Threats to the Beluga Whale (Delphinapterus leucas)
Info: 10745 words (43 pages) Dissertation
Published: 10th Dec 2019
Beluga Whale (Delphinapterus leucas)
Abstract
Beluga whales (Delphinapterus leucas), also known as white whales, are migratory marine mammals that are distributed across Arctic and Subarctic waters around Canada, Russia and Alaska (Hauser et al., 2014). They breed during the summer months in the north and migrate south to spend the winter months foraging (Hauser, et al., 2014). Beluga whales live in pods, which consist of two to twenty-five individuals and rarely have more than one calf at a time (Hill et al., 2015). They are a very social species and use at least forty different types of acoustic signals to communicate with conspecifics (Alekseeva et al., 2013). Warmer sea surface temperatures, hazardous waste runoff, and increased vessel traffic are some examples of major threats to beluga whale populations (Norman et al., 2015). Based on the International Union for Conservation of Nature (IUCN) criteria, beluga whales are considered to be a near threatened species (Jefferson et al., 2012). Recent research has found that there are two genetically distinct lineages of beluga whales, which carries important implications for conservation efforts (Gladden et al., 1999).
Systematics
Beluga whales (Delphinapterus leucus) are members of the order Cetacea, which they share with dolphins, porpoises and other whales (Harrington, 2008). Within this order are two suborders: Mysticeti (i.e. baleen whales) and Odontoceti (i.e. toothed whales including belugas; Harington, 2008; Milinkovitch et al., 1994). There is some uncertainty about which species is the sister group of all whales, but tentatively, it is the beaked whales (Milinkovitch et al., 1994).
Beluga whales can be further classified into the monophyletic superfamily called Delphinoidea with porpoises and dolphins (Milinkovitch et al., 1994; Waddell et al., 2000). There is confusion regarding whether porpoises are more closely related to beluga whales or to dolphins (Milinkovitch et al., 1994; Waddell et al., 2000). Previously, there were speculations that porpoises were more closely related to dolphins (Milinkovitch et al., 1994). However, recent molecular data provides support for a clade consisting of porpoises and beluga whales (Waddell et al., 2000).
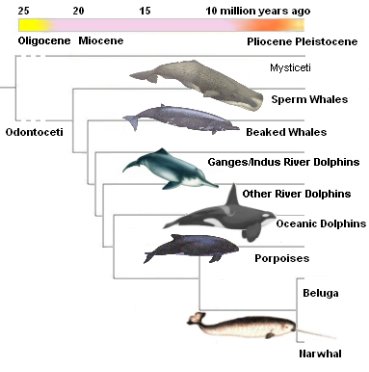 The monophyletic family, Monodontidae, includes two marine mammals: beluga whales and narwhals, and the common ancestor of this family is the Denebola (Harington, 2008; Fig. 1.). In the past, taxonomists believed that beluga whales were closely related to both the Irrawaddy dolphins and the narwhals but this has been revised (O’Corry-Crowe, 2009). Therefore, presently, narwhals are the most closely related species to beluga whales (O’Corry-Crowe, 2009).
The monophyletic family, Monodontidae, includes two marine mammals: beluga whales and narwhals, and the common ancestor of this family is the Denebola (Harington, 2008; Fig. 1.). In the past, taxonomists believed that beluga whales were closely related to both the Irrawaddy dolphins and the narwhals but this has been revised (O’Corry-Crowe, 2009). Therefore, presently, narwhals are the most closely related species to beluga whales (O’Corry-Crowe, 2009).
Monodontidae
Fig. 1. Phylogeny of beluga whales and their closest relatives. The family Monodontidae consists of beluga whales and narwhals (Morbey, T. (Photographer). Phylogenetic tree of the odontocetes. [Digital imgae]. Retrieved from http://palaeo.gly.bris.ac.uk/palaeofiles/whales/odontoceti.htm)
Nomenclature
Kingdom: Animalia
Phylum: Chordata
Class: Mammalia
Order: Cetacea
Suborder: Odontoceti
Family: Monodontidae
Genus: Delphinapterus
Species: leucas
Fig. 2. The taxonomy of the beluga whale (IUCN Red List, 2017)
Throughout the literature, the beluga whale has always been referred to as the Delphinapterus leucas (Fig.2.), and there does not appear to be evidence of synonymy or misuse of the name. The beluga whale is known by several common names such as beluga whale, white whale and Belukha (IUCN Red list,2017). The name beluga comes from the Russian word for white (O’Corry-Crowe, 2009). The beluga whale has also been nicknamed the “sea canary” due to its ability to produce a wide range of calls (O’Corry-Crowe, 2009).
Distinguishing Characteristics
The beluga whale is one of the most distinct looking marine mammals due to their white colouration (Harington, 2008; O’Corry-Crowe, 2009). Movement in the beluga whale’s melon-shaped head and mouth allow them to display different facial expressions (Harington, 2008; O’Corry-Crowe, 2009). They are slightly smaller than their closest and only relative in the Monodontidae family, the narwhal (Macdonald, 2007; O’Corry-Crowe, 2009; Fig. 3.).
Unlike the narwhals that have two nonfunctional teeth, beluga whales have up to 40 teeth, none of which are tusks (Macdonald, 2007; O’Corry-Crowe, 2009). Another distinct characteristic of beluga whales is their ability to move their head and neck laterally (Harrington, 2008;O’Corry-Crowe, 2009). This distinguishes them from narwhals that are incapable of performing this movement (Macdonald, 2007).

Fig. 3. Morphological comparison between the beluga whale and the narwhal. (Toni Llobet & Lynx Edicions. (n.d.). [Digital Image]. Retrieved from http://animalia-life.club/openphoto.php?img=http://www.tonillobet.com/wp-content/uploads/2013/12/tonillobet_wildlifeart_HMW4_beluga_narwhal.jpg)
General Description
Belugas are a whale of medium size and build and can reach a maximum length of about 16 feet and a maximum weight of 1500 kg (Leatherwood et al., 1976; O’Corry-Crowe, 2009). Size and body mass varies depending on age and sex, with male belugas tending to be larger (Leatherwood et al., 1976). In comparison to females, males can grow to be 25% longer and their body structure appears to be more robust due to their larger size (O’Corry-Crowe, 2009). The flippers of male belugas also have an upward recurve which sets them apart from the females (Harington, 2008). Relative to the rest of their body, a beluga whale’s head, flippers and tail appear to be small (Leatherwood et al., 1976; O’Corry-Crowe, 2009).
Beluga whales do not have a dorsal fin but they do possess a dorsal ridge/crest on their back which can be 1 to 3 cm tall and 50 cm long and is important for breaking sea ice (Harington, 2008; O’Corry-Crowe, 2009). This ridge is notched laterally forming small bumps (Leatherwood et al., 1976). They have a muscular body that turns into a distinct neck before reaching the head (Leatherwood et al., 1976). Their cervical vertebrae are unfused which allows them to move their head and neck from side to side (Harrington, 2008; O’Corry-Crowe, 2009).
At birth, beluga calves are originally greyish cream in colour (O’Corry-Crowe, 2009). They then change their colour almost immediately to either a blue-grey or a dark-brown (O’Corry-Crowe, 2009). They begin to lighten at the age of 6 or 7 (females and males respectively) and do not turn fully white until they are 14 or 18 (females and males respectively) (Brodie, 1971; Harington, 2008; Leatherwood et al., 1976; O’Corry-Crowe, 2009).
A large part of their teeth are made of cementum (Stock et al., 2017). Cementum is mineralized tissue consisting mostly of apatite minerals and collagen (Stock et al., 2017). Annular growth bands or growth layer groups form within the cementum and are used to identify the age of the animal (Stock et al., 2017). These bands can be light or dark in colour (Stock et al., 2017). Furthermore, they have dentine layers incorporated every year which can also be used to determine their age (Sergeant 1973; Stewart et al., 2006).
Distribution and Habitat
Beluga whales are found in the Arctic and Subarctic waters of Canada, Alaska, and Russia (Hauser et al., 2014; Fig. 4.). There are both migratory and non-migratory beluga whales (Quakenbush et al., 2015). Migratory beluga whales travel to North Arctic estuaries or other shallow waters for summer breeding (June-October) (Hauser et al., 2014). During the fall and winter months (October-June), they disperse southwards to deeper waters with sea ice cover (Hauser et al., 2014). It is thought that there are five main sites that beluga whales migrate to for the winter: the Bering Sea, Hudson Strait, Baffin Bay, Cook Inlet, and St. Lawrence River (Gladden et al., 1999). Several different summer populations share the same wintering sites (MacLaren & Davis, 1982), but do not often occupy them at the same time (O’Corry Crowe et al., 1997). Non-migratory populations of beluga whales reside in the Cook Inlet and St. Lawrence River regions (Gladden et al., 1999). As a result, they are more geographically isolated from the other wintering populations (Gladden et al., 1999).
It is difficult to track all the summer sites that beluga whales inhabit, but some of the regions include the Hudson Bay (East and West), Ungava Bay, Beaufort Sea, Chukchi Sea, Baffin Bay, St. Lawrence River, Norton Sound, and Bristol Bay (Smith & Hammill, 1986). Researchers have investigated the beluga whale populations of the Eastern Chukchi and Beaufort Seas during the summer months (Hauser et al., 2014). In fact, the largest population of summer beluga whales has been found in the Beaufort Sea (Hauser et al., 2014; Lesoto et al., 2006). Beluga whales have also been found to disperse along the southern estuaries of the Hudson Bay among the Churchill, Nelson, and Seal Rivers (Smith et al., 2017). Shallow coastal waters are preferable during breeding season because it is safer for breeding and easier to protect themselves from predators when necessary (Smith et al., 2017). Beluga whales are also more densely concentrated in waters with greater freshwater influx during the breeding season, which may correspond with the seasonal moult they conduct for that year (Smith et al., 2017).
There is also a gender difference in habitat distribution for beluga whales. Female distributions are generally more condensed than those of males, where males disperse greater distances to deeper waters from their summer habitat (Hauser et al., 2014; Hauser et al., 2017). Researchers suggest this is because males have more energy stores, thus being adapted to dive for longer periods of time (Hauser et al., 2017). The distribution of female beluga whales is also more dependent on their proximity to the shoreline, whereas male distribution is more dependent on their proximity to the ice edge (Hauser et al., 2017). Research on Western Hudson Bay beluga whales found that their water habitat preference varies with wet and dry seasons (Smith et al., 2017). During the wet season, they prefer deeper, saline water and during dry seasons, their preference shifts towards shoreline waters (Smith et al., 2017).
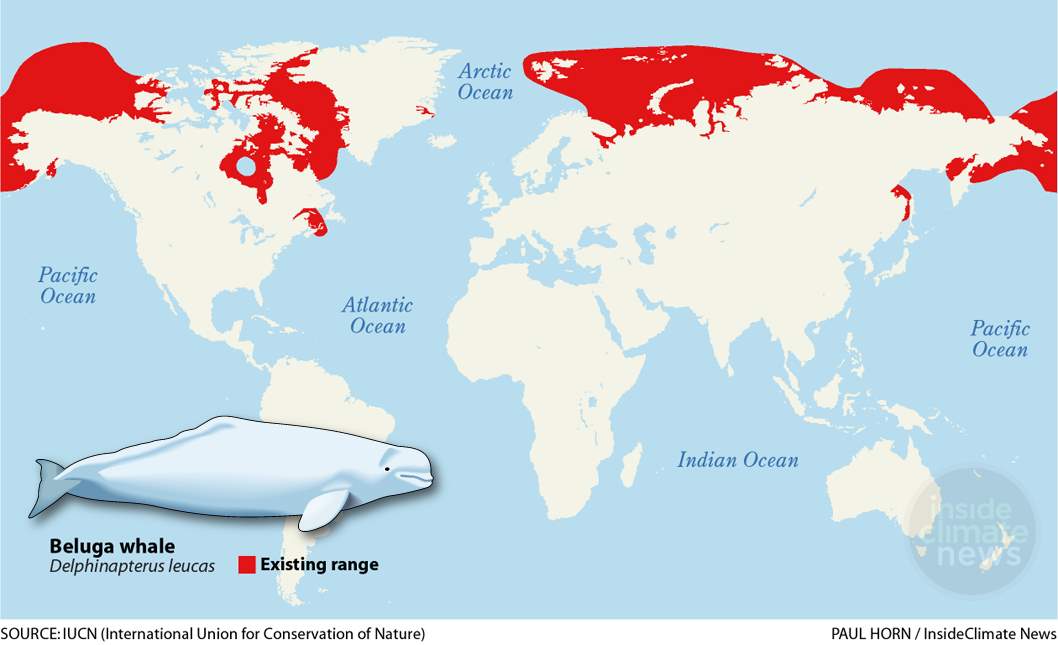
Fig. 4. Geographical distribution of Beluga whales. (Horn, P. (n.d.). [Digital Image]. Retrieved from https://insideclimatenews.org/species/mammals/beluga-whale
Behaviour
Parental Care
Beluga whales rarely have more than one calf at a time, where the responsibilities of parental care solely reside with female beluga whales, also known as sows (Hill et al., 2015). Notably, the most physical contact is shared between a sow and her calf than in comparison to all other interactions within a pod (Hill et al., 2016; Fig. 5.). Physical contact is essential during a calf’s first year of life in order to teach them how to socially interact, bond with others, and cope with agonistic interactions (Hill et al., 2016). This close social connection between a sow and her calf lasts for at least 3 years, beginning from the time of the calf’s birth (Krasnova et al., 2014).
Starting from the day of its birth, the calf cannot go anywhere unaccompanied without its mother or another adult female from the pod (Krasnova, et al., 2014). In the following four days after the calf’s birth, they start becoming independent from their mother (Krasnova et al., 2006). The calf swims slightly farther from the mother, so long as there are other adult females from the pod present (Krasnova et al., 2006). The calf’s swim position in relation to the mother changes as they mature, from being at the mother’s side and tail for the first month, to swimming ahead of her occasionally at two months (Krasnova et al., 2006). A beluga whale calf increases its social activity through interaction with other calves, juveniles, and adults as they develop within their pod (Krasnova et al., 2006). There is also more noticeable head butting and circling conducted by the calf with their mother, which perhaps allows them to practice testing social hierarchies and maintaining relationships within the pod (Krasnova et al., 2006).
Alloparental care has been observed in many mammals, especially the Cetaceans (Leung et al., 2010). Beluga whales are no exception, where female beluga whales, other than the mother of the calf, help nurse it (Leung et al., 2010). This is known as allonursing, where unrelated female beluga whales have the ability to spontaneously lactate when a calf in its pod is in close proximity (Leung et al., 2010). Alloparental care benefits the reproductive success of beluga whales because it increases the calf’s protection from predators, keeps them close to the pod, and increases their rate of survival (Leung et al., 2010). Upon maturation, female calves typically stay in their natal pods for the rest of their lives whereas males leave the maternal pod at four to five years old (Krasnova et al., 2006). The males may return during the breeding season (Krasnova et al., 2006).

Fig. 5. Beluga whale sow and calf (Flood, S. (Photographer). (n.d.). Beluga whale with calf. [Digital Image]. Retrieved from http://www.arkive.org/beluga-whale/delphinapterus-leucas/image-G5973.html
Vocalization
Beluga whales can conduct at least 44 to 48 different types of acoustic signals (Alekseeva et al., 2013). These signals can be divided into four categories: tonal signals, pulsive signals, noise signals, and mechanical signals (Alekseeva et al., 2013). Beluga whale acoustic signals vary in category and frequency depending on the social behaviour (e.g.: whether they are feeding, playing, migrating, etc.) and depending on who they are communicating with in the social hierarchy (Panova et al., 2013; Recchia, 1994; Fig. 6.). With recent advances in technology, researchers are only beginning to decipher the meaning behind all their acoustic signals (Alekseeva et al., 2013; Panova et al., 2013).
Acoustic signaling is most actively used in social interactions (Alekseeva et al., 2013). Short distance communication normally involves pulsive and variable frequency tonal signals, whereas long distance communication is associated with high frequency tonal signals (Alekseeva et al., 2013). It is hypothesized that tonal signals in general are used for group coordination, as they have been most frequently heard in the context of foraging and directional motion behaviours (Alekseeva et al., 2013; Panova et al., 2012). Peeps are often heard during every type of behavioural activity, possibly acting as contact signals between individuals (Alekseeva et al., 2013). On the other hand, squeaks are used when the visibility of other pod members is unclear in an attempt to reconnect with them, whereas pulse and click signals are associated with social affiliation (Panova et al., 2012).

Fig. 6. “Canaries of the Sea” communicating with conspecifics using acoustic signals. (N. A. (n.d.) [Digital Image]. Retrieved from http://www.animalspot.net/beluga-whale.html
Social Interaction with Conspecifics
Beluga whale pods consist of anywhere between 2 to 25 individuals, where the smallest pod unit includes a sow and her calf (Hill et al., 2015; Recchia, 1994). Beluga whales display a multitude of swimming behaviours; this includes mother and calf swims (i.e. involves the most physical contact), pair swims (i.e. swim in close proximity without physical contact), and solitary swimming (Hill et al., 2015). Affiliative interactions (i.e. positive playful behaviours between two or more whales), sexual interactions (i.e. contact or display of the genitals), agonistic interactions (i.e. threatening or aggressive behaviour), and play activity (i.e. manipulation and contact with surrounding stimuli) are other classified behaviours within beluga whale pods (Hill et al., 2015).
Socially hierarchal behaviour is also present in beluga whale pods (Recchia, 1994). This evidence comes from beluga whales in captivity where there are observed differences in acoustical signaling depending on the age and size of the individual being communicated to (Recchia, 1994). For example, an older bull will communicate differently with a juvenile male as opposed to a juvenile female (Recchia, 1994). Dominance in the pod seems to correlate with size, so the bigger the whale, the more dominant behaviours they are entitled to (Recchia, 1994). An example of a dominant behaviour is displacing other pod members from one part of the water to the other (Recchia, 1994). When observed in captivity, larger older females often displaced smaller juvenile females from certain parts of the pool (Recchia, 1994). Moreover, juvenile males could displace juvenile females, but not larger adult females, further indicating that size contributes to social hierarchy in pods (Recchia, 1994).
Beluga whales also instigate a lot of play behaviours, which are particularly crucial to a calf’s social development (Delfour & Aulagnier, 1997). Bubble blowing is an interesting play behaviour that has been observed in beluga whales, although its true purpose remains unclear (Delfour & Aulagnier, 1997). They blow rings of bubbles, followed by circling or pushing them around in the water (Delfour & Aulagnier, 1997). It appears to be a solitary play behaviour, and may help advance an individual’s cognitive and motor development (Delfour & Aulagnier, 1997). Play behaviours in beluga whales decrease with increasing age, so older beluga whales do not initiate play as much as juveniles and calves (Delfour & Aulagnier, 1997). Interestingly, the presence of calves in the pod increases the amount of play behaviour witnessed in the adults (Delfour & Aulagnier, 1997). In fact, the most playful adult beluga whales are sows who are mothers (Delfour & Aulagnier, 1997). The reasoning as to why these play behaviours radiate throughout adult members of the pod in the presence of calves is currently unknown (Delfour & Aulagnier, 1997).
Mating Behaviours and Strategies
Female beluga whales reach sexual maturity at 4 years old and males reach sexual maturity later on at 8 years old (Lomac-Macnair et al., 2015). Socio-sexual behaviour is seasonal, where mating occurs during the late winter and early spring (Lomac-Macnair et al., 2015). Socio-sexual behaviours that have been observed include ventral to ventral contact, ventral presents (i.e. rolling the body so that the ventral side faces their partner), pelvic thrusting, nodding, and physical touching (Glabicky et al., 2010; Lomac-Macnair et al., 2015). Beluga whale mating pairs also perform echelon swims where both of them alter their positions so that they are swimming closely parallel to each other (Glabicky et al., 2010). Male to male sexual contact has also been observed in captive beluga whales (Glabicky et al., 2010). Although the true function is unknown, it is predicted to help establish dominance (Glabicky et al., 2010).
Beluga whales form reproductive gatherings at the breeding location during the summer to breed safely and allow the adults to teach calves survival strategies required to be independent (Alekseeva et al., 2013). These reproductive gatherings consist of females, calves, and a few males who have migrated to join the group during the breeding period (Alekseeva, et al., 2013). Other behaviours displayed in reproductive gatherings include: relaxed swimming, playing, and diving behaviours (Alekseeva et al., 2013).
Ecology
Feeding, diet and Trophic level
Beluga whales are known to eat species that occur in concentrated groups or schools such as fish and shrimp (Quakenbush et al., 2015). Fish make up the majority of the beluga whale diet, with benthic invertebrates and small pelagic species coming second, followed by mesopelagic species (Dauly et al., 1998, Fig.7). They feed on a variety of fish species such as Arctic cod and capelin and Inuit knowledge also suggests they feed on halibut (Marcoux et al., 2012). Invertebrates such as shrimp, crabs and bivalves also make up an important part of the beluga whale diet (Quakenbush et al., 2015). A small part of their diet may also consist of squid (Dauly et al., 1998). Fish species tend to have a bottom up control on the Beluga whale population (Loseto et al., 2009). Beluga whales consume their prey using a mixture of suction feeding and ram feeding (Kane & Marshall, 2009). This suction feeding strategy allows them to consume prey at a high velocity (Kane &, Marshall 2009).
Diet differences among belugas are related to energy requirements and age class (Marcoux et al., 2012). Older belugas tend to feed more on benthic prey and prey in higher trophic levels (Marcoux et al., 2012). Unweaned calves can eat small fish and shrimp, yearlings tend to eat small fish and a few adult salmon and subadults eat a large amount of invertebrates (Quakenbush et al., 2015). There are also some differences between the sexes, with adult females consuming smaller fish and adult males consuming larger fish (Quakenbush et al., 2015). The size of the prey also differs across age classes (Marcoux et al., 2012). Belugas swallow their prey whole, so the size of the chosen prey is limited by the size of the beluga whale’s esophagus (Quakenbush et al., 2012). The different diet amongst age classes comes from the different energy requirements at these ages, such as those for growth and reproduction (Marcoux et al., 2012). Energy requirements differ between sexes with the females requiring more energy during lactation, which can last around 20 months (Marcoux et al., 2012). This results in females needing to consume more prey or more energy rich prey during this time (Marcoux et al., 2012). In addition to energy requirements and age class, diet is also affected by body size (Marcoux et al., 2012).
Diet varies widely based on the population and location of the beluga whales (Ferguson et al., 2010). Populations in the far north eat a lot of shrimp, octopus and Arctic cod (Quakenbush et al., 2015). As you go south, Arctic cod is replaced by saffron cod and in the southern most populations, salmon and smelt play a larger role in their diet (Quakenbush et al., 2015). In Hudson Bay, Arctic cod is most important, while redfish, halibut and shrimp are consumed closer to Greenland (Ferguson et al., 2010). Alaskan populations consume more Pacific salmon, while the Churchill population consumes more capelin (Ferguson et al., 2010). Some of these regional differences can be attributed to latitude and the sea ice cover (Ferguson et al., 2010).
Over time there has been a shift in the beluga diet (Marcoux et al., 2012). Capelin has become increasingly important because as this species continues to spread and invade new areas, it becomes a more available food source for belugas (Marcoux et al., 2012). Climate change will likely result in capelin spreading farther north and becoming even more important to the beluga diet (Ferguson et al., 2010). Beluga diets and foraging are also affected by humans (Ferguson et al., 2016). Mercury contamination is stronger in the fish near the shore, forcing some belugas to forage farther out (Ferguson et al., 2016).
Beluga whale diets can also change throughout the year (Marcoux et al., 2012). Belugas will have changes in feeding in relation to the migration patterns of fish such as halibut and salmon, eating more when they are present in shallow, nearshore waters and less when they migrate to other locations (Quakenbush et al., 2015). Belugas also tend to consume more food during the spring and summer months in some populations while in others they feed more in the fall and winter (Quakenbush et al., 2015, Breton-Honeyman et al., 2016). This occurs because at other times of the year, less energy is spent on foraging and more energy is put into migration, moulting and reproduction (Quakenbush et al., 2015). Beluga whales tend to eat more food during the productive seasons in order to prepare for the less productive seasons (Breton-Honeyman et al., 2016).
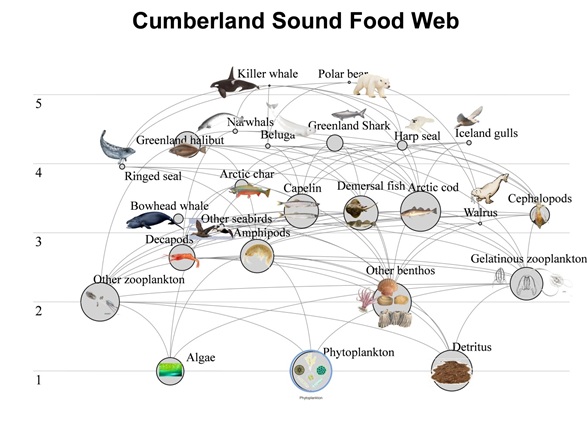
Fig. 7. Trophic food web of an Arctic aquatic ecosystem (Janjua et al. (n.d.). [Digital Image]. Retrieved from http://www.dfo-mpo.gc.ca/science/rp-pr/accasp-psaccma/projects-projets/051-eng.html
Predators
A common predator of the beluga is the killer whale (Quakenbush et al., 2015). The presence of killer whales can affect the feeding patterns of belugas and sometimes result in them not leaving shallow waters to search for food (Quakenbush et al., 2015). Predators also influence habitat choice and reproductive females show strong predator avoidance (Loseto et al., 2006).
Other predators of the beluga whale include polar bears and humans (Loseto et al., 2006). Young calves are also sometimes at risk of attack from adult male belugas, who sometimes commit infanticide (Loseto et al., 2006).
Competition
Beluga whales must compete for prey with several seal species, such as ringed seals and spotted seals, as well as sea lions and harbour porpoises (Quakenbush et al., 2015). They compete with bearded seals for fish such as cod, flatfish and sculpins and compete with bearded seals and walrus for invertebrate prey (Quakenbush et al., 2015). Competition also occurs with humans for fish such as herring and salmon (Quakenbush et al., 2015).
Habitat, Distribution and Migration
Beluga whales are found in the Arctic and Subarctic, and have a circumpolar distribution (Dunn et al., 2014). As the Pleistocene ice sheets retreated, beluga whales moved into new habitats and the Beaufort Sea was likely populated by belugas traveling through the channel created by the Bering land bridge (O’Corry-Crowe et al., 1997). In the post glacial period, some beluga populations then moved to lower latitudes (O’Corry-Crowe et al., 1997).
Overall, habitat selection is based on the availability of food, the suitability of the area for moulting or calving, protection from predators and the level of human disturbance (Quakenbush et al., 2015). Habitat choice is also influenced by the major ocean currents and the flow of nutrients (Hauser et al., 2017).
There are two life strategies in beluga populations, migratory and non-migratory (Quakenbush et al., 2015). The migratory populations travel between shallow, near shore habitats and offshore habitats with deeper water (Quakenbush et al., 2015). Non-migratory populations stay in the shallow, estuary environments (Quakenbush et al., 2015). These estuaries are usually heavily influenced by the tide and have areas with shallow water (Quakenbush et al., 2015). They also contain several different fish species, providing a good food source, as well as providing protection from predators like the killer whale (Quakenbush et al., 2015). Belugas also tend to favour areas with sea ice (Quakenbush et al., 2015). Areas with broken sea ice still give belugas access to the bays and shores while providing protection (Quakenbush et al., 2015). This sea ice is relied upon by migratory belugas as a way to escape predators (Quakenbush et al., 2015). Beluga whales tend to return to familiar habitats each year in order to take advantage of the predictable climate, ocean conditions and food sources (O’Corry-Crowe et al., 1997). Not only do they make use of the salt water oceans and estuaries but they also seasonally use freshwater rivers (Watts & Draper, 1988).
Some beluga whale populations are separated by sex (Marcoux et al., 2012). In the summer months, female belugas form large groups with their calves and males form separate smaller groups (Marcoux et al., 2012). Nursing females and older females are most often found in the estuaries with their calves (Loseto et al., 2006). Calves remain in the shallower waters due to their limited lung capacity and their inability to dive as deep as adult whales (Loseto et al., 2006). The large male belugas spend more time by the sea ice in order to feed on fish species such as Arctic cod, which swim beneath the ice (Loseto et al., 2006). They forage in high risk environments but take advantage of this productive environment to gain the energy needed to support growth (Loseto et al., 2006). There are also groupings that consist of smaller males and females with older calves and these groupings tend to stay near the ice edge (Loseto et al., 2006). These smaller males could be avoiding the aggression of the larger male populations or are potentially providing protection for the young (Loseto et al., 2006). Populations are also separated by age, with older, larger individuals tending to be found farther offshore and smaller, younger individuals being found inshore (Marcoux et al., 2012).
Some belugas will disperse to new populations and this results in some gene flow between the populations (O’Corry-Crowe et al., 1997). Most of these dispersal events involve males; females disperse less often (O’Corry-Crowe et al., 1997). This dispersal is likely due to competition for females (O’Corry-Crowe et al., 1997). Despite this gene flow, populations tend to remain separate and genetically isolated (O’Corry-Crowe et al., 1997). Though different beluga populations may use the same habitat, this tends to occur at different times of year, minimizing the chances of dispersal and gene flow between the populations (Hauser et al., 2014).
The distribution of some beluga populations is shifting due to changes to their habitat and declines in their population (Quakenbush et al., 2015). As the climate changes, beluga ranges will likely change too (O’Corry-Crowe et al., 1997).
Physiology
Moulting
Beluga whales go through a process of moulting approximately once a year and the turnover for epidermal cells is around 70 to 75 days (St. Aubin et al., 1990). There are seasonal differences in the surface of the epidermis (St. Aubin et al., 1990). They tend to have a shade of yellow on dorsal parts of their body, flippers and flukes in the spring as well as a stratum externum on top of the epidermis (St. Aubin et al., 1990). When they enter the estuaries in the summer, they slowly lose the tint of yellow due to the sloughing of epidermal cells and the thinning of the stratum externum (St. Aubin et al., 1990).
The rate of epidermal cell degeneration varies based on the temperature of the waters, and to a certain degree, their behaviour as well (Boily, 1995; Smith et al., 1992; St. Aubin et al., 1990). The warm waters of the estuaries cause a faster rate of sloughing and thinning of the stratum externum while lower temperature waters (around 5 °C) slow down the process (Boily, 1995; St. Aubin et al., 1990). This means that for 8 to 10 months of the year, when they are not in the estuaries, the epidermal cells are replaced very slowly (St. Aubin et al., 1990). By using the warm waters to increase the temperature of their epidermis rather than raising their metabolic rate, they save energy (Boily, 1995). During the time in the estuary, adult belugas also rub against surfaces such as limestone, mud and pebbles, which results in abrasions on the surfaces of their skin (Smith et al., 1992). The skin is then softened by the warm water causing moulting to occur faster and new skin to grow quicker (Smith et al., 1992). By the time they leave for their migration in the fall, their epidermis and stratum externum will be quite thin (St. Aubin et al., 1990). This same moulting process will repeat when they return to the estuary in subsequent years (St. Aubin et al., 1990).
Reproductive System
Some mature male belugas can begin to copulate at around 13 years of age but this is dependent on testes size and testosterone levels (Robeck et al., 2005). If the volume of their testes is 130 cc or smaller, there will not be any spermatozoa present, meaning they will not be able to copulate (Brodie, 1971). Testes with a volume that is greater than 360 cc will contain a small amount of spermatozoa (Brodie, 1971). Testosterone is found at lower levels in immature males than in mature males between January and April (highest was between January and March) which corresponds to their mating season (O’Corry-Crowe, 2009; Robeck et al., 2005). However, there may be some variation since some belugas breed later in the year in May (Brodie, 1971).
Mature female belugas go through annual estrous cycles (2 to 7 each year) that last for approximately 37 days, starting from April to May of each year (Katsumata, 2010). Some belugas can have longer estrous cycles that last up to 48 days (Robeck et al., 2005). Furthermore, it is normal for belugas to have multiple ovulations as well as multiple accessory corpora lutea (Brodie, 1971). Once maturity has been reached, the level of progesterone elevates and shortly after mating, the rectal temperature also increases in response (Katsumata, 2010).
The length of gestation in beluga whales is around 14 months and calves are usually birthed sometime between late July and early August (Brodie, 1971; Sergeant, 1973). In captive belugas, the gestation period was observed to be around 15 and half months (Robeck et al., 2005). After gestation, it takes a total of approximately 55 hours for birth to occur (Robeck et al., 2005). At this time, females will generally fast or decrease the amount of food consumption (Robeck et al., 2005). Some females will lactate for around 2 years (Brodie, 1971), while others have been seen to lactate for a shorter period of about 20 months (Sergeant, 1973). Beluga whales will rarely have a second calf while they are still providing parental care to the first (Sergeant, 1973). Therefore, their reproductive cycle is around 3 years and they can be pregnant up to 10 times in their lifetime (Brodie, 1971; Sergeant, 1973).
Developmental Stages
There are inconsistencies between studies regarding the growth of both sexes of beluga whales which may be due to the limited data on calves (Brodie, 1971). Fetus sizes can range from 156 cm to 174 cm in length and 63.7 kg to 94 kg in weight (Brodie, 1971). However, there is a possibility for fetuses to fall outside of these ranges since this was only observed in one study using two beluga whales (Brodie, 1971). At birth, these two belugas averaged to be 159.6 cm in length and weighed 78.3 kg (Brodie, 1971). From another observational study, it seems that their length can vary between 120 cm to 183 cm (Sergeant, 1973), which implies that their weights can vary as well. Beluga whale sizes and weights during development are not identical between individuals and more studies will be necessary in order to find an accurate range (Brodie, 1971; Sergeant, 1973).
There are also discrepancies regarding when female and male beluga whales mature (e.g.: Brodie (1971), Katsumata (2010)). Females can mature at an age as young as 4 to 7 years old (Brodie, 1971; Sergeant, 1973), but have also been observed to mature between the ages of 9 and 13 (Katsumata, 2010; O’Corry-Crowe, 2009). Males mature later than females at around 8 to 9 years old (Brodie, 1971; Sergeant, 1973). Sometimes, male maturation can occur past 12 years of age (O’Corry-Crowe, 2009). Similarly to beluga whale sizes, more studies are required to pinpoint the ages at which maturation occurs and whether there are factors that cause differences. In the past, beluga whales were thought to only live up to 25 years old (Sergeant, 1973), but recently, they have discovered that they can live up to 80 years old (O’Corry-Crowe, 2009; Stewart et al., 2006). This may be due to the discovery regarding teeth growth.
Beluga whales do not begin to grow teeth until they are 2 years old and they continue to emerge throughout the 3rd year (Brodie, 1971). There is an outer layer on top of the dentine, known as the cementum, which grows throughout their lifetime and provides a point of attachment for periodontal ligaments (Stock et al., 2017). In the past, it was thought that two layers of dentine were incorporated annually (Sergeant, 1973; Fig. 8.). However, recent evidence shows that only one layer of dentine is added each year (Stewart et al., 2006). As a result, the method for aging beluga whales have changed throughout history; thereby providing an explanation for the age discrepancies observed.
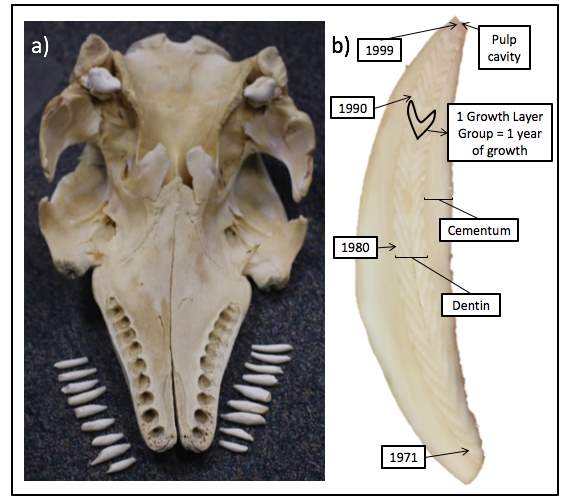
Fig. 8. Anatomy of the beluga whale skull and the layers of a tooth. (Nelson, M. (Photographer). (n.d.). A beluga whale skull with a labeled tooth. [Digital Image]. Retrieved from https://news.uaf.edu/new-research-to-investigate-factors-limiting-beluga-whale-recovery-hold/
Beluga whale calves are born a grey-cream colour (O’Corry-Crowe, 2009). Not long after, they change to either a blue-grey or dark-brown colour (O’Corry-Crowe, 2009). At the age of 6 or 7 (females and males respectively), their skin begins to get lighter and by the age of 14 or 18 (females and males respectively), they will be fully white (Brodie, 1971; Harington, 2008; O’Corry-Crowe, 2009).
Beluga calves seem to develop vocalization through learning rather than through genetics (Vergara & Barrett-Lennard, 2008). The development of vocalization was only observed in one beluga whale and although there could be variation between different beluga whales, it can still provide an approximate timeline (Vergara & Barrett-Lennard, 2008). An hour after the calf was born, he could already start making pulsed calls that did not require much energy (Vergara & Barrett-Lennard, 2008). By the time he was 4 months old, he had developed different calls and was able to mix them (Vergara & Barrett-Lennard, 2008). As he continued to grow, his vocalization also improved and he was able to vocalize like an adult at the age of 20 months (Vergara & Barrett-Lennard, 2008).
Thermoregulation
Arctic marine mammals use thermoregulation to withstand cold temperature waters (Kasting et al., 1989; Ryg et al., 1993). The amount of heat loss through convection is dependent on the body length and appendages of the animal (Ryg et al., 1993). Most cetaceans lose the same amount of heat per unit2 in their trunk as they do in their appendages, but in beluga whales, out of the total amount of heat loss, they only lose 20% through their appendages (Kasting et al, 1989; Ryg et al., 1993). When an animal is swimming, the amount of heat loss increases slightly due to forced convection (Ryg et al., 1993). It has been theorized that their appendages and the mass of blubber have evolved in a way that maintains hydrodynamics as well as insulation (Ryg et al., 1993).
Heat can also be loss through respiration, but it is not as notable of a concern as convection (Kasting et al., 1989; Ryg et al., 1993). The exhaled air of beluga whales has a much lower temperature when compared to their core temperature and thus not much heat is loss (Kasting et al., 1989). Although the mechanism for this phenomenon is not certain, there are theories that can possibly explain it (Kasting et al., 1989). Before exhalation, the pressure within the blowhole may have been elevated which can cause condensation to occur (Kasting et al., 1989). This will reduce the amount of water evaporation, and in turn will decrease the amount of heat lost (Kasting et al., 1989). More research is required in order to be certain that this is a probable mechanism.
Similarly to other cetaceans, the metabolic rates of beluga whales are elevated, which is both energetically costly and inefficient since they require more food (Kasting et al., 1989). In addition, their body shape does not maximize the amount of heat retention due to the large surface area of their appendages and their thermal conductance is high in comparison to smaller cetaceans (Kasting et al., 1989). From these observations, it has been suggested that they are able to survive without further improvements to maximize insulation (Kasting et al., 1989). The reason for increasing their metabolic rate may be to prepare for unforeseen circumstances like sickness, injury, etc. and not exclusively for thermoregulation (Kasting et al., 1989). It might be more beneficial for Arctic marine mammals to create excess heat (i.e. increase metabolic rate) rather than to store heat (i.e. increase metabolic rate and reduce heat loss simultaneously; Kasting et al., 1989).
Blubber is important for a number of reasons; two of which include storing energy and thermoregulation (Cornick et al., 2016). Blubber is distributed differently across the beluga whale’s body, with an increasing amount of blubber beginning at the end of the head and thickening towards the dorsal ridge where the thickest region is right past the dorsal ridge (Cornick et al., 2016). The total amount of blubber in their body is dependent on age, home range, sex and reproductive activity (Cornick et al., 2016). Adult beluga whales tend to have less blubber in the spring and more in the autumn while there is no storage difference in juveniles between seasons (Cornick et al., 2016). There is a possibility that juveniles do not alter their blubber mass because the amount that they possess has reached a thermoregulation threshold (Cornick et al., 2016). Furthermore, since juveniles are able to receive parental care (i.e. milk from their mom) for a few years, they may not need to store as much blubber (Cornick et al., 2016).
Diving and Respiration
Respiration rates are not consistent throughout all individuals but vary depending on the season, their body size, their age and their sex (George & Noonan, 2014). Generally, males, calves and smaller adults have higher respiration rates (George & Noonan, 2014). In addition, beluga whales have higher rates during the winter and spring in comparison to the summer and fall (George & Noonan, 2014).
Beluga whales will dive great distances to reach the seabed where there is an abundant amount of food for them to forage (Martin & Smith, 1992; Martin & Smith, 1999; Ridgway et al., 1984). They can dive as deep as 600 m but their range preference appears to be no further than 400 m (Ridgway et al., 1984). The maximum length of time that they can stay submerged (i.e. aerobic dive limit) is limited by the amount of oxygen that they had stored within their blood and tissues as well as oxygen consumption rates (Shaffer et al., 1997). The aerobic dive limit (ADL) varies just as respiration rates are different between individuals (Shaffer et al., 1997). Some beluga whales are able to stay underwater for 9 to 10 minutes while others could stay submerged for approximately 23 minutes (Martin & Smith, 1999; Shaffer et al., 1997). Once the ADL is passed, the low levels of oxygen will cause lactate to be produced and accumulate (Shaffer et al., 1997).
To remain underwater for extended periods of time, a number of adaptations have evolved. Firstly, beluga whales have been observed to descend slowly (less than or equal to 2 m/s) when diving, which may be a way to conserve oxygen (Martin & Smith, 1999; Shaffer et al., 1997). Their ascents are generally faster in comparison but some beluga whales may slow down at around 140 m to prevent gas bubbles from forming too quickly either in their blood or tissues (Martin & Smith, 1992). This has not been observed in many beluga whales and more data would be necessary to confirm that this is common between all individuals.
Interestingly, to store more oxygen for long dives, beluga whales do not have increased lung capacities but instead have greater amounts of respiratory pigments which are located in their muscles and blood (Dhindsa et al., 1974; Snyder, 1983). Their lungs hold a small volume of oxygen and will collapse when under the pressure of diving (Snyder, 1983). Beluga whales also have a high P50 (i.e. a low affinity for oxygen) allowing for more oxygen to be transferred from the blood to the tissues, keeping them oxygenated (Dhindsa et al., 1974; Snyder, 1983).
Genetics and Conservation
According to IUCN, beluga whales are a near threatened species (Jefferson et al., 2012). Several populations of belugas are threatened and have low population sizes, such as the population in Cumberland Sound (Marcoux et al., 2012). The Cook Island population also is decreasing due to overharvesting (Quakenbush et al., 2015).
Natural Threats
Natural factors contributing to the decline in beluga whale populations include disease, competition, and live stranding (Norman, Hobbs, Goertz, Burek-Huntington, Shelden et al., 2015). In terms of pathogens, parasites in the kidney, pneumonia, and lungworm infestation are some of the most common ones to invade a population (Norman et al., 2015). Climate change may exacerbate the spread of parasites among beluga whales as it will limit the beluga whales’ spatial distribution, thereby increasing the density of the population and making it easier for transmission (Norman et al., 2015). In addition, warmer sea surface temperatures give pathogens the potential to adapt and increase in frequency (Norman et al., 2015). It is also predicted to trigger distress in some of their prey items, subsequently affecting their nutritional value (Norman et al., 2015).
There is also the potential threat of increased competition with other predatory species for prey, as shark and pike fish habitat ranges shift to overlap with those of the beluga whale (Carter & Nielson, 2011). Notably, the warmer sea surface temperatures instigate the melting of glaciers, thus creating an influx of freshwater into saline regions where beluga whales forage (Carter & Nielson, 2011). Consequently, the change in water salinity negatively affects the timing of prey migrations and food availability for the beluga whales (Norman et al., 2015). It is important to note that live stranding behaviour displays evidence of threats to a beluga population (Norman et al., 2015). Reasons behind live stranding include escaping predation, being swept up by high tides, or illness (Norman et al., 2015).
Anthropogenic Threats
Some anthropogenic threats negatively affecting the beluga whale population include pollution runoff, accidental catches from commercial fishing, pressure on prey items as a result of commercial fishing, and underwater noise as a result of vessel traffic in and around the coastline (Carter & Nielson, 2011; Norman et al., 2015). The hazardous runoff and pollution that affect the surrounding water can increase the chances of disease and chemical borne illness amongst beluga whales (Carter & Nielson, 2011). Increases in sport fishing for salmon populations and noise pollution due to coastline trains also make it distressing for belugas to occupy certain habitats close to the shoreline in Alaska (Carter & Nielson, 2011).
Belugas themselves have been victims of whaling, both for food and sport (Marcoux et al., 2012). Fisheries can also threaten beluga populations by decreasing their food populations (Marcoux et al., 2012). Belugas are also very important to the Aboriginal/Indigenous communities and are used as a food source, as well as carry cultural significance (Breton-Honeyman et al., 2016).
Implications of Population Genetics to Conservation
Upon inspecting the mitochondrial DNA in beluga whales, two genetically distinct groups have been identified (Gladden et al., 1999). There are the western (Pacific) North American beluga whales and the eastern (Atlantic) North American beluga whales (Gladden et al., 1999). Beluga whale populations are philopatric, meaning they return to the same breeding ground every year (Hoezel, 1998). Despite sympatric gatherings in summer, populations in these regions are genetically heterogeneous at the breeding sites (Hoezel, 1998). Genetic variation in a population is critical for a population’s ecological resilience; thus indicating that it is extremely important to leave the summer breeding sites undisturbed by commercial fishing and other human activity in order to preserve the population’s genetic diversity (Hoezel, 1998).
There is specific concern regarding the Cook Inlet beluga whale population, as they are geographically isolated from the other populations of beluga whales and there is a lot of commercial development occurring along their breeding estuaries (Carter & Nielson, 2011; Norman et al., 2015).
References
Alekseeva, Y. I., Panova, E. M., & Bel’kovich, V. M. (2013). Behavioral and acoustical characteristics of the reproductive gathering of beluga whales (Delphinapterus leucas) in the vicinity of Myagostrov, Golyi Sosnovets, and Roganka Islands (Onega Bay, the White Sea). Biology Bulletin, 40(3), 307-317.
Boily, P. (1995). Theoretical heat flux in water and habitat selection of phocid seals and beluga whales during the annual molt. Journal of Theoretical Biology, 172, 235-144.
Breton-Honeyman, K., Hummill, M.O., Furyal, C.M., & Hickie, B. (2016). Inuit knowledge of beluga whale (Delphinapterus leucas) foraging ecology in Nunavik (Arctic Quebec), Canada. Canadian Journal of Zoology, 94(10), 713-726.
Brodie, P. F. (1971). A reconsideration of aspects of growth, reproduction, and behavior of the white whale (Delphinapterus leucas), with reference to the Cumberland Sound, Baffin Island, Population. Journal of the Fisheries Research Board of Canada, 28: 1309-1318.
Carter, B. T. G., & Nielsen, E. A. (2011). Exploring ecological changes in Cook Inlet beluga whale habitat though traditional and local ecological knowledge of contributing factors for population decline. Marine Policy, 35(3), 299-308.
Cornick, L. A., Quakenbush, L. T., Norman, S. A., Pasi, C., Maslyk, P., Burek, K. A., Goertz, C. E. C., & Hobbs, R. C. (2016). Seasonal and developmental differences in blubber stores of beluga whales in Bristol Bay, Alaska using high-resolution ultrasound. Journal of Mammalogy, 97(4), 1238-1248.
Dauly, D., Trites, A.W., Capuli, E., & Christensen, V. (1998). Diet composition and trophic levels of marine mammals. ICES Journal of Marine Science, 55(3), 467-481.
Delfour, F., & Aulagnier, S. (1997). Bubbleblow in beluga whales (Delphinapterus leucas): a play activity? Behavioural Processes, 40(2), 183-186.
Dhindsa, D. S., Metcalfe, J., Hoversland, A. S., Hartman, R. A. (1974). Comparative studies of the respiratory functions of mammalian blood X. Killer whale (Orcinus orca Linnaeus) and beluga whale (Delphinapterus leucas). Respiration Physiology, 20, 93-103.
Dunn, J., Still, R. & Harrop, H., (2014). Britain’s sea mammals; whales, dolphins, porpoises and seals and where to find them. Princeton: Princeton University Press.
Ferguson, S.H., Loseto, L.L. & Mallory, M.L. (2010). A little less arctic top predators in the world’s largest northern inland sea, Hudson Bay. New York: Springer.
George, E. M., & Noonan, M. (2014). Respiration rates in captive beluga whales (Delphinapterus leucas): Effects of season, sex, age and body size. Aquatic Mammals, 40(4), 350-356.
Glabicky, N., DuBrava, A., & Noonan, M. (2010). Social-sexual behaviour seasonality in captive beluga whales (Delphinapterus leucas). Polar Biology, 33, 1145-1147.
Gladden, J. G., Ferguson, M. M., Friesen, M. K., & Clayton, J. W. (1999). Population structure of North American beluga whales (Delphinapterus leucas) based on nuclear DNA microsatellite variation and contrasted with the population structure revealed by mitochondrial DNA variation. Molecular Ecology, 8(3), 347-363.
Harington, C. R. (2008). The evolution of arctic marine mammals. Ecological Applications, 18(2), S23-S40.
Hauser, D. D. W., Laidre, K. L., Stern, H. L., Moore, S. E., Suydam, R. S., & Richard, P. R. (2017). Habitat selection by two beluga whale populations in the Chukchi and Beaufort seas. PLoS ONE, 12(2), 1-15.
Hauser, D. D. W., Laidre, K. L., Suydam, R. S., & Richard, P. R. (2014). Population-specific home ranges and migration timing of Pacific Arctic beluga whales (Delphinapterus leucas). Polar Biology, 37(8), 1171-1183.
Hill, H. M., Alvarez, C. J., Dietrich, S., & Lacy, K. (2016). Preliminary findings in beluga (Delphinapterus leucas) tactile interactions. Aquatic Mammals, 42(3), 277-291.
Jefferson, T.A., Karkzmarski, L., Laidre, K., O’Corry-Crowe, G., Reeves, R., Rojas-Bracho, L., Secchi, E., Slooten, E., Smith, B.D., Wang, J.Y., & Zhou, K. (2012). Delphinapterus leucas. The IUCN Red List of Threatened Species 2012: e.T6335A17690692. Retreived from http://dx.doi.org/10.2305/IUCN.UK.2012.RLTS.T6335A17690692.en.
Kane, E.A., & Marshall, C.D. (2009). Comparative feeding kinematics and performance of odontocetes: Belugas, Pacific white sided-dolphins and long-finned pilot whales. The Journal of Experimental Biology, 212(24), 3939-3950.
Kasting, N. W., Adderley, S. A. L., Safford, T., & Gilbey Hewlett, K. (1989). Thermoregulation in beluga (Delphinapterus leucas) and killer (Orcinus orca) whales. Physiological Zoology, 62(3), 687-701.
Katsumata, E. (2010). Study on reproduction of captive marine mammals. Journal of Reproduction and Development, 56(1), 1-8.
Krasnova, V. V., Bel’kovich, V. M., & Chernetsky, A. D. (2006). Mother-infant spatial relations in wild beluga (Delphinapterus leucas) during postnatal development under natural conditions, Biology Bulletin, 33(1), 53-58.
Leatherwood, S., Caldwell, D. K., & Winn, H. E. (1976). Whales, dolphins and porpoises of the Western North Atlantic; a guide to their identification. Retrieved from http://www.gutenberg.org/files/33527/33527-h/33527-h.htm
Leung, E. S., Vergara, V., & Barrett-Lennard, L. G. (2010). Allonursing in captive belugas. Zoo Biology, 29(5), 633-637.
Lomac-Macnair, K. S., Smultea, M. A., Cotter, M. P., Thissen, C., & Parker, L. (2015). Socio-sexual and probable mating behaviour of Cook Inlet beluga whales, Delphinapterus leucas, observed from an aircraft. Marine Fisheries Review, 77(2), 32.
Loseto, L. L., Richard, P., Stern, G. A., Orr, J., & Ferguson, S. H. (2006). Segregation of Beaufort sea beluga whales during the open-water season. Canadian Journal of Zoology, 84(12), 1743-1751.
Loseto, L. L., Stern, G. A., Connelly, T. L., Deibel, D., Gemmill, B., Prokopowicz, A., Frotier, L., & Ferguson, S. H. (2009). Summer diet of beluga whales inferred by fatty acid analysis of the eastern beaufort sea food web. Journal of Experimental Marine Biology and Ecology, 374(1), 12-18.
Macdonald, D. W. (2007). Beluga and narwhal. In T. Martin & P. Brotherton (3rd ed.). The encyclopedia of mammals. doi: 10.1093/acref/9780199206087.001.0001
MacLaren, P. L., & Davis, R. A. (1982). Winter distribution of Arctic marine mammals in ice-covered waters of eastern North America. Offshore Labrador Biological Studies Report, p.13-114.
Marcoux, M., McMeans, B.C., Fisk, A.T., & Ferguson, S.H. (2012). Composition and temporal variation in the diet of Beluga whales, derived from stable isotopes. Marine Ecology Progress Series, 471, 283-291.
Martin, A. R., & Smith, T. G. (1992). Deep diving in wild, free-ranging beluga whales, Delphinapterus leucas. Canadian Journal of Fisheries and Aquatic Sciences, 49, 462-466.
Martin, A. R., & Smith, T. G. (1999). Strategy and capability of wild belugas, Delphinapterus leucas, during deep, benthic diving. Canadian Journal of Zoology, 77, 1783-1793.
Milinkovitch, M. C., Meyer, A., & Powell, J. R. (1994). Phylogeny of all major groups of cetaceans based on DNA sequences from three mitochondrial genes. Molecular Biology and Evolution, 11(6), 939-948.
Norman, S. A., Hobbs, R. C., Goertz, C. E. C., Burek-Huntington, K. A., Shelden, K. E. W., & Smith, W. A. (2015). Potential natural and anthropogenic impediments to the conservation and recovery of Cook Inlet beluga whales, Delphinapterus leucas. Marine Fisheries Review, 77(2), 89.
O’Corry-Crowe, G. M. (2009). Beluga whale Delphinapterus leucas. In W. F. Perrin, B. G. Würsig & J. G. M. Thewissen (2nd ed.), Encyclopedia of marine mammals (108-112). Retrieved from ScienceDirect database.
O’Carry-Crowe, G. M., Suydam, R. S., Rosenberg, A., Frost, K. J., & Dizon, A. E. (1997). Phylogeography, population structure and dispersal patterns of the beluga whale, Delphinapterus leucas in the western near arctic revealed by mitochondrial DNA. Molecular Ecology, 6(10), 955-970.
Panova, E. M., Belikov, R. A., Agafonov, A. V., & Bel’kovich, V. M. (2012). The relationship between the behavioral activity and the underwater vocalization of the beluga whale (Delphinapterus leucas). Marine Biology, 52(1), 79-87.
Quakenbush, L. T., Suydam, R. S., Bryan, A. L., Lowry, L. F., Frost, K. J., & Mahoney, B. A. (2015). Diet of beluga whales, Delphinapterus leucas, in Alaska from stomach contents, March-November. Marine Fisheries Review, 77(1), 70-84.
Recchia, C. A. Social behaviour of captive belugas, Delphinapterus leucas. Rep. no. WHOI 94-03. Woods Hole Oceanographic Institute, MA, 1994, 206.
Ridgway, S. H., Bowers, C. A., Miller, D., Schultz, M. L., Jacobs, C. A., & Dooley, C. A. (1984). Diving and blood oxygen in the white whale. Canadian Journal of Zoology, 62, 2349-2351.
Robeck, T. R., Monfort, S. L., Calle, P. P., Lawrence Dunn, J., Jensen, E., Boehm, J. R., Young, S., & Clark, S. T. (2005). Reproduction, growth and development in captive beluga (Delphinapterus leucas). Zoo Biology, 24, 29-49.
Ryg, M., Lydersen, C., Knutsen, L. Ø., Bjørge, A., Smith, T. G., & Øritsland, N. A. (1993). Scaling of insulation in seals and whales. Journal of Zoology, 230, 193-206.
Sergeant, D. E. (1973). Biology of white whales (Delphinapterus leucas) in Western Hudson Bay. Journal of the Fisheries Research Board of Canada, 30, 1065-1090.
Shaffer, S. A., Costa, D. P., Williams, T. M., & Ridgway, S. H. (1997). Diving and swimming performance of white whales, Delphinapterus leucas: An assessment of plasma lactate and blood gas levels and respiratory rates. Journal of Experimental Biology, 200, 3091-3099.
Smith, T. G., & Hammill, M. O. (1986). Population estimates of white whale, Delphinapterus leucas in James Bay, eastern Hudson Bay, and Ungava Bay. Canadian Journal of Fisheries and Aquatic Science, 43, 1982-1987.
Smith, A. J., Higdon, J. W., Richard, P., Orr, J., Bernhardt, W., & Ferguson, S. H. (2017). Beluga whale summer habitat associations in the Nelson River estuary, western Hudson Bay, Canada. PLoS One, 12(8), 1-19.
Smith, T. G., St. Aubin, D. J., & Hammill, M. O. (1992). Rubbing behaviour of belugas, Delphinapterus leucas, in a high Arctic estuary. Canadian Journal of Zoology, 70, 2405-2409.
Snyder, G. K. (1983). Respiratory adaptations in diving mammals. Respiration Physiology, 54, 269-294.
St. Aubin, D. J., Smith, T. G., & Geract, J. R. (1990). Seasonal epidermal molt in beluga whales, Delphinapterus leucas. Canadian Journal of Zoology, 68, 359-367.
Stewart, R. E. A., Campana, S. E., Jones, C. M., & Stewart, B. E. (2006). Bomb radiocarbon dating calibrates beluga (Delphinapterus leucas) age estimates. Canadian Journal Zoology, 84, 1840-1852.
Stock, S.R., Finney, L.A., Telser, A., Maxey, E., Vogt, S., & Okasinski, J.S. (2017). Cementum structure in beluga whale teeth. Acta Biomaterialia, 48, 289-299.
Vergara, V., & Barrett-Lennard. (2008). Vocal development in a beluga calf (Delphinapterus leucas). Aquatic Mammals, 34(1), 123-143.
Waddell, V. G., Milinkovitch M. C., Bérubé, M., & Stanhope, M. J. (2000). Molecular phylogenetic examination of the Delphinoidea trichotomy: Congruent from three nuclear loci indicates that porpoises (Phocoenidae) share a more recent common ancestry with white whales (Monodontidae) than they do with true dolphins (Delphinidae). Molecular Phylogenetics and Evolution, 15(2), 314-318.
Watts, P. D., & Draper, B. A. (1988). Beluga whale ecology: a concern for freshwater biologists? Freshwater Biology, 20(2), 211-213.
Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allRelated Content
All TagsContent relating to: "Marine Studies"
Marine studies look at the oceans and seas from the resources they provide, the diverse organisms and life that live in them to the threats and effects of climate change on marine habitats.
Related Articles
DMCA / Removal Request
If you are the original writer of this dissertation and no longer wish to have your work published on the UKDiss.com website then please:




