a confirmatory analysis (theory-based).
For the short-term goals. First, clustering the different test; to see which test that may relate closely to particular emotion (for example, negative emotion and inhibition test). Second, the result of this clustering then will be applied to cup catastrophe model to find out the threshold or juncture where there is a sudden change of emotion based on different cognitive tests. Third, to test the theory-based model using basic and advanced structural equation modelling.
- Estimated Work Plan for The Remaining Time
This section presents the task of the HDR tasks and PhD plans. Table 1 summarises the work carried out thus far. Table 2 provides the work plan for the remaining duration of the project.
During my first year, several practical skills had been achieved. I studied FTD through documentations (files, videos) and observing patients in Frontier labs. I learned R programming language which is important tool for statistics and computational modelling. I learned how to export and manage data using FileMaker. I learned how to manage my references using EndNote and manage my research using ENotebook. I also attended several advance statistical workshops including complex statistical modelling using MPlus programming language.
| HDR tasks and PhD Plans | Year/Month | ||||||||||||
| 2017 | 2018 | ||||||||||||
| 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 1 | 2 | 3 | |
| Literature review/ watching recordings | |||||||||||||
| First refinement of the PhD research topic | |||||||||||||
| HDR compulsory modules: health and safety, ethics | |||||||||||||
| Observing patients | |||||||||||||
| Learn about the FileMaker: database | |||||||||||||
| Attending Frontier lab meeting | |||||||||||||
| Attending seminars | |||||||||||||
| Attending colloquium | |||||||||||||
| Refinement of the research plan | |||||||||||||
| Review of the research plan | |||||||||||||
| Presenting the research plan | |||||||||||||
| Learn about a programming language: R | |||||||||||||
| Attending Mplus SEM workshop | |||||||||||||
| Exporting, cleaning, and impute missing data. | |||||||||||||
| Analysis of preliminary models | |||||||||||||
| Writing of the First Year Report | |||||||||||||
Table 1 – Completed Work-Plan Timetable
| HDR tasks and PhD Plans | Year/Month | ||||||||||||
| 2018 | 2019 | ||||||||||||
| 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 1 | 2 | 3 | |
| Attending colloquium | |||||||||||||
| Attending seminars at a symposium | |||||||||||||
| Refinement of the prototype for theory-based models. | |||||||||||||
| Refinement of the prototype for data-driven models. | |||||||||||||
| Attending workshops on computational modelling. | |||||||||||||
| Presenting in the Psychfest | |||||||||||||
| Testing the model with different clinical case/ populations | |||||||||||||
| Attending conferences | |||||||||||||
| Usability tests of the model for diagnosis | |||||||||||||
| Publication and dissemination of the work | |||||||||||||
| HDR tasks and PhD Plans | Year/Month | ||||||||||||
| 2019 | 2020 | ||||||||||||
| 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 1 | 2 | 3 | 4 | |
| Constructing emotional tests for a final usability test of the model. | |||||||||||||
| Attending colloquium | |||||||||||||
| Attending seminars in the symposium | |||||||||||||
| Presenting for the final year in the symposium | |||||||||||||
| Conducting experiments | |||||||||||||
| Analysis of the results | |||||||||||||
| Publication and dissemination of the work | |||||||||||||
| Attending conferences | |||||||||||||
| Writing the thesis | |||||||||||||
| Submitting the thesis | |||||||||||||
Table 2 – Estimated Work-Plan Timetable for the remaining time
References
Adolphs, R., Nummenmaa, L., Todorov, A., & Haxby, J. V. (2016). Data-driven approaches in the investigation of social perception. Phil. Trans. R. Soc. B, 371(1693), 20150367.
Anticevic, A., Schleifer, C., & Youngsun, T. C. (2015). Emotional and cognitive dysregulation in schizophrenia and depression: understanding common and distinct behavioral and neural mechanisms. Dialogues Clin Neurosci, 17(4), 421-434.
Averill, J. R. (1980). A constructivist view of emotion. In Theories of emotion (pp. 305-339).
Baez, S., Garcia, A. M., & Ibanez, A. (2017). The Social Context Network Model in Psychiatric and Neurological Diseases. Curr Top Behav Neurosci, 30, 379-396. doi:10.1007/7854_2016_443
Bandura, A. (2001). Social cognitive theory: An agentic perspective. Annual review of psychology, 52(1), 1-26.
Barrett, L. F. (2006). Are emotions natural kinds?. Perspectives on psychological science, 1(1), 28-58.
Barrett, L. F. (2017). Categories and their role in the science of emotion. Psychological inquiry, 28(1), 20-26.
Bathgate, D., Snowden, J. S., Varma, A., Blackshaw, A., & Neary, D. (2001). Behaviour in frontotemporal dementia, Alzheimer’s disease and vascular dementia. Acta neurológica scandinavica, 103(6), 367-378.’
Bediou, B., Ryff, I., Mercier, B., Milliery, M., Henaff, M. A., D’Amato, T., … & Krolak-Salmon, P. (2009). Impaired social cognition in mild Alzheimer disease. Journal of geriatric psychiatry and neurology, 22(2), 130-140.
Busemeyer, J., Townsend, J., Wang, Z., & Eidels, A. (2015). Review of Basic Mathematical Concepts Used in Computational and Mathematical Psychology. In The Oxford Handbook of Computational and Mathematical Psychology. : Oxford University Press.
Bosse, T., Pontier, M., & Treur, J. (2010). A computational model based on Gross’ emotion regulation theory. Cognitive systems research, 11(3), 211-230.
Chare, L., Hodges, J. R., Leyton, C. E., McGinley, C., Tan, R. H., Kril, J. J., & Halliday, G. M. (2014). New criteria for frontotemporal dementia syndromes: clinical and pathological diagnostic implications. J Neurol Neurosurg Psychiatry, 85(8), 865-870.
Dayan, P., & Williams, J. (2006). Putting the computation back into computational modeling. Pharmacopsychiatry, 39, S50-S51. doi:10.1055/s-2006-931494
Devinsky, O., Morrell, M. J., & Vogt, B. A. (1995). Contributions of anterior cingulate cortex to behaviour. Brain, 118(1), 279-306.
Devenney, E., Bartley, L., Hoon, C., O’Callaghan, C., Kumfor, F., Hornberger, M., … & Hodges, J. R. (2015). Progression in behavioral variant frontotemporal dementia: a longitudinal study. JAMA neurology, 72(12), 1501-1509.
Dickerson, B. C. (2016). Hodges’ Frontotemporal Dementia (Vol. 2nd). Cambridge: Cambridge University Press.
Duch, W. (2007). Computational Models of Dementia and Neurological Problems, 305–336. http://doi.org/10.1007/978-1-59745-520-6_17
Ekman, P. (1992). An argument for basic emotions. Cognition & emotion, 6(3-4), 169-200.
Eslinger, P. J., Moore, P., Troiani, V., Antani, S., Cross, K., Kwok, S., & Grossman, M. (2007). Oops! Resolving social dilemmas in frontotemporal dementia. Journal of Neurology, Neurosurgery & Psychiatry, 78(5), 457-460.
Ekman, P. (2016). What scientists who study emotion agree about. Perspectives on Psychological Science, 11(1), 31-34
Flay, B. R. (1978). Catastrophe theory in social psychology: Some applications to attitudes and social behavior. Systems Research and Behavioral Science, 23(4), 335-350.
Fodor, J. A. (2001). The mind doesn’t work that way: The scope and limits of computational psychology. MIT press.
Frith, C. D., & Frith, U. (2008). Implicit and explicit processes in social cognition. Neuron, 60(3), 503-510. doi:10.1016/j.neuron.2008.10.032
Frith, C. (2009). Role of facial expressions in social interactions. Philosophical Transactions of the Royal Society B: Biological Sciences, 364(1535), 3453-3458.
Gorno‐Tempini, M. L., Dronkers, N. F., Rankin, K. P., Ogar, J. M., Phengrasamy, L., Rosen, H. J., … & Miller, B. L. (2004). Cognition and anatomy in three variants of primary progressive aphasia. Annals of neurology, 55(3), 335-346.
Gorno-Tempini, M. L., Hillis, A. E., Weintraub, S., Kertesz, A., Mendez, M., Cappa, S. E. E. A., … & Manes, F. (2011). Classification of primary progressive aphasia and its variants. Neurology, 76(11), 1006-1014.
Gray, J. A. (1990). Brain systems that mediate both emotion and cognition. Cognition & emotion, 4(3), 269-288.
Grossman, M., Mickanin, J., Onishi, K., Hughes, E., D’Esposito, M., Ding, X. S., … & Reivich, M. (1996). Progressive nonfluent aphasia: language, cognitive, and PET measures contrasted with probable Alzheimer’s disease. Journal of Cognitive Neuroscience, 8(2), 135-154.
Gossink, F., Schouws, S., Krudop, W., Scheltens, P., Stek, M., Pijnenburg, Y., & Dols, A. (2018). Social Cognition Differentiates Behavioral Variant Frontotemporal Dementia From Other Neurodegenerative Diseases and Psychiatric Disorders. The American Journal of Geriatric Psychiatry, 26(5), 569-579.
Guyon, I., Weston, J., Barnhill, S., & Vapnik, V. (2002). Gene selection for cancer classification using support vector machines. Machine learning, 46(1-3), 389-422.
Herland, M., Khoshgoftaar, T. M., & Wald, R. (2014). A review of data mining using big data in health informatics. Journal of Big Data, 1(1), 2.
Herrmann, E., Call, J., Hernández-Lloreda, M. V., Hare, B., & Tomasello, M. (2007). Humans have evolved specialized skills of social cognition: The cultural intelligence hypothesis. science, 317(5843), 1360-1366.
Hodges, J. R., Patterson, K., Oxbury, S., & Funnell, E. (1992). Semantic dementia: Progressive fluent aphasia with temporal lobe atrophy. Brain, 115(6), 1783-1806.
Hodges, J. R., Davies, R. R., Xuereb, J. H., Casey, B., Broe, M., Bak, T. H., … & Halliday, G. M. (2004). Clinicopathological correlates in frontotemporal dementia. Annals of neurology, 56(3), 399-406.
Hornberger, M., Piguet, O., Graham, A. J., Nestor, P. J., & Hodges, J. R. (2010). How preserved is episodic memory in behavioral variant frontotemporal dementia?. Neurology, 74(6), 472-479.
Hsieh, S., Schubert, S., Hoon, C., Mioshi, E., & Hodges, J. R. (2013). Validation of the Addenbrooke’s Cognitive Examination III in frontotemporal dementia and Alzheimer’s disease. Dementia and geriatric cognitive disorders, 36(3-4), 242-250.
Hudlicka, E. (2011). Guidelines for designing computational models of emotions. International Journal of Synthetic Emotions (IJSE), 2(1), 26-79.
Hudlicka, E. (2014). From habits to standards: towards systematic design of emotion models and affective architectures. In Emotion Modeling (pp. 3-23). Springer, Cham.
Hutchings, R., Palermo, R., Piguet, O., & Kumfor, F. (2017). Disrupted face processing in frontotemporal dementia: a review of the clinical and neuroanatomical evidence. Neuropsychology review, 27(1), 18-30.
Ibañez, A., & Manes, F. (2012). Contextual social cognition and the behavioral variant of frontotemporal dementia. Neurology, 78(17), 1354-1362.
Iturria-Medina, Y., & Evans, A. C. (2015). On the central role of brain connectivity in neurodegenerative disease progression. Front Aging Neurosci, 7. doi:10.3389/fnagi.2015.00090
James, T. T., Jerome, R. B., Jerome, R. B., James, T. T., Zheng, W., & Ami, E. (2015). Review of Basic Mathematical Concepts Used in Computational and Mathematical Psychology.
Knibb, J. A., Xuereb, J. H., Patterson, K., & Hodges, J. R. (2006). Clinical and pathological characterization of progressive aphasia. Annals of neurology, 59(1), 156-165.
Knapp, M. L., Hall, J. A., & Horgan, T. G. (2013). Nonverbal communication in human interaction. Cengage Learning
Körding, K. P., Beierholm, U., Ma, W. J., Quartz, S., Tenenbaum, J. B., & Shams, L. (2007). Causal Inference in Multisensory Perception. PLoS One, 2(9). doi:10.1371/journal.pone.0000943
Kamminga, J., Kumfor, F., Burrell, J. R., Piguet, O., Hodges, J. R., & Irish, M. (2014). Differentiating between right-lateralised semantic dementia and behavioural-variant frontotemporal dementia: an examination of clinical characteristics and emotion processing. J Neurol Neurosurg Psychiatry, jnnp-2014.
Kumfor, F., & Piguet, O. (2012). Disturbance of emotion processing in frontotemporal dementia: a synthesis of cognitive and neuroimaging findings. Neuropsychol Rev, 22(3), 280-297. doi:10.1007/s11065-012-9201-6
Kumfor, F., Landin-Romero, R., Devenney, E., Hutchings, R., Grasso, R., Hodges, J. R., & Piguet, O. (2016). On the right side? A longitudinal study of left- versus right lateralized semantic dementia. Brain, 139(Pt 3), 986-998. doi:10.1093/brain/awv387
Kursa, M. B., & Rudnicki, W. R. (2010). Feature selection with the Boruta package. J Stat Softw, 36(11), 1-13.
Lam, B. Y., Halliday, G. M., Irish, M., Hodges, J. R., & Piguet, O. (2014). Longitudinal white matter changes in frontotemporal dementia subtypes. Human brain mapping, 35(7), 3547-3557.
Lavenu, I., Pasquier, F., Lebert, F., Petit, H., & Van der Linden, M. (1999). Perception of emotion in frontotemporal dementia and Alzheimer disease. Alzheimer disease and associated disorders.
Lavenu, I., & Pasquier, F. (2005). Perception of emotion on faces in frontotemporal dementia and Alzheimer’s disease: a longitudinal study. Dementia and geriatric cognitive disorders, 19(1), 37-41.
Lazarus, R. S. (1991). Progress on a cognitive-motivational-relational theory of emotion. American psychologist, 46(8), 819.
Lindau, M., Almkvist, O., Kushi, J., Boone, K., Johansson, S. E., Wahlund, L. O., … & Miller, B. L. (2000). First symptoms–frontotemporal dementia versus Alzheimer’s disease. Dementia and geriatric cognitive disorders, 11(5), 286-293.
Lu, P. H., Mendez, M. F., Lee, G. J., Leow, A. D., Lee, H.-W., Shapira, J., … Knopman, D. S. (2013). Patterns of Brain Atrophy in Clinical Variants of Frontotemporal Lobar Degeneration. Dementia and Geriatric Cognitive Disorders, 35(1-2), 34–50. http://doi.org/10.1159/000345523
Mackenzie, I. R., Neumann, M., Bigio, E. H., Cairns, N. J., Alafuzoff, I., Kril, J., … & Ince, P. G. (2010). Nomenclature and nosology for neuropathologic subtypes of frontotemporal lobar degeneration: an update. Acta neuropathologica, 119(1), 1.
Mackenzie, I. R., Neumann, M., Baborie, A., Sampathu, D. M., Du Plessis, D., Jaros, E., … & Lee, V. M. (2011). A harmonized classification system for FTLD-TDP pathology. Acta neuropathologica, 122(1), 111-113.
Marsella, S., Gratch, J., & Petta, P. (2010). Computational models of emotion. A Blueprint for Affective Computing-A sourcebook and manual, 11(1), 21-46.
McDonald, S., Bornhofen, C., Shum, D., Long, E., Saunders, C., & Neulinger, K. (2006). Reliability and validity of The Awareness of Social Inference Test (TASIT): a clinical test of social perception. Disability and rehabilitation, 28(24), 1529-1542.
Mendez, M. F., Joshi, A., Tassniyom, K., Teng, E., & Shapira, J. S. (2013). Clinicopathologic differences among patients with behavioral variant frontotemporal dementia. Neurology, 80(6), 561-568.
Mesulam, M. M. (2001). Primary progressive aphasia. Ann Neurol, 49(4), 425-432.
Miller, L. A., Hsieh, S., Lah, S., Savage, S., Hodges, J. R., & Piguet, O. (2012). One size does not fit all: face emotion processing impairments in semantic dementia, behavioural-variant frontotemporal dementia and Alzheimer’s disease are mediated by distinct cognitive deficits. Behavioural neurology, 25(1), 53-60.
Mioshi, E., Hsieh, S., Savage, S., Hornberger, M., & Hodges, J. R. (2010). Clinical staging and disease progression in frontotemporal dementia. Neurology, 74(20), 1591-1597.
Neary, D., Snowden, J. S., Gustafson, L., Passant, U., Stuss, D., Black, S. A., … & Boone, K. (1998). Frontotemporal lobar degeneration A consensus on clinical diagnostic criteria. Neurology, 51(6), 1546-1554.
Neumann, M., Sampathu, D. M., Kwong, L. K., Truax, A. C., Micsenyi, M. C., Chou, T. T., … & McCluskey, L. F. (2006). Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Science, 314(5796), 130-133.
Neumann, M., Rademakers, R., Roeber, S., Baker, M., Kretzschmar, H. A., & Mackenzie, I. R. (2009). A new subtype of frontotemporal lobar degeneration with FUS pathology. Brain, 132(11), 2922-2931.
Pennington, C., Hodges, J. R., & Hornberger, M. (2011). Neural correlates of episodic memory in behavioral variant frontotemporal dementia. Journal of Alzheimer’s Disease, 24(2), 261-268.
Piguet, O., Hornberger, M., Mioshi, E., & Hodges, J. R. (2011). Behavioural-variant frontotemporal dementia: diagnosis, clinical staging, and management. Lancet Neurol, 10(2), 162-172. doi:10.1016/S1474-4422(10)70299-4
Poorkaj, P., Bird, T. D., Wijsman, E., Nemens, E., Garruto, R. M., Anderson, L., … & Schellenberg, G. D. (1998). Tau is a candidate gene for chromosome 17 frontotemporal dementia. Annals of neurology, 43(6), 815-825.
Rascovsky, K., Hodges, J. R., Knopman, D., Mendez, M. F., Kramer, J. H., Neuhaus, J., … & Hillis, A. E. (2011). Sensitivity of revised diagnostic criteria for the behavioural variant of frontotemporal dementia. Brain, 134(9), 2456-2477.
Reisenzein, R., Hudlicka, E., Dastani, M., Gratch, J., Hindriks, K., Lorini, E., & Meyer, J. J. C. (2013). Computational modeling of emotion: Toward improving the inter-and intradisciplinary exchange. IEEE Transactions on Affective Computing, 4(3), 246-266.
Rodríguez, L. F., & Ramos, F. (2015). Computational models of emotions for autonomous agents: major challenges. Artificial Intelligence Review, 43(3), 437-465.
Rohrer, J. D., Sauter, D., Scott, S., Rossor, M. N., & Warren, J. D. (2012). Receptive prosody in nonfluent primary progressive aphasias. cortex, 48(3), 308-316.
Rosen, H. J., Gorno–Tempini, M. L., Goldman, W. P., Perry, R. J., Schuff, N., Weiner, M., … & Miller, B. L. (2002). Patterns of brain atrophy in frontotemporal dementia and semantic dementia. Neurology, 58(2), 198-208.
R Core Team (2013). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL http://www.R-project.org
Russell, J. A. (2003). Core affect and the psychological construction of emotion. Psychological Review, 110(1), 145-172.
Savage, S., Hsieh, S., Leslie, F., Foxe, D., Piguet, O., & Hodges, J. R. (2013). Distinguishing subtypes in primary progressive aphasia: application of the Sydney language battery. Dementia and geriatric cognitive disorders, 35(3-4), 208-218.
Seeley, W. W., Crawford, R., Rascovsky, K., Kramer, J. H., Weiner, M., Miller, B. L., & Gorno-Tempini, M. L. (2008). Frontal paralimbic network atrophy in very mild behavioral variant frontotemporal dementia. Archives of neurology, 65(2), 249-255.
Scherer, K. R., Bänziger, T., & Roesch, E. (Eds.). (2010). A Blueprint for Affective Computing: A sourcebook and manual. Oxford University Press.
Scherer, K. R. (2009). Emotions are emergent processes: they require a dynamic computational architecture. Philosophical Transactions of the Royal Society of London B: Biological Sciences, 364(1535), 3459-3474.
Scherling, C. S., Zakrzewski, J., Datta, S., Levenson, R. W., Shimamura, A. P., Sturm, V. E., … & Rosen, H. J. (2017). Mistakes, too few to mention? Impaired self-conscious emotional processing of errors in the behavioral variant of frontotemporal dementia. Frontiers in behavioral neuroscience, 11, 189.
Schreuder, E., van Erp, J., Toet, A., & Kallen, V. L. (2016). Emotional responses to Multisensory Environmental Stimuli: A conceptual framework and literature review. SAGE Open, 6(1), 2158244016630591.
Snowden, J. S., Goulding, P. J., & Neary, D. (1989). Semantic dementia: a form of circumscribed cerebral atrophy. Behavioural Neurology.
Snowden, J. S., Bathgate, D., Varma, A., Blackshaw, A., Gibbons, Z. C., & Neary, D. (2001). Distinct behavioural profiles in frontotemporal dementia and semantic dementia. J Neurol Neurosurg Psychiatry, 70(3), 323-332.
Snowden, J. S., Gibbons, Z. C., Blackshaw, A., Doubleday, E., Thompson, J., Craufurd, D., … & Neary, D. (2003). Social cognition in frontotemporal dementia and Huntington’s disease. sNeuropsychologia, 41(6), 688-701.
Snowden, J. S., Harris, J. M., Thompson, J. C., Kobylecki, C., Jones, M., Richardson, A. M., & Neary, D. (2017). Semantic dementia and the left and right temporal lobes. Cortex.
Sprong, M., Schothorst, P., Vos, E., Hox, J., & Van Engeland, H. (2007). Theory of mind in schizophrenia. The British Journal of Psychiatry, 191(1), 5-13.
Sun, R. (Ed.). (2008). The Cambridge handbook of computational psychology. Cambridge University Press.
Tariot, P. N., Mack, J. L., Patterson, M. B., & Edland, S. D. (1995). The behavior rating scale for dementia of the consortium to establish a registry for Alzheimer’s disease. The American journal of psychiatry, 152(9), 1349.
Teufel, C., & Fletcher, P. C. (2016). The promises and pitfalls of applying computational models to neurological and psychiatric disorders. Brain, 139(10), 2600-2608. doi:10.1093/brain/aww209
Turner, R. S., Kenyon, L. C., Trojanowski, J. Q., Gonatas, N., & Grossman, M. (1996). Clinical, neuroimaging, and pathologic features of progressive nonfluent aphasia. Annals of neurology, 39(2), 166-173.
Vallacher, R. R., Read, S. J., & Nowak, A. (Eds.). (2017). Computational Social Psychology. Routledge.
Von Der Heide, R. J., Skipper, L. M., & Olson, I. R. (2013). Anterior temporal face patches: a meta-analysis and empirical study. Front Hum Neurosci, 7, 17.
Whitwell, J. L., Boeve, B. F., Weigand, S. D., Senjem, M. L., Gunter, J. L., Baker, M. C., … & Rademakers, R. (2015). Brain atrophy over time in genetic and sporadic frontotemporal dementia: a study of 198 serial magnetic resonance images. European journal of neurology, 22(5), 745-752.
Yim, O., & Ramdeen, K. T. (2015). Hierarchical cluster analysis: comparison of three linkage measures and application to psychological data. The quantitative methods for psychology, 11(1), 8-21.
Zeeman, E. C. (1976). Catastrophe theory. Scientific American, 234(4), 65-83.
Zhang, Y., Tartaglia, M. C., Schuff, N., Chiang, G. C., Ching, C., Rosen, H. J., … & Weiner, M. W. (2013). MRI signatures of brain macrostructural atrophy and microstructural degradation in frontotemporal lobar degeneration subtypes. Journal of Alzheimer’s Disease, 33(2), 431-444.
Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allRelated Content
All TagsContent relating to: "Alzheimers"
Alzheimer’s disease is a neurodegenerative disease that causes a progressive decline in brain function, affecting memory, language, orientation, and reasoning. Symptoms usually begin mild and get progressively worse.
Related Articles
DMCA / Removal Request
If you are the original writer of this dissertation and no longer wish to have your work published on the UKDiss.com website then please:





