Bacteria Resistance to Antibiotics in Blood Cancer Patients
Info: 16136 words (65 pages) Dissertation
Published: 5th Jul 2021
Abstract
In the UK alone there are 70 patients every day being diagnosed with blood cancer. With the harsh treatment of cancer these patients’ immune system are being immunocompromised leaving them vulnerable to opportunistic organisms such as coagulase negative staphylococcus bacteria.
In Gwynedd hospital these bacterial infection of cancer patients are treated with the glycopeptides antibiotic, called Teicoplanin. Unfortunately coagulase negative bacteria appear to be less susceptible to glycopeptides antibiotics. In this study coagulase negative staphylococci strains are taken from patients with blood cancer and are tested for potential heterogeneity if the MIC/MBC ratio appear to be higher or equal to 32. The result of this study did not justify the hypothesis as no strains appear to be potentially heterogenic, with this studies hypothesis resulting into a null hypothesis with all the strains appearing to be sensitive towards Teicoplanin.
Contents
Other types of drugs used to treat cancer
Coagulase negative staphylococci infections (CoNS)
Staphylococcus epidermidis (S. epidermidis)
Teicoplanin vs Vancomycin – why do we use Teicoplanin?
Summary of Antibacterial Activity of Teicoplanin
Vancomycin resistance protein (vanA)
Vancomycin resistance protein B (vanB)
Conclusion for both vanA and vanB genes
Introduction
What is blood cancer?
In the UK, 70 patients per day are diagnosed with blood cancer; this includes cancer affecting blood, bone marrow and lymphatic system. (Nolan, 2017) There are three categories of blood cancer, leukemia, lymphoma and myeloma. (Hematology.org, 2017) Diagram 1 explains the differences between them with table 1 comparing the different types of blood cancer with each other.
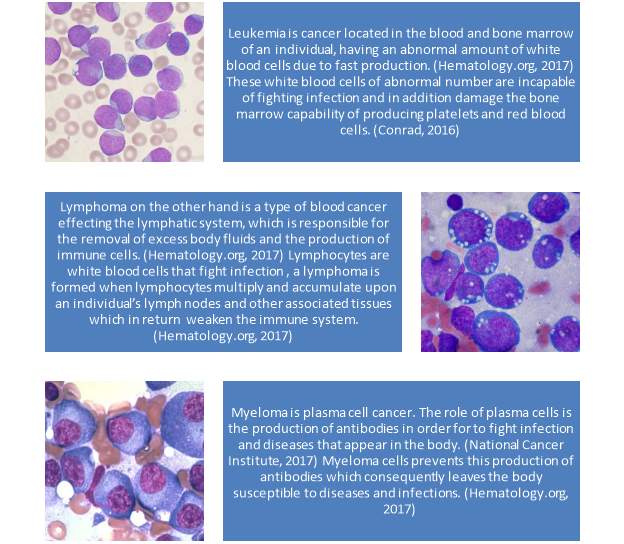
Diagram 1 – illustrates the 3 different types of blood cancer (types by Nolan, 2017) – images from Christaras. A (2016)
Table 1 shows the comparison of the different blood cancer types (based on Nolan, 2017)
| Type of blood cancer | Affect | Cause | Development | Sub-types |
| Leukaemia
|
White blood cells | Production of abnormal number of immature white blood cells clogging up your bone marrow. Preventing a balanced immune system and healthy blood. | Acute leukemia progresses quickly and needs to be treated urgently. Chronic leukemia develops more slowly, over months or years. | Acute myeloid leukemia (AML)
Acute lymphoblastic leukemia (ALL) Chronic myeloid leukemia (CML) Chronic lymphocytic leukemia (CLL) Acute promyelocytic leukemia (APL) Hairy cell leukemia (HCL) Large granular lymphocytic leukemia (LGL) T-cell acute lymphoblastic leukemia (T-ALL) Chronic myelomonocytic leukemia (CMML) |
| Lymphoma
|
Lymphatic system. | Overproduction of lymphocytes, overloading and compromising your immune system. | Lymphoma can grow in various portions of your body, for example lymph nodes, bone marrow, blood, spleen and other organs. | Non-Hodgkin lymphoma (NHL).
Hodgkin lymphoma |
| Myeloma | Plasma cells found in your bone marrow. | Large numbers of abnormal plasma cells gather in your bone marrow and stop producing an important part of your immune system. | The risk of myeloma increases up and till 40% of cases in people aged over 70. | Myelodysplastic syndromes (MDS) |
Treatment of blood cancer
Treatment of cancer has a strong impact on the quality of life of the patients. (Megari, 2013)
Symptoms such as fatigue pain and or vitality seem to be the most common symptoms affecting hematological cancers with these symptoms getting worse in progressive stages. These symptoms do appear in other type of cancers as well with fatigue appearing greater in patients of hematological cancers. (Niederhuber et al., 2013)
Long term survival prognosis after treatment favors younger individuals that have undergone treatment since children. (Megari, 2013)
The most common therapies associated with blood cancer are chemotherapy and radiotherapy where in some cases a bone marrow transplant or stem cell transplant may be necessary. (Niederhuber et al., 2013) Chemotherapy is a combination of different drugs that kill cancer cells or stop them from increasing or spreading any further with the ultimate goal to cure the patient from cancer or in some cases to slow down the outcome of the disease. Chemotherapy attacks cells that actively multiplying since cancer cells have the attribute of constant growth and multiplication chemotherapy seems to appear as the perfect therapy. However chemotherapy also affects healthy cells in the individual’s body hence why side effects such as hair loss occur. Once chemotherapy is over, the healthy cells will be repaired and develop normally once again. (Skeel and Khlief, 2015)
Intense chemotherapy can cause neutropenia, cytotoxic effect and affecting the cells that line the gastrointestinal. The type of treatment is determined by the type of blood cancer as well as the stage, aggressiveness and the overall health of the individual tract. (Gedik et al., 2014) (O’Brien et al., 2017)
Many times a combination of chemotherapy drugs are used or even a combination of chemotherapy with radiotherapy. (Ko, Dollinger and Rosenbaum, 2008)
How Chemotherapy Drugs Work
There are over 100 different chemotherapy drugs, used individually or as combination, in order to treat various types of cancer (Ko, Dollinger and Rosenbaum, 2008)
Table 2 indicates the different drugs, with treatment and working. These specific drugs have different chemical composition are taken differently and each is used to treat a specific type of cancer and have different side effects (Skeel and Khlief, 2015)
Table 2 – Chemotherapy treatment created from (Skeel and Khlief, 2015)
| Treatment name | Working | Cancer type treatment | examples |
| Alkylating agents
|
Alkylating agents keep the cell from reproducing by damaging its DNA | Cancers of the lung, breast, ovary , leukemia, lymphoma, Hodgkin disease, multiple myeloma, and sarcoma |
|
| Antimetabolites
|
Interfere with DNA and RNA growth by replacing normal building blocks of RNA and DNA. Cells of cancer are damaged during the phase when the cell’s chromosomes are being copied. | Treatment of leukemia, breast cancer, ovary and of the gastro intestinal tract cancers for example stomach and pancreatic cancer. |
|
| Anti-tumor antibiotics
|
They work by altering the DNA inside cancer cells to preserve them from increasing and reproducing. | Variety of cancers. |
Examples of anthracyclines include:
|
| Topoisomerase inhibitors
|
These drugs affect topoisomerases, which help separate the strands of DNA so they can be copied. | Certain leukemia, lung, ovarian, gastrointestinal, and other cancers. |
|
| Mitotic inhibitors
|
Prevent cells from separating to form new cells. | Breast, lung, myelomas, lymphomas, and leukemia. |
|
| Corticosteroids
|
Hormone treatment | Many types of cancer, as well as other illnesses. |
|
Other types of drugs used to treat cancer
There are a variety of alternative treatments available such as radiotherapy that functions by using particles of high energy such x-rays and other type of radiation to terminate or damage cancer cells. Targeted therapies are used to attack cancer cells, to keep the cancer under control or keep it from coming back. Differentiating agent act on the cancer cells to make them mature into normal cells. (Ruddon, 2007) The usage of sex hormones or hormone-like drugs, also called hormone therapy, slow the growth of breast, prostate, and endometrial (uterine) cancers. (Niederhuber et al., 2013) Lastly immunotherapy can also be used to assist the immune systems to recognize and attack cancer cells. (Ruddon, 2007)
Immunocompromised
When an individual has hematological cancer the immune system can be weakened either by the malignancies itself or by the treatment of chemotherapy, leading to possible infections and bacterial invasion. (Tessier and D. Sifri, 2011)
The paper by Staudinger et all, indicates that patients with cancer appear to be immunocompromised due to the illness itself or due to the cytotoxic chemotherapy that they undergo resulting in neutropenia that happens when the myelosuppression which happens due to the malignant infiltration occurs. (Staudinger and Pène, 2014)
In addition to measureable deficiencies, neutrophils also display numerous efficient flaws in chemotaxis, phagocytosis, bactericidal capacity and respiratory burst. This flaws account for impairment in skin and mucosal reliability. (Staudinger and Pène, 2014) The alteration seen in the immune cells such as the reduced number of lymphocytes undergoing apoptosis or the increase of suppressor cells that derived from the myeloid are linked with the immune system reaction upon infection. (Murray, Rosenthal and Pfaller, 2005)
Cancer patients who have septicemia have encouraging survival rate outlook due to the constant updates in medicine , as patients who are admitted due to severe septicemia to the hospital not only improve in more than 50 % of the time they have a better outlook for a better and longer life. (Anderson and May, 2010) A way to reduce the risk of septicemia is by the removal of catheters on time by following the survival sepsis campaign rules. (Murray, Rosenthal and Pfaller, 2005)
Bacterial infection that occurs due to septicemia, converse to an amplified hazard of cancer. this indicates the importance of sepsis in the prognosis of cancer patients as due to their immunosuppressant immune system their health is in risk to not only the initial disease of cancer but also of bacterial infection that can be lethal. (Anderson and May, 2010), the survival rate is directly linked to the prevention of the infection and the managements of its complication if it occurs with. (Gedik et al., 2014)
Coagulase negative staphylococci infections (CoNS)
CoNS are considered one of the leading causes of infections in hospitals and there are often isolated in clinical microbiology laboratories. It has been suggested since the 70s that CoNS are important pathogens that have the ability to cause serious infections with infections usually occurring in particular group of patients such as patients suffering from neutropenia and of patients that had to use indwelling foreign devices such as catheters. (Sydnor and Perl, 2011)
In hematologic malignancies, one of the most serious complications that can occur is bacterial infections and the corresponding treatment to eliminate them. (Tessier and D. Sifri, 2011)
When looking at hematology wards and infection, graph 1 summarizes the type of pathogens found in hematology wards. This is for a period from 1967 – 2013, covering data from 34 countries. Graph 2 indicates the source of these. All Data was obtained from Outbreak-database.
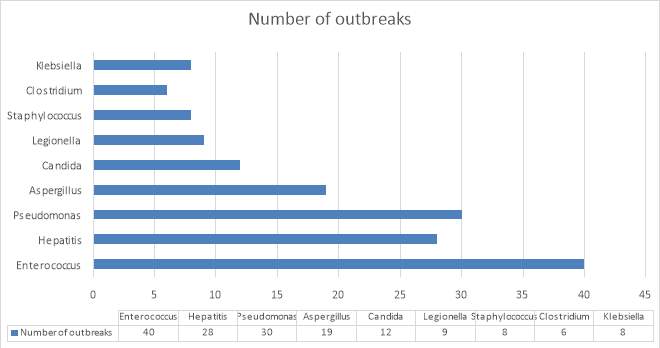
Graph 1 – number of outbreaks measured against different pathogens. – created from statistics of outbreak
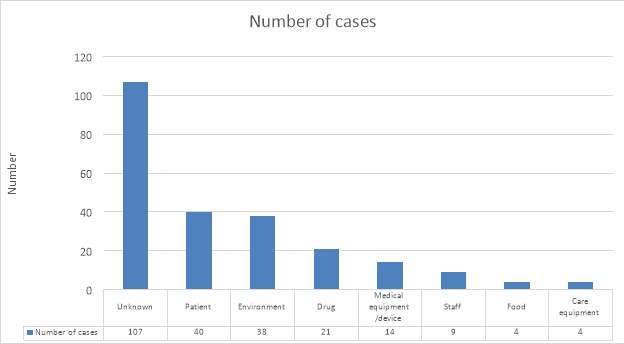
Graph 2 – number of cases contributed to different factors effecting infection in patients – created from statistics of outbreak
In order to secure the survival of these hematological cancer patients it is important to prevent antibacterial infections. The main source of infection here appears to be CoNS. (Zakhour, Chaftari and Raad, 2017).
CoNS are progressively being recognized as major nosocomial infections with one of their basic characteristics being their resistance to many antimicrobial agents that are regularly used as treatment for staph infections. (Y. Peleg and Hooper, 2011)
The bacteria that are found to be responsible for most of the infections in the population group of hematological malignancies develop during the introduction of both preventive and preemptive therapies of blood cancers for example the introduction of dwelling foreign devices such as catheters.
This catheter-related sepsis or infections by CoNS are known to be resistant to methilicin. (Gahlot et al., 2017)
Avoidance of bacterial infection in hematological malignancies patients throughout cancer treatment can be seen table 3.
Table 3 – methods avoiding infection in hematological malignancies patients (created from (Ruhnke, 2014)

The usage of catheters during treatment of patients with hematological malignancies and stem-cell transplants are important, however this put them at high risk for catheter-related bloodstream infections, leading to illness, death and high cost of care. (Zakhour, Chaftari and Raad, 2017)
The microorganisms can infiltrate onto the catheter through skin, via direct contact or contamination. (Zakhour, Chaftari and Raad, 2017) Furthermore, small thrombi can be formed as the result of micro trauma during catheter placement, creating another breeding ground for bacteria. (Zakhour, Chaftari and Raad, 2017)
Staphylococcus epidermidis (S. epidermidis)
Common in people that use catheters such as hematological malignancies patients are the CoNS and in particularly the S. epidermidis which is mainly found as part of the skin microflora but can also be found as part of the endogenous flora. (O’Brien et al, 2017).
Staphylococcus are a genus of gram and catalase positive and appear as single or and in irregular clusters looking like grapes hence the origin of their name. (Shivaprakasha, 2017) There are two groups of interest in this study, these that coagulase (such as S.aureus) are considered coagulase positive and these that don’t coagulase negative. (Fidanoski.ca, 2017) Throughout time S.aureus has been considered one of the most serious infections that can be caused in humans though Coagulase negative staphylococci have for the past few decades rise as a factor of infections in humans. (Fidanoski.ca, 2017) S.epidermidis is part of the microflora with suggestions that it might play a role as a probiotic manner and with this way being able to prevent the colonization of bacteria that appear more pathogenic such as S.aureus with no clear evidence existing to support this theory with information regarding S.epidermidis as a non-pathogenic nature being limited.
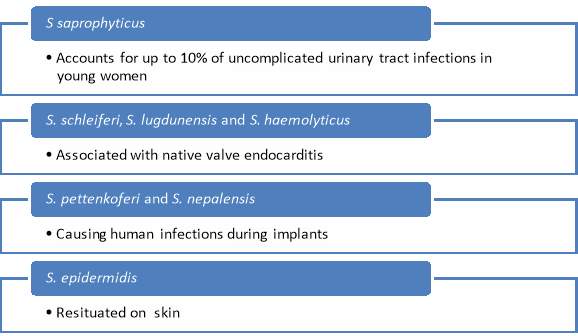
Table 4 – illustrate the different CoNS types and the infections that they play an important role in (created by Spyridoula Trakaki) (Fidanoski.ca, 2017)
Studies have shown that role of S.epidermidis as part of the commensal environment; maintain the equilibrium of the epithelial flora and to serve as a reservoir for resistant genes. Although S.epidermidis usually are in abundance on the human skin as part of harmless commensal bacterium., under specific circumstances though like when there is an implantation of a medical device S.epidermidis has the ability to become pathogenic. (Battner, Mack and Rohde, 2015) This causes challenges to isolates invasion. Reason being that a line needs to be drawn between what is clinical relevant and what is contaminations. S.epidermidis is seen as one of the most important bacteria related to hospital infections, especially for people that has been infected by HIV, bone marrow or organ translation, medical device implantation and chemotherapy related neutropenia. (Battner, Mack and Rohde, 2015)This research shows that S.epidermidis can interfere with the colonization mechanisms of S. aureus, e.g., biofilm formation.
In this context, this paper will look at S.epidermidis, as it is the main specie of CoNS causing infection in humans related to skin. (Battner, Mack and Rohde, 2015). The main difference is that the virulence factor associated with S. epidermidis has the ability to form biofilm and colonize biomaterials. (Fey and Olson, 2010) Biofilms are multifaceted interaction of unicellular organisms that are covered in a mixture of nucleic acid protein and extracellular matrix of polysaccharide with the biofilm. (Battner, Mack and Rohde, 2015)
S.epidermidis as an opportunistic pathogen is seen to cause infection in patients with an immunocompromised immune system (neonatal, receiving therapy that causes immunosuppressive results such as cancer therapy (chemotherapy, patients with AIDS etc.) (LIindsey, 2008) and rarely cases infection in healthy individuals.
Intravascular catheters are used in hospitalized patients for fluid administration medications, parenteral nutrition, and blood products; to monitor hemodynamic status; and finally for hemodialysis. This use of intravascular catheters however is linked with the risk of central line–associated bloodstream infection (CLABSI). This infection causes a biofilm formation. (Donlan, 2011)
The biofilm formed, play an important role also in infections of ocular matter and are being observed on abiotic material such as glaucoma tubes, stents, punctual plugs, corneal sutures. Patients that are infected by S.epidermidis can develop septicemia, endocarditis, cystic fibrosis pneumonia, periodontitis, chronic rhinosinusitis, and otitis media. (Morgenstern et al., 2016)
Catheter biofilms
Biofilm may act as a barrier between cellular and immune functionality, therefore inhibit the absorption of antibiotics. S.epidermidis biofilm, in specific forms a resistant type of biofilm. Once this biofilm is formed, it also contributes to the persistence of other pathogens such as Candida albicans (Neal, 2001) Biofilms are determined as are multicellular, surface-attached agglomerations of microorganisms with a specific type of physiology and structure that aids to the biofilm characteristic of showing resistance to a variety of antibiotics. (Meskin, 1998)As biofilm contains genes that are antibiotic resistant, with research showing that it might have the ability to cause chronic illness, treatment is difficult. (Otto, 2009)
This infection is called catheter colonization and is seen as infection when the measures are at >15 colony-forming units (CFU) in semi quantitative culture or >100 CFU in quantitative culture. (Trecarichi et al., 2017) This formation of colonies, leads to the formation of a biofilm from bacteria of the skins normal flora or a pathogenic organism. Crystal deposits instigating from flushed fluids may further assist attaching of bacteria to the catheter surface. The biofilm that surrounds the microorganisms protects it from the host immune system defense system and from antibiotics.
Plasmid pattern analysis on CoNS showed mucosa known as a biofilm in 70% of hematology patients within 2 weeks after the catheter introduction. (O’Brien et al., 2017)
The steps of the formation of biofilm can be seen in Diagram 4.

Diagram 4 – Biofilm development in Staphylococcus epidermidis. The steps include: step 1 – attachment to uncoated material due to hydrophobicity. Step 2 – After adhesion to the surface, exopolysaccharide (for example, poly-N-acetylglucosamine (PNAG), also known as PIA), Bap (Bhp and Aap) and teichoic acids help in intercellular aggregation. Step 3 – Maturation and structuring. Step 4 – detachment. For Steps 3 -4, quorum sensing-controlled expression of detergent-like peptides and proteolytic activity is detected. (Information from (Moormeier and Bayles, 2017)
Antibiotic resistance
According to Ventola (2015), worldwide bacteria are becoming rapidly resistant, endangering the efficiency of the effectiveness of antibiotics, which over the past decades have saved many lives. Shortly after the first person was cured with antibiotics, the first signs of resistance started; with bacterial infections threaten the health of individuals, with even more bacteria gaining resistance to antibiotics the last decades. Research indicated that the unsuitable prescription of antibiotics reaches 30-50 % of all antibiotics prescribed. This can be due to the influence on humans via the wide-ranging usage of antibiotic agriculturally, the decrease and lack of funding in antibiotic development and research. (Ventola, 2015)
Glycopeptides
Glycopeptides are the antibiotics used to treat gram positive bacterial infection with the 2 most known glycopeptide antibiotics being Teicoplanin and Vancomycin. In general the susceptibility degree of Gram-positive bacteria is 97.4% to vancomycin and 94.2% to Teicoplanin. (Trecarichi et al., 2017)
The way that glycopeptide antibiotics work is by inhibiting the synthesis of the cell wall by creating complexes with the peptidyl-D-alanyl–D-alanine (D-Ala–D-Ala) which is a termini of the peptidoglycan precursors located at the cell surface. (Courvalin, 2016) Furthermore resistances to glycopeptide antibiotics are mostly due to 2 gene clusters known as vanA and vanB, which are further discussed in the in silico part (See Appendix A). (Courvalin, 2016)
Resistance occurs due to the formation of the peptidoglycan precursors ending in the depsipeptideD-alanyl–D-lactate (D-Ala–D-Lac) which connects with weak affinity to the glycopeptides. (Arthur, Molinas and Courvalin, 1992)
Teicoplanin vs Vancomycin – why do we use Teicoplanin?
Although Teicoplanin and Vancomycin are similar in structures, they are different at antimicrobial and pharmacokinetic level, Teicoplanin has the lipophilic activity. (Newsom, 1996) Teicoplanin is a glycopeptide antibiotic that has been associated with successfully treating infectious diseases that are linked to coagulase negative staphylococci infections. (Smith et al., 1989) Teicoplanin, a lipoglycopeptide, is regularly compared to Vancomycin, a glycopeptide antibiotic that is used to treat caused by invasive beta-lactam-resistant gram-positive organisms. (Smith et al., 1989) For more information regarding Teicoplanin refer to Appendix B for the related leaflet of Targocid.
Dosages of these glycopeptides need to be monitored for renal toxicity. One daily dosage for uncomplicated infections and for more complex infections, higher dosages are given. (Newsom, 1996)
Glycopeptide antibiotics are used as the initial treatment for febrile neutropenic patient. In addition they are used as a second line agent when a patients fever don’t seem to response to the antibiotics that where given primarily as treatment. (Smith et al., 1989) A culture test needs to be done to determine if the infection of the biofilm formation on the catheter or septicemia is due to gram-positive or gram-negative bacteria.
Gram positive isolates in general are susceptible to Teicoplanin but some isolates that can infect patients becoming potentially a risk to the patient for example, S. haemolyticus can show some resistance to Teicoplanin although it has been showed that Teicoplanin may still be successful in cases as such. (Newsom, 1996)
Although Vancomycin is the most frequently used glycopeptide it’s required to be given many times a day which can lead to pruritus or even hypotension also known in this case as red man syndrome with signs of synergistic nephrotoxicity. (Vancomycin associated with hypersensitivity reactions, 2016)
The drug of interest in this study is Teicoplanin, as it is as an empirical treatment in blood cancer patients that have bacterial infection in Gwynedd hospital .Teicoplanin is administered as a once-daily dose and thus have lower chance to create nephrotoxicity. Because of this Teicoplanin serum concentration assays are not usually routinely needed. (Yano et al., 2007)
When Teicoplanin and Vancomycin effectiveness where compared in the treatment of infections in hematology and oncology patients, using Teicoplanin seems beneficial for the patient as it is easy to administrate, without routine serum monitoring and reduce rick of nephrotoxicity due to the low dosage given to the patients. (Yano et al., 2007) In cases where patients develop side effects while been treated with Teicoplanin such as rashes then Vancomycin will be administered instead and vice versa.
The use of Teicoplanin and Vancomycin is used as part of the initial management of clinically infected right atrial catheters in patients with malignancy. (Yano et al., 2007)
.
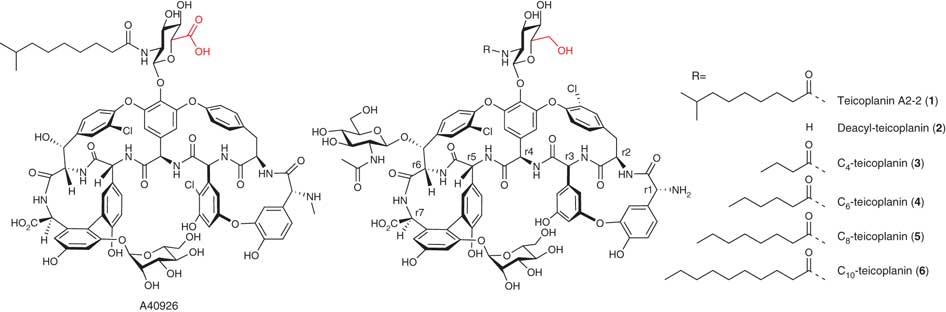
Diagram 3 – Structures of relevant glycopeptide antibiotics – with glucosamine and aliphatic acid as the main distinction of Teicoplanin (Liu et al., 2011)
A summary of both Teicoplanin and Vancomycin is made in table 4 within regards the mode of action, the side effects of the 2 glycopeptides as well as their cellular target and their medical purpose.
Table 4 – information regarding the 2 glycopeptide antibiotics ;Teicoplanin and Vancomycin information by Wilson and Grüneberg (1994), AUBERT et al., (1990) and Petersen et al., (1993)
| Teicoplanin | Vancomycin |
| Once-daily bolus administration | Constantly monitor of serum concentrations |
| Intravenously or intramuscularly | |
| A difference between Vancomycin and Teicoplanin lies within their diverse side-chain linkage patterns and carbohydrate groups | |
| A lower rate of side effects | |
| Teicoplanin has also been used to treat at home patients suffering from endocarditis specifically endocarditis caused by streptococcus | Treatment sometimes leads to the increased duration of systemic antibiotic treatment and thus postponing the cyclic parenteral nutrition |
| Teicoplanin is used for treatment of respiratory tract infections, infections linked to catheters and soft tissues | |
| Teicoplanin was given to patients as home therapy so they wouldn’t have to be hospitalized. (Hui et al., 2014) | |
| Teicoplanin is showed to be successful in treating infections caused by staphylococcus caused by venous catheter sepsis. | Not commonly used in cases of treating infection of the skin and soft tissue and infections of the bones and joints |
| It was shown that treatment with combination of Teicoplanin and Vancomycin or Teicoplanin and linezolid have more influence over the treatment process | |
| Teicoplanin inhibits more transpeptidation while | Vancomycin is more inclined to inhibit transglycosylation |
| The reason of its success as an antibiotic lays upon its carbon chain length | |
| More nephrotoxic than Teicoplanin | |
| Overall side effects including red man syndrome and skin rashes | |
| Is used as alternative therapy to Vancomycin only in Europe | |
Summary of Antibacterial Activity of Teicoplanin
In the point of view of Teicoplanin antibacterial activity it is shown in studies that streptococci in general appear sensitive to Teicoplanin with varying relative susceptibility have been shown from CoNS towards both Teicoplanin and Vancomycin with clostridium species such as C. diffilce and C.perfringens, appear to be sensitive to even low concentrations of Teicoplanin.(de Lalla et al., 1992) Teicoplanin is seen to be more tolerant, more effective for secondary treatment of patients who had a negative response to experimental treatment with either ceftazidime alone or in combination with amikacin, piperacillin and amikacin, or one of the cephalosporin antibiotics with the combination of an aminoglycoside antibiotics. (Davies, 2009)
MIC/MBC ratio – heterogeneity
Both Vancomycin and Teicoplanin, standard therapies for staphylococcal bacteremia, has shown a tolerance to vancomycin and Teicoplanin in CoNS, staphylococci, S. aureus and other Streptococci species. (Gonzalez et al., 2013)
Complicated infection, causing antibiotic tolerance may be more difficult to treat, particularly when they cause toxicities such as endocarditis, meningitis, osteomyelitis or cause infections in immunocompromised patients. (Traczewski et al., 2017)
This cause in return disagreement regarding the suitable approaches in the calculation of tolerance in the clinical laboratory where the heterogeneity is calculated for a better and more suitable solution. (Gonzalez et al., 2013)
Minimum inhibitory concentrations (MICs) is the term used for the lowest concentration of antimicrobial that has the ability to inhibit the noticeable growth after incubation occurs overnight of an microorganism and the minimum bactericidal concentrations ( MBCs)shows the lowest concentration of an antimicrobial that has the ability to stop the organisms growth after sub-culture on to antibiotic free media. (Andrews, 2002)
In general MICs are being used more frequently than MBC. They are used in laboratories for diagnostic purposes usually to confirm when resistance is suspected, but also in research as a research tool in order to determine the in-vitro activity of new antimicrobials, and data from such studies have been used to determine MIC breakpoints. (Andrews, 2002) MBC are used mainly for isolates taken from the patients’ blood suffering from endocarditis. The golden standard to determine whether or not an organism is susceptible to antimicrobials is the MICs which are used to judge the performance of all other methods of susceptibility testing. Mics are used to confirm uncommon resistance. (Pankey and Sabath, 2004)
Using both MIC and MBC, calculates heterogeneity of the organism towards the antibiotic. (Determination of minimum inhibitory concentrations (MICs) of antibacterial agents by broth dilution, 2003) in the case when Teicoplanin is used to treat infections caused by CoNS, this measurement will help to determine the overall resistance. (Pankey and Sabath, 2004)
A value of equal or greater than 32 from the MBC/MIC indicates possible heterogeneity that will show an occurrence of resistance of the CoNS strains that are tested. This is due to the fact that subpopulation of artificial isogenic bacteria displays a weakness to Teicoplanin in the case of this study or any other antibiotic in general. (El-Halfawy and Valvano, 2017) This measurement can then be used as a baseline to find the correct concentration of antibiotic to be administered. Once the ratio is measured the baseline has to be calculated to find the right amount of antibiotic concentration to be able to differentiate between the sensitive and resistant isolates. (Pankey and Sabath, 2004)
This study took 20 strains of CoNS that were isolated from oncology patients’ blood culture at the Gwynedd hospital in order to measure the ratio of MIC/MBC, if the ratio shows an equal or higher value of 32, then there is potentially heterogeneity.
Materials and Methods
Due to time constrictions the methodology was a 5 day procedure. 20 CoNS strains are tested with each strain being duplicated which results in a total of 40 strains, in order to have more accurate results by comparing the 2 tests done for each strain. The procedure was done twice for convenience and in order to avoid human errors such as contamination, so 10 strains with their duplicates the first 5 days and the other 10 strains with their duplicates the other 5 days. A control organism was introduced, which in this case, was the Staphylococcus aureus was used.
Day 1:
The first day consists of the broth making and the sub culturing of the strains (in the particular study. Each week 10 strains where tested together with their duplicate that would be tested onto agar plates)
Inoculation of the control organism which was the Staphylococcus aureus organism was done the next day with the other 10 strains and their duplicates in 10 ml of LB broth, each in sterile universal containers following overnight incubation at 37 degrees.
The broth is done for a 1 Liter and consisted of 10g of tryptone, 5g of yeast extractor, 10g of Nacl mixed thoroughly up until homogeneous with 1L of distilled water. Once this step is done the broth solution had to be done and incubated for at least 6-9 hours.
The Subculture of organism onto plates is done in order to isolate the bacteria and proceed the next day.
Antibiotic stock making is done by diluting the antibiotic of Teicoplanin in order for the solution that is being used to contain 10 x the required concentration upon the 1st tube.
400mg/3ml=133.3 μg/ml
133.333mg/ml requires 1280 μg/ml
The dilution needs to occur by 104.16 (133.333/1280)
1ml of Teicoplanin 99ml of distilled water=1333 mg/ml
1ml Teicoplanin 103 ml =1280 μg/ml (103ml) are split into 10 ml tubes and are frozen in order to be used when required
Day 2:
Inoculation of the control organism Staphylococcus aureus and of the strains that will be tested into 10 ml of broth in sterile universal container and incubation is followed overnight at 37 degrees
The tubes that are used for MIC are labelled to be used for the MIC measurements.
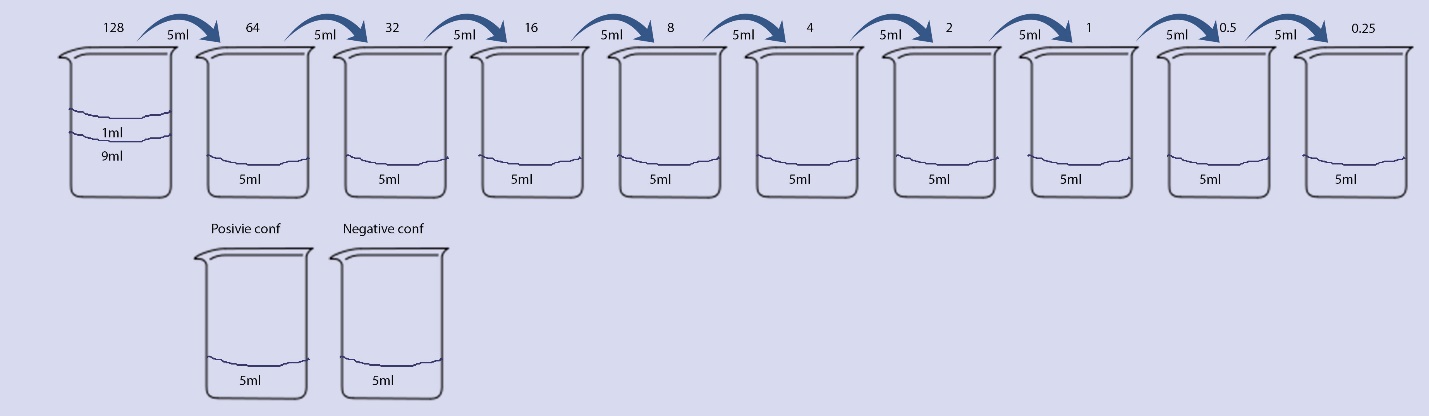
Sketch 1 – 12 sterile universal containers are used for each organism. Since each organism is duplicated is duplicated 24 containers for each strain is used. In each of the 128 μg/ml tubes, 9mls of the LB is inserted and in the rest of the tubes 5 ml are inserted. In the first tube of 128 μg/ml, 1ml of Teicoplanin is inserted (1024 ug/ml) and then by using a 5000 pipette mix tube 1 of 128 μg/ml is mixed and 5 mls are taken and placed into tube 2 which then have 512 ug/ml of Teicoplanin. The same procedure is repeated with the mixture and extraction of 5ml and then once again placed onto the next tube, tube 3. The same procedure occurs up until the last tube is reached of 0.25 μg/ml, where the 5ml extraction is disposed.
Day 3:
12 sterile universal containers are used for each organism. Since each organism is duplicated is duplicated 24 containers for each strain is used. In each of the 128 μg/ml tubes, 9mls of the LB is inserted and in the rest of the tubes 5 ml are inserted. In the first tube of 128 μg/ml,1ml of Teicoplanin is inserted (1024 ug/ml) and then by using a 5000 pipette mix tube 1 of 128 is mixed and 5 mls are taken and placed into tube 2 which then have 512 ug/ml of Teicoplanin. The same procedure is repeated with the mixture and extraction of 5ml and then once again placed onto the next tube, tube 3. The same procedure occurs up until the last tube is reached of 0.25 μg/ml, where the 5ml extraction is disposed. This procedure can be seen in sketch 1. Once this step is completed a 100ul of the broth dilution each corresponding strains is added into all the tubes (1-12 and the positive control tube) each organism has a positive control and a negative control where 5 ml of LB is placed. The positive control has 100ul of the organism whereas the negative has only the broth. This way there controls can specify if the batch that was done were contaminated or not. All the tubes are incubated overnight at 37 degrees.
Day 4:
The MIC is recorded by placing all the tubes in a row and by eye detecting the tubes with no visible growth. The tubes that appear not to have visually any growth are subculture into a blood agar plate which is divided in 4 areas. A 1ul loop is used to mark each of the quarters with the concentration. The control organism is also sub cultured (Staphylococcus aureus) upon the agar plates. The plates are then incubated overnight at 37 degrees C
Day 5:
The lowest concentration appearing without any growth is recorded in order to calculate the MBC. At this point all the strains are tested and the MBC/MIC ratio is calculated with any results appearing equal or greater than 32ug/ml being potentially heterogenic.
Picture – 1 illustrates the method that the MIC was recorded with the tubes being placed in a row in order to visually detect any tubes with no noticeable growth.

 Picture 2 MBC: below illustrates how the plates where divided in 4 sections
Picture 2 MBC: below illustrates how the plates where divided in 4 sections
Strain 17 MBC appearing to have growth

Picture 3 illustrate the wrapping of the agar plates with plastic wrap in order to be placed into the incubator where is will stay overnight at 37 degrees C
Picture 4 shows strain 17 MBC appearing to have growth
Results
The MIC is carried out to see if there is any peculiar resistance, to give answers when other tests give unclear answers or marginal results regarding resistance or in cases where the method of disc diffusion can’t be used like such in the case of coagulase negative staphylococci determination of susceptibility towards Teicoplanin as when staphylococci are tested using disc diffusion they do not give dependable results (Andrews, 2002)
The MIC dilution test, examines to see which strains where capable to grow in the broth dilution which contained an antimicrobial agent, which in this case is Teicoplanin. The lowest concentration of Teicoplanin that stops the visible growth of the CoNS strain is known as the organisms MIC measurement. (Rodríguez-Tudela et al., 2003)
Table 5 illustrates the results obtained from doing the MIC upon the selected 20 CoNS strains alongside the duplicated results with the selected antimicrobial agent of this study, Teicoplanin
| Organism | First clear tube | Positive control | Negative control |
| 1 | 0,5 | + | – |
| 1b | 0.25 | + | – |
| 2 | 0.25 | + | – |
| 2b | 0.25 | + | – |
| 3 | 0.5 | + | – |
| 3b | 0.25 | + | – |
| 4 | 0.5 | + | – |
| 4b | 0.5 | + | – |
| 5 | 1 | + | – |
| 5b | 0.5 | + | – |
| 6 | – | – | |
| 6b | – | – | |
| 7 | 1 | + | – |
| 7b | 0.5 | + | – |
| 8 | 0.5 | + | – |
| 8b | 0.5 | + | – |
| 9 | 0.25 | + | – |
| 9b | 0.25 | + | – |
| 10 | 1 | + | – |
| 10b | 1 | + | – |
| 11 | 0.5 | + | – |
| 11b | 1 | + | – |
| 12 | 2 | + | – |
| 12b | 1 | + | – |
| 13 | 0.25 | + | – |
| 13b | 0.25 | + | – |
| 14 | 1 | + | – |
| 14b | 0.5 | + | – |
| 15 | 0.25 | + | – |
| 15b | 0.25 | + | – |
| 16 | 0.5 | + | – |
| 16b | 0.5 | + | – |
| 17 | 4 | + | – |
| 17b | 4 | + | – |
| 18 | 0.25 | + | – |
| 18b | 0.25 | + | – |
| 19 | 0.25 | + | – |
| 19b | 0.25 | + | – |
| 20 | 0,5 | + | – |
| 20b | 0.5 | + | – |
The MBC calculated the lowest level of concentration of Teicoplanin that will stop the growth of the CoNS after their culture upon agar plates. The MBC are not done as frequently as the MIC and are usually used more commonly in for the blood isolates of endocarditis patients.
Table 6 illustrates the MBC results obtained upon the selected 20 CoNS strains alongside the duplicated results with the selected antimicrobial agent of this study, Teicoplanin
| Organism | First clear section in agar blood plate |
| 1 | 1 |
| 1b | 2 |
| 2 | 0.5 |
| 2b | 0.25 |
| 3 | 1 |
| 3b | 0.5 |
| 4 | 1 |
| 4b | 2 |
| 5 | 1 |
| 5b | 1 |
| 6 | |
| 6b | |
| 7 | 2 |
| 7b | 2 |
| 8 | 2 |
| 8b | 0.25 |
| 9 | 0.5 |
| 9b | 0.5 |
| 10 | 4 |
| 10b | 2 |
| 11 | 1 |
| 11b | 1 |
| 12 | 4 |
| 12b | 4 |
| 13 | 0.25 |
| 13b | 2 |
| 14 | 1 |
| 14b | 0.5 |
| 15 | 0.25 |
| 15b | 0.25 |
| 16 | 1 |
| 16b | 2 |
| 17 | 4 |
| 17b | 4 |
| 18 | 0.25 |
| 18b | 0.25 |
| 19 | 0.25 |
| 19b | 1 |
| 20 | 1 |
| 20b | 1 |
Table 7 illustrates the results obtained after completing the MIC and MBC and the equation of MBC/MIC on the CoNS strains and on the control organism which was Staphylococcus aureus to determine possible heterogeneity if the ratio appears to be more than 32. The organism on Sample 6 died hence why no measurements had been taken. Briefly observing table 3, it can be observed that there is no strain at the MIC/MBC ratio which surpasses or is equal to 32.
| Organism | MIC results μg/ml | MBC results μg/ml | MBC/MIC ratio |
| Staphylococcus aureus ( control organism)NCRC strain | 0.25 | 0.25 | 1 |
| 1 | 0,5 | 1 | 2 |
| 1b | 0.25 | 2 | 8 |
| 2 | 0.25 | 0.5 | 2 |
| 2b | 0.25 | 0.25 | 1 |
| 3 | 0.5 | 1 | 2 |
| 3b | 0.25 | 0.5 | 2 |
| 4 | 0.5 | 1 | 2 |
| 4b | 0.5 | 2 | 4 |
| 5 | 1 | 1 | 1 |
| 5b | 0.5 | 1 | 2 |
| 6 | |||
| 6b | |||
| 7 | 1 | 2 | 2 |
| 7b | 0.5 | 2 | 4 |
| 8 | 0.5 | 2 | 4 |
| 8b | 0.5 | 0.25 | 0.5 |
| 9 | 0.25 | 0.5 | 2 |
| 9b | 0.25 | 0.5 | 2 |
| 10 | 1 | 4 | 4 |
| 10b | 1 | 2 | 2 |
| 11 | 0.5 | 1 | 2 |
| 11b | 1 | 1 | 1 |
| 12 | 2 | 4 | 2 |
| 12b | 1 | 4 | 4 |
| 13 | 0.25 | 0.25 | 1 |
| 13b | 0.25 | 2 | 8 |
| 14 | 1 | 1 | 1 |
| 14b | 0.5 | 0.5 | 1 |
| 15 | 0.25 | 0.25 | 1 |
| 15b | 0.25 | 0.25 | 1 |
| 16 | 0.5 | 1 | 2 |
| 16b | 0.5 | 2 | 4 |
| 17 | 4 | 4 | 1 |
| 17b | 4 | 4 | 1 |
| 18 | 0.25 | 0.25 | 1 |
| 18b | 0.25 | 0.25 | 1 |
| 19 | 0.25 | 0.25 | 1 |
| 19b | 0.25 | 1 | 4 |
| 20 | 0,5 | 1 | 0.5 |
| 20b | 0.5 | 1 | 0.5 |
Discussion
The results obtained from the MIC and MBC ratio, were used to calculate the possible heterogeneity of the CoNS strains that where tested. Results of MBC/MIC ratio of higher or equal value than 32, indicates heterogeneity. Out of the 20 strains that were tested for possible heterogeneity none of them had shown a sign of possible heterogeneity, turning the theory of this study into a null hypothesis as the initial hypothesis was not proven true.
The positive outcome of this study is that the patient of whom the strains are taken from will be able to be treated with Teicoplanin, by given the standard dosage of 6mg/L without any suspected complications during treatment since all strains tested resulted as sensitive towards Teicoplanin.
The heterogeneity results of the study ranged from 0.5 to 8 µg. This is an indication that the test cases delivered no possible heterogeneity. When comparing the MIC results of the study with the EUCAST (Matuschek, 2017) results of glycopeptides and particularly Teicoplanin used upon coagulase negative staphylococci, the MIC breakpoint states that sensitivity is achieved with results equal and less than 4mg/L and results above 4mg/L indicates resistance. These results of sensitivity and resistance are identical with Vancomycin, indicating once again the similarity of effect of the 2 glycopeptides. When these MIC results were compared to the study done we observed that none of the 20 strains that had higher values than 4 mg/L resulting in all 20 strains of the study fall under the sensitivity range.
When taking in consideration published papers of similar nature we see the worldwide concern of the upcoming resistant that bacteria have towards glycopeptides. One study (Ma et al., 2011) indicated the increase of antimicrobial resistance rates on CoNS, specifying the increased resistance of CoNS towards Teicoplanin. In particular an increase from 4.5 to 6.7 % between 2008 and 2009 of the MIC values were observed (the test indicated six CoNS strains with MIC values of 16 and 64 mg ml in 2008; seven CoNS strains with a MIC value of 128 mg ml in 2009). Each year a steady increase is observed which is of serious concern for the medical treatment of these strains. (Tor and Fair, 2014)
A study undertaken by a Chinese medical university (Ma et al., 2011) indicated that the upcoming resistance of Teicoplanin among the CoNS strains could due to the selective pressure from the regular use of Vancomycin. Out of the 12 patients receiving Vancomycin, 8 received b-lactam antibiotics before or concurrently with Vancomycin, validating the likelihood that preceding use of b-lactams might help to induce the manifestation of vancomycin resistance.
Another study that was carried out over a period of 8 months in North India to test the occurrence and antimicrobial susceptibility pattern of Staphylococci causing blood stream infection, resulted that out of the 157 infected with bacteremia, 77 of them was caused by gram positive bacteria with staphylococci being responsible for 74 of them . Out of these results only 4 strains of CoNS appear to be sensitive with only one being totally resistant towards Teicoplanin. Interestingly enough, it seems that all CoNS appear to be sensitive to Vancomycin. (Ahmed, Baruah and Grover, 2015)
Another study deducted from 240 surgical events from a tested or distrusted device-associated with bone and joint infections, isolated 315 CoNS strains with 16 strains indicating resistance to glycopeptides. Fifteen of them showed resistance to Teicoplanin alone and only one to both. There was a 100% decreased in the susceptibility of strains taken from S.epidermidis towards Teicoplanin. (Cremniter et al., 2017)
This information is problematic as S.epidermidis as pre-mentioned is one of the leading causes of biofilm formation on catheters in cancer patients. (Faller and Kohler, 2017)
Recent studies in Italy that where undertaken by Trecarichi et al., (2009), regarding epidemiologically and antimicrobial resistance for bacterial infections of the bloodstream in patients with blood cancer with hematologic malignancies. The study was conducted in 9 hematological wards with all the bacterial infections of the bloodstream being in patients with blood cancer with the majority of the patients being neutropenic .The overall results showed that the susceptibility rates of gram positive bacteria indicates 97.4% to vancomycin and 94.2% to Teicoplanin.
Gram negative bacteria though appears to be more frequent in microorganisms that were isolated form infections in patients with cancer which consistent with the information of reports indicating the shift of occurrence from Gram-positive to Gram-negative bacteria in serious bacterial infections in cancer patients
A literature review carried out by Mikulska et al , compared the resistance of 18 countries of patients with cancer with septicemia from 2005-2011 illustrated an important current new reduction in the Gram-positive to Gram-negative ratio illustrating the swap of “power “ and preference.
The Italian study (Trecarichi et al., 2009) conducted showed that the resistance of the CoNS strains towards Teicoplanin were lower than reported in the literature review of Mikulska et al.
The overall conclusion taken from comparing studies from all over the world is that depending on the area and study and depending on the time taken to do the research and the number of strains results vary from place to place with an important factor being as well the usage of the glycopeptides as well like in the case of the study done in China. (Ma et al., 2011)
With many studies indicating the increase of CoNS resistance towards glycopeptides (Sujatha and Praharaj, 2012) and as CoNS are the most predominant organism causing infection on medical devices (such as catheters) infection in immunosuppressant patients (Sydnor and Perl, 2011) and the study of antibiotic resistance and susceptibility is crucial from an epidemiological aspect as well as to offer guidelines for the use of antibiotics and for better and more efficient treatment by taking control measurements.
The overall picture of glycopeptide antibiotics and their susceptibility appear to range from country to country though the overall image obtained from studies around the world is that there is an increase in resistance of CoNS towards Teicoplanin and that it is of major concern. (Ma et al., 2011)
The conclusion is still in a lower or higher rate in some countries, that CoNS resistance is an occurrence that scientist are aware of and that caution measurements should be taken .
Within perspective, the reason behind the null hypothesis obtained in this study, could be accounted to possible human errors during testing and result calculation. As an undergraduate with a lack of experience in a microbiology lab and with delicate work and concentration needed with the specific techniques required to obtain the MIC and MBC results, it is not unlikely that mistakes could have occurred. Since all the procedures had to be carried out under sterile conditions contamination is very easy to occur even with an experience personnel. A wrong calculations could provide falsify results. Experience and practice is key in this study to successfully gain true positive results.
Also the time frame where the laboratory work was done was very limited, opening up the window for errors. A larger time frame would be desirable for not only testing a larger number of strains but at the same time have time to redo any experiments if contamination occurs without time pressure.
Further studies could isolate and test other strains as well of CoNS in order to check for possible heterogeneity. In future studies more strains could be used to check for heterogeneity and could be compared with previous years of the study done by the University in order to have an overview of the heterogeneity the last years drawing conclusions whether or not there is an increase in resistance in the strains taken by Gwynedd Hospital.
Acknowledgments
I want to add a special thanks to my supervisor, Dr. Merfyn Williams, his support, guidance and understanding motivated me to complete this thesis with excitement. It is because of him that I will continue and have an interest now in doing research.
References
- Ahmed, N., Baruah, F. and Grover, R. (2015). Staphylococcal blood stream infections in cancer patients. Annals of Medical and Health Sciences Research, 5(3), p.226.
- Anderson, R. and May, R. (2010). Infectious diseases of humans. 1st ed. Oxford: Oxford Univ. Press.
- Andrews, J. (2002). Determination of minimum inhibitory concentrations. Journal of Antimicrobial Chemotherapy, 49(6), pp.1049-1049.
- Andrews, J. (2002). Determination of minimum inhibitory concentrations. Journal of Antimicrobial Chemotherapy, 49(6), pp.1049-1049.
- Arthur, M., Molinas, C. and Courvalin, P. (1992). The VanS-VanR two-component regulatory system controls synthesis of depsipeptide peptidoglycan precursors in Enterococcus faecium BM4147. Journal of Bacteriology, 174(8), pp.2582-2591.
- Aubert, G., Passot, S., Lucht, F. And Dorche, G. (1990). Selection of vancomycin- and Teicoplanin resistant Staphylococcus haemolyticus during Teicoplanin treatment of S. epidermidis infection. Journal of Antimicrobial Chemotherapy, 25(3), pp.491-493.
- Barros, E., Ceotto, H., Bastos, M., dos Santos, K. and Giambiagi-deMarval, M. (2011). Staphylococcus haemolyticus as an Important Hospital Pathogen and Carrier of Methicillin Resistance Genes. Journal of Clinical Microbiology, 50(1), pp.166-168.
- Battner, H., Mack, D. and Rohde, H. (2015). Structural basis of Staphylococcus epidermidis biofilm formation: mechanisms and molecular interactions. Frontiers in Cellular and Infection Microbiology, 5.
- CancerCenter.com. (2017). Radiation Therapy Options for Cancer Patients | CTCA. [online] Available at: http://www.cancercenter.com/treatments/radiation-therapy/? [Accessed 6 Apr. 2017].
- Conrad, M. (2016). Leukemia: Symptoms, Survival Rate, and Diagnosis. [online] MedicineNet. Available at: http://www.medicinenet.com/leukemia/article.htm [Accessed 14 Mar. 2017].
- Costerton, J. (1999). Bacterial Biofilms: A Common Cause of Persistent Infections. Science, 284(5418), pp.1318-1322.
- Courvalin, P. (2016). Vancomycin Resistance in Gram-Positive Cocci.
- Cremniter, J., Slassi, A., Quincampoix, J., Sivadon-Tardy, V., Bauer, T., Porcher, R., Lortat-Jacob, A., Piriou, P., Judet, T., Herrmann, J., Gaillard, J. and Rottman, M. (2017). Decreased Susceptibility to Teicoplanin and Vancomycin in Coagulase-Negative Staphylococci Isolated from Orthopedic-Device-Associated Infections. [online] Jcm.asm.org. Available at: http://jcm.asm.org/content/48/4/1428.full?ck=nck [Accessed 6 Apr. 2017].
- Davies, J. (2009). A review of the use of Teicoplanin in haematological malignancy. European Journal of Haematology, 59(S62), pp.2-5.
- De Lalla, F., Nicolin, R., Rinaldi, E., Scarpellini, P., Rigoli, R., Manfrin, V. and Tramarin, A. (1992). Prospective study of oral Teicoplanin versus oral vancomycin for therapy of pseudomembranous colitis and Clostridium difficile-associated diarrhea. Antimicrobial Agents and Chemotherapy, 36(10), pp.2192-2196.
- Determination of minimum inhibitory concentrations (MICs) of antibacterial agents by broth dilution. (2003). Clinical Microbiology and Infection, 9(8), p.ix-xv.
- Donlan, R. (2011). Biofilm Elimination on Intravascular Catheters: Important Considerations for the Infectious Disease Practitioner. Clinical Infectious Diseases, 52(8), pp.1038-1045.
- El-Halfawy, O. and Valvano, M. (2017). Antimicrobial Heteroresistance: an Emerging Field in Need of Clarity.
- Emedicine.medscape.com. (2017). Enterococcal Infections: Background, Pathophysiology, Epidemiology. [Online] Available at: http://emedicine.medscape.com/article/216993-overview [Accessed 2 Mar. 2017].
- Enger, E. and Ross, F. (n.d.). Concepts in biology. 1st Ed.
- Faller, M. and Kohler, T. (2017). The Status of Biofilms in Penile Implants. Microorganisms, 5(2), p.19.
- Fey, P. and Olson, M. (2010). Current concepts in biofilm formation of Staphylococcus epidermidis. Future Microbiology, 5(6), pp.917-933.
- Fidanoski.ca. (2017). Staphylococcus Vs Streptococcus, a comprehensive analysis – Dr.B.Fidanoski, DMD. [online] Available at: http://www.fidanoski.ca/medicine/staphylococcus-streptococcus.htm [Accessed 12 Apr. 2017].
- Gahlot, R., Nigam, C., Kumar, V., Yadav, G. and Anupurba, S. (2017). Catheter-related bloodstream infections.
- Gedik, H., Şimşek, F., Kantürk, A., Yildirmak, T., Arica, D., Aydin, D., Demirel, N. and Yokuş, O. (2014). Bloodstream infections in patients with hematological malignancies: which is more fatal– cancer or resistant pathogens? PMC, 10, pp.743–752.
- Gonzalez, N., Sevillano, D., Alou, L., Cafini, F., Gimenez, M., Gomez-Lus, M., Prieto, J. and Aguilar, L. (2013). Influence of the MBC/MIC ratio on the antibacterial activity of vancomycin versus linezolid against methicillin-resistant Staphylococcus aureus isolates in a pharmacodynamic model simulating serum and soft tissue interstitial fluid concentrations reported in diabetic patients. Journal of Antimicrobial Chemotherapy.
- Hematology.org. (2017). Blood Cancers. [online] Available at: http://www.hematology.org/Patients/Cancers/ [Accessed 1 Apr. 2017].
- Henderson, I., Navarro-Garcia, F., Desvaux, M., Fernandez, R. and Ala’Aldeen, D. (2004). Type V Protein Secretion Pathway: the Autotransporter Story. Microbiology and Molecular Biology Reviews, 68(4), pp.692-744.
- Hui, D., Didwaniya, N., Vidal, M., Shin, S., Chisholm, G., Roquemore, J. and Bruera, E. (2014). Quality of end-of-life care in patients with hematologic malignancies: A retrospective cohort study. Cancer, 120(10), pp.1572-1578.
- Ko, A., Dollinger, M. and Rosenbaum, E. (2008). Everyone’s guide to cancer therapy. 1st ed. Kansas City: Andrews McMeel Pub.
- Kuzin, A., Sun, T., Jorczak-Baillass, J., Healy, V., Walsh, C. and Knox, J. (2000). Enzymes of vancomycin resistance: the structure of d-alanine–d-lactate ligase of naturally resistant Leuconostoc mesenteroides. Structure, 8(5), pp.463-470.
- Lindsey, H. (2008). Preventing Infection in Immunocompromised Cancer Patients. Oncology Times, 30(18), pp.25-26.
- Liu, Y., Li, Y., Lyu, S., Hsu, L., Chen, Y., Huang, Y., Chan, H., Huang, C., Chen, G., Chou, C., Tsai, M. and Li, T. (2011). Interception of Teicoplanin oxidation intermediates yields new antimicrobial scaffolds.
- Ma, X., Wang, E., Liu, Y. and Luo, E. (2011). Antibiotic susceptibility of coagulase-negative staphylococci (CoNS): emergence of Teicoplanin-non-susceptible CoNS strains with inducible resistance to vancomycin. Journal of Medical Microbiology, 60(11), pp.1661-1668.
- Matuschek, E. (2017). EUCAST: MIC distributions and ECOFFs. [online] Eucast.org. Available at: http://www.eucast.org/mic_distributions_and_ecoffs/.
- Megari, K. (2013). Quality of life in chronic disease patients. Health Psychology Research, 1(3), p.27.
- Meskin, I. (1998). Staphylococcus epidermidis. Pediatrics in Review, 19(3), pp.105-106.
- Michaud, L., Turck, D., Cuntz, D., Husson, M., Dubar, G. and Gottrand, F. (1998). Antibiotic lock technique with Teicoplanin for control of staphylococcus central venous catheter-related sepsis in children with long term home parenteral nutrition. Journal of Paediatric Gastroenterology & Nutrition, 26(5), p.593.
- Moormeier, D. and Bayles, K. (2017). Staphylococcus aureus biofilm: a complex developmental organism. Molecular Microbiology, 104(3), pp.365-376.
- Morgenstern, M., Post, V., Erichsen, C., Hungerer, S., Bühren, V., Militz, M., Richards, R. and Moriarty, T. (2016). Biofilm formation increases treatment failure in Staphylococcus epidermidis device-related osteomyelitis of the lower extremity in human patients. Journal of Orthopaedic Research, 34(11), pp.1905-1913.
- Murray, P., Rosenthal, K. and Pfaller, M. (2005). Medical microbiology. 1st ed. Philadelphia: Elsevier Mosby.
- National Cancer Institute. (2017). Plasma Cell Neoplasms (Including Multiple Myeloma) Treatment. [online] Available at: https://www.cancer.gov/types/myeloma/patient/myeloma-treatment-pdq [Accessed 16 Mar. 2017].
- Neal, C. (2001). Catecholamine inotropes as growth factors for Staphylococcus epidermidis and other coagulase-negative staphylococci. FEMS Microbiology Letters, 194(2), pp.163-169.
- Newsom, S. (1996). Susceptibility of coagulase-negative staphylococci to Teicoplanin. Clinical Microbiology and Infection, 2(3), pp.155-158.
- Niederhuber, J., Armitage, J., Doroshow, J., Kastan, M. and Tepper, J. (2013). Abeloff’s Clinical Oncology Review. 1st ed. London: Elsevier Health Sciences.
- Nolan, A. (2017). What is blood cancer? [online] Available at: https://www.anthonynolan.org/patients-and-families/blood-cancers-and-blood-disorders/what-blood-cancer [Accessed 16 Mar. 2017].
- O’Brien, S., Blijlevens, N., Mahfouz, T. and Anaissie, E. (2017). Infections in Patients with Hematological Cancer: Recent Developments. [online] Asheducationbook.hematologylibrary.org. Available at: http://asheducationbook.hematologylibrary.org/content/2003/1/438.full [Accessed 3 Apr. 2017].
- Otto, M. (2009). Staphylococcus epidermidis — the ‘accidental’ pathogen. Nature Reviews Microbiology, 7(8), pp.555-567.
- Outbreak-database.com. (2017). Outbreak Database – Home. [online] Available at: https://www.outbreak-database.com [Accessed 4 Apr. 2017].
- Pankey, G. and Sabath, L. (2004). Clinical Relevance of Bacteriostatic versus Bactericidal Mechanisms of Action in the Treatment of Gram‐Positive Bacterial Infections. Clinical Infectious Diseases, 38(6), pp.864-870.
- Petersen, D., Steffen, A., Bautsch, W., Ziesing, S. and Link, H. (1993). Therapeutic Drug Monitoring and in Vitro Activity of the Glycopeptide Antibiotic Teicoplanin. Therapeutic Drug Monitoring, 15(2), p.141.
- Rodríguez-Tudela, J., Barchiesi, F., Bille, J., Chryssanthou, E., Cuenca-Estrella, M., Denning, D., Donnelly, J., Dupont, B., Fegeler, W., Moore, C., Richardson, M. and Verweij, P. (2003). Determination of minimum inhibitory concentrations (MICs) of antibacterial agents by broth dilution. Clinical Microbiology and Infection, 9(8), p.ix-xv.
- Ruddon, R. (2007). Cancer biology. 1st ed. Oxford: Oxford University Press.
- Ruhnke, M. (2014). Infection control issues in patients with haematological malignancies in the era of multidrug-resistant bacteria. [online] Sciencedirect.com. Available at: http://www.sciencedirect.com/science/article/pii/S1470204514703444 [Accessed 10 Apr. 2017].
- Ruhnke, M., Arnold, R. and Gastmeier, P. (2017). Infection control issues in patients with haematological malignancies in the era of multidrug-resistant bacteria.
- Sepsis Alliance. (2017). Sepsis and Cancer – Sepsis Alliance. [online] Available at: http://www.sepsis.org/sepsis-and/cancer/ [Accessed 4 Apr. 2017].
- Shivaprakasha, S. (2017). Determination of Vancomycin, Teicoplanin and Linezolid resistance among Staphylococcal isolates from a tertiary care hospital.
- Skeel, R. and Khlief, S. (2015). Handbook of Cancer Chemotherapy. 1st ed. Philadelphia: Wolters Kluwer.
- Smith, S., Cheesbrough, J., Spearing, R. and Davies, J. (1989). Randomized prospective study comparing vancomycin with Teicoplanin in the treatment of infections associated with Hickman catheters. Antimicrobial Agents and Chemotherapy, 33(8), pp.1193-1197.
- Staudinger, T. and Pène, F. (2014). Current insights into severe sepsis in cancer patients. Rev Bras Ter Intensiva, 26(4), pp.335-338.
- Sujatha, S. and Praharaj, I. (2012). Glycopeptide Resistance in Gram-Positive Cocci: A Review. Interdisciplinary Perspectives on Infectious Diseases, 2012, pp.1-10.
- Sydnor, E. and Perl, T. (2011). Hospital Epidemiology and Infection Control in Acute-Care Settings. Clinical Microbiology Reviews, 24(1), pp.141-173.
- Sydnor, E. and Perl, T. (2011). Hospital Epidemiology and Infection Control in Acute-Care Settings. Clinical Microbiology Reviews, 24(1), pp.141-173.
- Tessier, J. and D. Sifri, C. (2011). Epidemiology and Prevention of Bacterial Infections in Patients with Hematologic Malignancies. Infectious Disorders – Drug Targets, 11(1), pp.11-17.
- Tor, Y. and Fair, R. (2014). Antibiotics and Bacterial Resistance in the 21st Century. Perspectives in Medicinal Chemistry, p.25.
- Traczewski, M., Katz, B., Steenbergen, J. and Brown, S. (2017). Inhibitory and Bactericidal Activities of Daptomycin, Vancomycin, and Teicoplanin against Methicillin-Resistant Staphylococcus aureus Isolates collected from 1985 to 2007.
- Trecarichi, E., Pagano, L., Candoni, A., Pastore, D., Cattaneo, C., Fanci, R., Nosari, A., Caira, M., Spadea, A., Busca, A., Vianelli, N. and Tumbarello, M. (2017). Current epidemiology and antimicrobial resistance data for bacterial bloodstream infections in patients with hematologic malignancies: an Italian multicentre prospective survey.
- Trecarichi, E., Pagano, L., Candoni, A., Pastore, D., Cattaneo, C., Fanci, R., Nosari, A., Caira, M., Spadea, A., Busca, A., Vianelli, N. and Tumbarello, M. (2017). Current epidemiology and antimicrobial resistance data for bacterial bloodstream infections in patients with hematologic malignancies: an Italian multicentre prospective survey.
- Vancomycin associated with hypersensitivity reactions. (2016). Reactions Weekly, 1603(1), pp.12-12.
- Ventola, C. (2015). The Antibiotic Resistance Crisis Part 1: Causes and Threats. P&T, Apr; 40(4), pp.277–283.
- Verheij, M. (2014). SP-0370: Radiotherapy. Radiotherapy and Oncology, 111, p.S143.
- Wilson, A. and Grüneberg, R. (1994). Use of Teicoplanin in community medicine. European Journal of Clinical Microbiology & Infectious Diseases, 13(9), pp.701-710.
- Y. Peleg, A. and Hooper, D. (2011). Hospital-Acquired Infections Due to Gram-Negative Bacteria. PMC, 13(362(19), pp.1804–1813.
- Yano, R., Nakamura, T., Tsukamoto, H., Igarashi, T., Goto, N., Wakiya, Y. and Masada, M. (2007). Variability in Teicoplanin Protein Binding and Its Prediction Using Serum Albumin Concentrations. Therapeutic Drug Monitoring, 29(4), pp.399-403.
- Zakhour, R. and Chaftari, A. (2017). Catheter-related infections in patients with haematological malignancies: novel preventive and therapeutic strategies. [online] Sciencedirect.com.
Appendix A
IN SILICO
Analysis of vanA and vanB
The analysis of vanA and vanB genes is conducted, in coagulase negative staphylococci species to identify them in silico part.
Both genes were selected for further investigation, due to the fact that they both contribute to the resistance towards glycopeptide antibiotics.
The creation of complexes with the peptidyl-D-alanyl–D-alanine (D-Ala–D-Ala), are attributed to Glycopeptide antibiotics functions, through the constraining of production of the cell wall.
Resistance contributes to the formation of the peptidoglycan precursors ending in the depsipeptideD-alanyl–D-lactate (D-Ala–D-Lac). This in return connects with weak attraction to the glycopeptides (Casadewall, Reynolds and Courvalin, 2001). This resistance is due to the fact that D-Ala– yields a peptidoglycan which does not terminate in D-alanine but in D-lactate, thus preventing vancomycin or Teicoplanin binding. (Kuzin et al., 2000)
By taking a deeper look into these two genes and comparing them to other species we can detect possible variations in the bacteria’s genome using the blast analysis that stands for Basic Local Alignment Search Tool. This tool allows us to compare similarities between sequences, comparing principal biological sequences of information (Blast.ncbi.nlm.nih.gov, 2016).
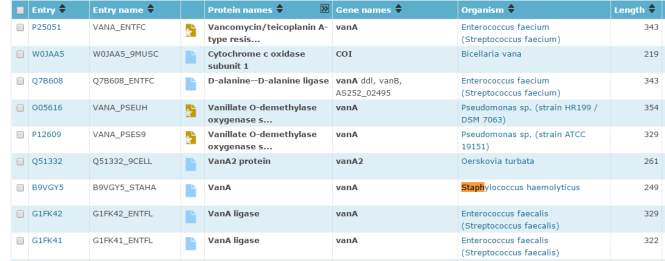
Figure 1 – illustrates the website Uniprot.org which was used to compares the different similarities between sequences
Vancomycin resistance protein (vanA)
The investigation started with the search of the gene vanA by using the uniprot.org website to compare the two genes to other species. The search revealed its protein name, the organism where it is located and the length of it as well as the entry name. (figure1) As we are interested in coagulase negative staphylococci, the vanA located in Staphylococcus haemolyticus was chosen as comparative organism as it is the second most common organism sequestered in human blood cultures is, having the uppermost level of antimicrobial resistance is Staphylococcus haemolyticus. (Barros et al., 2011)
VanA in enterococcus faecium was also chosen as its protein name Vancomycin/Teicoplanin A-type resistance protein VanA was of interest since the research is based upon Teicoplanin effect on coagulase negative organisms.
It was expected to see many Enterococcus faecium species as they are important human pathogens and are of major importance as they are most dominant cultures species taken from humans with at least 90% of clinical isolates from Enterococcus faecium being accountable for the majority of infections caused by vancomycin-resistant enterococci. (Emedicine.medscape.com, 2017)
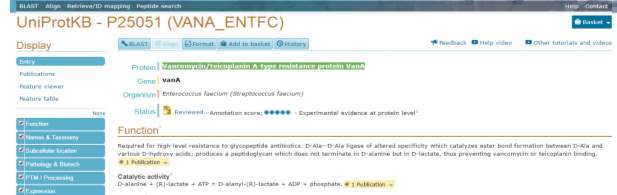
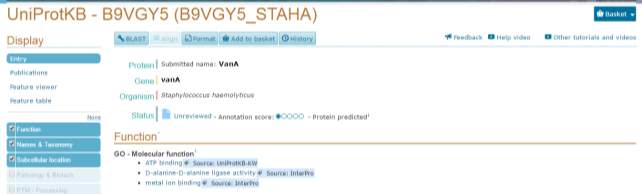
Figure 2 –indicates the organisms containing vanA that were chosen to be compared with other different sequence blast sequence
Once the blast was completed the first thing that was noticed is that vanA in Staphylococcus haemolyticus and in Enterococcus faecium are a 100% identical for the 249 residues that are identical between the 2 comparison organisms as can be seen in figure 3.
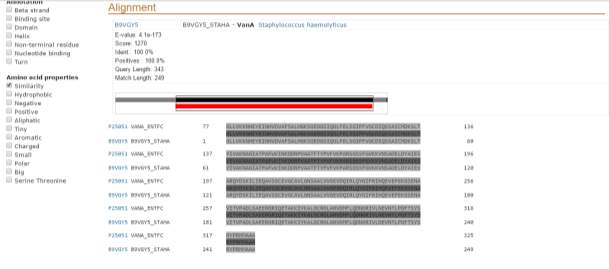
Figure 3: illustrates the comparison of vanA in Staphylococcus haemolyticus and in Enterococcus faecium showing a 100% similarity rate for the 249 residues that are identical between the 2 comparison organisms. The total size of the Enterococcus faecium is 343.There is another 100 amino acid present in the Enterococcus faecium that is not found in Staphylococcus haemolyticus
When VanA in enterococci was compared to other organisms it showed as expected a lot of similarities with vanA in other enterococci organisms. (Figure 4) As we are searching to compare it to coagulase negative staphylococci and more specific to Staphylococcus epidermidis as it is the main opportunistic nosocomial pathogen associated mainly with foreign body infection (Costerton, 1999) it was noticed that there appeared to have no Staphylococcus epidermidis in the data base and it was observed that the only CoNS that was in the data base was Staphylococcus haemolyticus being a 100% identical.
Next it was compared to Staphylococcus aureus as Staphylococcus aureus is seen to be the organism in the staph family causing the most infections and are part of the methicillin-resistant Staphylococcus aureus (MRSA)
It was found in 6 different Staphylococcus aureus organisms but strains with all of them being a 100% identical. (Figure 5)
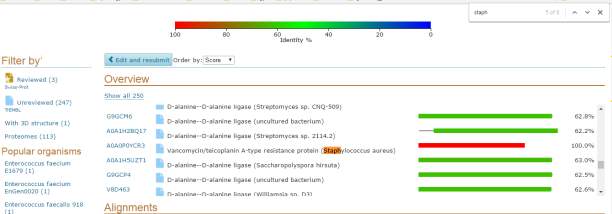

Figure 4 shows that the Enterococcus faecium with protein Vancomycin/Teicoplanin A-type resistance protein VanA when compared to Vancomycin/Teicoplanin A-type resistance protein VanA of Staphylococcus aureus are 100% identical
Figure 5 –illustrates Enterococcus faecium with protein Vancomycin/Teicoplanin A-type resistance protein VanA compared with Staphylococcus aureus illustrating 100% similarity at the 224 residues that they share, the other 119 amino acids are only found in Staphylococcus aureus
When vanA was compared from Staphylococci haemolyticus it was expected to be compared to other coagulase negative staphylococci such as Staphylococcus epidermidis. But as in the previous comparison Staphylococcus epidermidis doesn’t seem to have the vanA gene. So it was compared to other organisms.
It was found in more Staphylococci species than in vanA found in Enterococcus faecium having similarities with other Staphylococci species (see table 8 for more information)
Table 8 illustrates the staphylococci species of which were found after vanA in Staphylococci haemolyticus was compared by doing a BLAST and their similarity rate
| Protein name and type of staphylococci species | Similarity rate |
| Vancomycin/Teicoplanin A-type resistance protein (Staphylococcus aureus) | 100% |
| Vancomycin resistance protein A (Staphylococcus aureus) | 100% |
| VanA (Staphylococcus haemolyticus) | 100% |
| D-alanine: D-lactate ligase (Staphylococcus aureus) | 100% |
| D-alanine–D-alanine ligase (Staphylococcus aureus) | 100% |
| VanA (Staphylococcus sp. 95PS) | 100% |
The vanA of Staphylococci haemolyticus was compared to the vanA protein named D-alanine-D-alanine ligase in Staphylococci aureus appeared of 100% identical.
The bacterial enzyme D-alanine–D-alanine ligase plays an important role in the biosynthesis of cell walls. It helps in the formation of UDP-N-acetylmuramoyl pentapeptide which is a peptidoglycan precursor. These kind of enzyme are proteins that contain up to 360 amino acids which have plenty of conserved regions with the N-terminal Gly-rich region being thought to play a role in the binding of ATP (Henderson et al., 2004). These bacterial enzymes signify the chromosomal versions of not explicitly resilient species to glycopeptide antibiotics such as vancomycin.
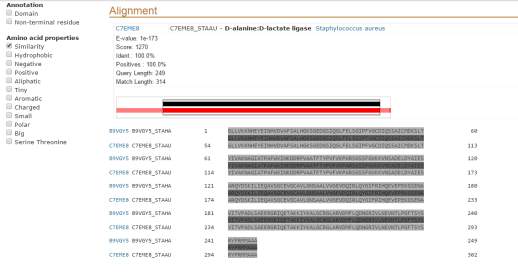
Figure 6- As can be seen the vanA of Staphylococcus haemolyticus was compared to the vanA protein named D-alanine-D-alanine ligase in Staphylococcus aureus appeared of 100% identical in the 249 residues they have in common the other 65 residues of amino acids are once again only found in Staphylococcus aureus
The vanA of Staphylococcus haemolyticus wasthen compared to Vancomycin/Teicoplanin A-type resistance protein VanA found in another enterococcus organism named enterococcus faecium EnGen0180 again due to Teicoplanin.
With a 99.6% of similarity it can be seen that although they are not a 100% identical there are amino acids that although appear different, have the same properties as it can be seen with the small amino acids. (Figure 8)
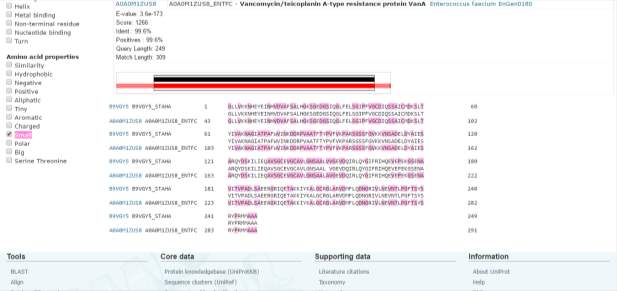
Figure 8 illustrates the blast comparison done between the vanA of Staphylococci haemolyticus to Vancomycin/Teicoplanin A-type resistance protein VanA found in Enterococcus faecium EnGen0180 with their similarity reaching 99.6% on their common residues of 249. The other 60 residues of amino acid is only found in Enterococcus faecium EnGen0180
Vancomycin resistance protein B (vanB)
Two organisms containing vanB were chosen to be compared with. It appeared that no other staphylococci organisms besides S.aureus contain vanB although it was expected for coagulase negative organisms to appear. So S.aureus was chosen as comparison, due to the fact that vancomycin and Teicoplanin are both glycopeptides and work in similar way. See figure 9 for results.
The mixture of altered peptidoglycan precursors terminating in D-lactate, produces VanA- and VanB-type glycopeptide resistance in enterococci is. When comparing VanA-type strains with vanB type, it is clear that vanA has resistant to both vancomycin and Teicoplanin, while VanB-type strains remain Teicoplanin susceptible. (Evers and Courvalin, 1996)
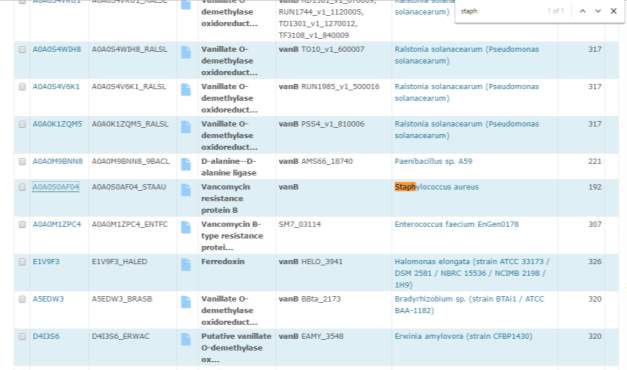
Figure 9 –. It appeared that no other staph organisms besides S.aureus contains vanB
A blast was done on the S.aureus with vanB.
With the blast done vanB of S.aureus appeared to have a 74.4% compatibility to vanA of S.haemolyticus .This indicates the close similarity of vanA and vanB gene which can be due to their similar role 2 genes in resistance towards glycopeptide antibiotics.
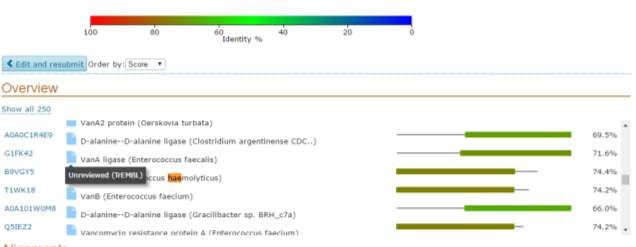
Figure 10– illustrates the vanB of S.aureus appearing to have a 74.4% compatibility to vanA of S.haemolyticus
When amino acids properties where compared though as can be seen in green the positive amino acids although different in some areas represented by different letters have the same properties. The same can be seen in pink with that illustrates the small amino acid properties. This indicates that although it is not 100% identical different amino acids appear to have the same properties. (Figure 13) The question arising is , does these small changes make a difference in the different resistance level towards glycopeptide antibiotics?
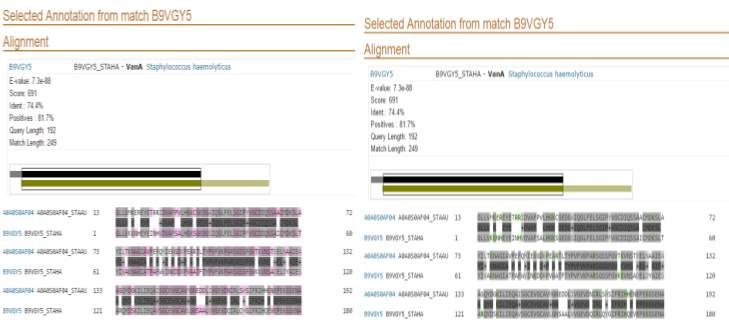
Figure 11 illustrates the vanB of Staphylococcus aureus appearing to have a 74.4% compatibility to vanA of Staphylococcus haemolyticus within regards their shared residues of 192 amino acids with the amino acids properties in green representing the positive amino acids and in pink illustrating the small amino acid properties. The other 57 amino acids are only found in Staphylococcus haemolyticus
Secondly vanB found in Enterococci faecalis with protein name vanB was chosen and underwent another blast. (Figure 12) At about 90% of clinical isolates, Enterococcus faecalis and Enterococcus faecium are the utmost flourishing species cultivated from humans, instigating a diversity of nosocomial toxicities, with urinary tract infections the most common. As many drugs are resistant to E. faecalis, the typical infections can be remarkably challenging to treat due to the drug resistance of many E. faecalis isolates. (Akhter, Ahmed and Anwar, 2017)
Figure 12 – illustrates vanB found in enterococci feacalis

Figure 13 shows the sequence of Enterococci feacalis with protein name Vancomycin resistance protein B used for BLAST
Once it was compared it was found with 75.7% similarities to S.aureus.

Figure 14- illustrates results found with 75.7% similarities to S.aureus.
Once again areas with different amino acids appear to have the same properties. As can be seen below( figure 15) in green are the areas of amino acids with positive properties and in pink polar properties again of different amino acids but contribute in some cases for the same property.
 Figure 15 – enterococci feacalis with protein name vanB when compares to amino acids with positive properties with green areas of amino acids representing areas with positive properties and in pink polar properties .some amino acids appear to be different but contribute in some cases for the same property.
Figure 15 – enterococci feacalis with protein name vanB when compares to amino acids with positive properties with green areas of amino acids representing areas with positive properties and in pink polar properties .some amino acids appear to be different but contribute in some cases for the same property.
Conclusion for both vanA and vanB genes
Although these genes were not found in the species of coagulase negative staphylococcus except for the case found in staph haemolyticus, further studies need to determine if Staphylococcus epidermidis has a gene that have similar properties as vanA and vanB that contribute to resistant .a closer look to the genes or proteins involved in the biofilm formation of Staphylococcus epidermidis such as the protein MSCRAMMs (Microbial surface components recognizing adhesive matrix molecules) which play an important role in the fast protection abiotic surfaces of indwelling medical devices.
As seen in the BLAST, a lot of sequences that weren’t 100% identical had the same amino acid properties so it could be of interest to find the genome of vanB and vanA might be similar to the one that epidermidis has. Another question raised is, do these small changes make a difference in the different resistance level towards glycopeptides antibiotics? with further research these questions may lead to answers that can be useful for understanding the mechanics of resistance and how to reverse this effect.
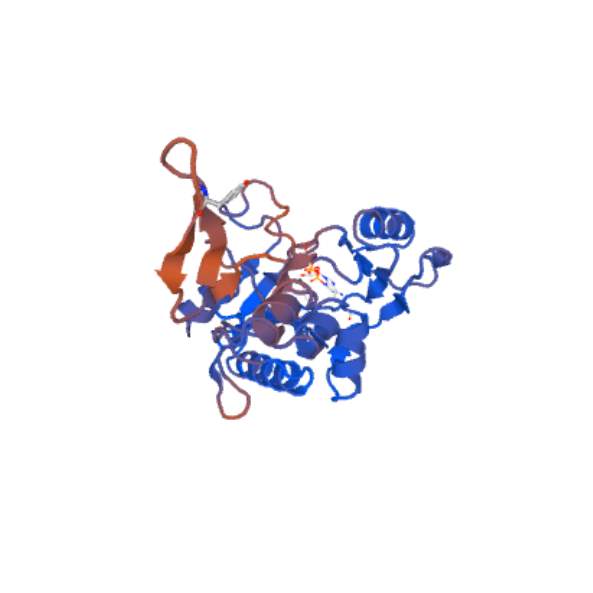 Figure 16– illustrates the Swiss model of vancomycin/Teicoplanin a-type resistance found in Enterococcus faecium vanA model.
Figure 16– illustrates the Swiss model of vancomycin/Teicoplanin a-type resistance found in Enterococcus faecium vanA model.
Appendix B
Targocid
Package leaflet: Information regarding this antibiotic drug Targocid which is the trade name of Teicoplanin.




Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allRelated Content
All TagsContent relating to: "Cancer"
Cancer is a disease in which cells grow or reproduce abnormally or uncontrollably. Cancerous cells have the potential to spread to other areas of the body in a process called metastasis.
Related Articles
DMCA / Removal Request
If you are the original writer of this dissertation and no longer wish to have your work published on the UKDiss.com website then please:




