Incorporating Uniform Drug Dispersions into Orally Dissolving Films (ODFs)
Info: 8974 words (36 pages) Example Dissertation Proposal
Published: 31st Jan 2022
Tagged: Medical
Proposal Summary
Despite the oral route of drug delivery being the most preferred amongst patients, and thereby having better adherence compared to other delivery routes, there remain certain administration issues, particularly in patients that may have difficulty swallowing (e.g. paediatric or geriatric patients). Orally Dissolving Films (ODFs) have great potential to ease these problems, whilst also improving acceptability and portability of many therapeutic agents.
One issue, however, is ensuring that the drug is evenly distributed throughout the film, to provide a uniform dose. This issue is more prevalent for poorly soluble drugs. Drug uniformity is of high importance because if the drug is not uniformly dispersed this can lead to underdosing or overdosing; resulting in no clinical benefit or serious side effects, and possibly death. This project will therefore explore a variety of stabilising excipients, mainly surfactants (i.e. Spans and Tweens, from standard quality to ultra-refined high purity grades), for their ability to produce uniform drug dispersions prior to casting in the films. It is intended that the solvent casting method will be used to manufacture the ODFs, with formulations subsequently analysed for their mechanical properties (e.g. thickness, elongation, etc.), drug content, uniformity, and stability.
Background
Orally dissolving films (ODFs), also known as oral strips, are thin films that are prepared using hydrophilic polymers that rapidly dissolve on the tongue or buccal cavity (Dixit and Puthli, 2009). They can be used for patient groups who find it difficult, or cannot physically swallow tablets (dysphagia), such as paediatrics, geriatrics, bedridden patients and those suffering from Parkinson’s disease, mucositis, or vomiting (Bala et al., 2013).
ODFs are administered via the oral cavity, which consists of the oral cavity mucosa, and the buccal membrane (Rathbone et al., 1994) and can be divided into two regions: the oral vestibule, and the oral cavity itself (Squier et al., 1976a). Although the surface area for absorption is much lower than the skin, it has a rich blood supply and the mucosa of the oral cavity has better permeability, allowing good absorption of the drugs given via this route (Rathbone et al., 1994; Galey et al., 1976; Lesch et al., 1989). Generally, the oral cavity is a rather complex structure and the thickness of certain areas varies vastly from 100-800 μm, so the ODFs must be optimised for the desired area, which would be the tongue (Ponchel, 1994).
Mucus and saliva are key for drug delivery in the oral cavity; the physiological turnover of saliva is 1-2 mL/min, which would need to be taken into consideration when formulating ODFs as saliva aids the disintegration of the ODFs in the oral cavity (Mattes, 1997). Mucus is made up of proteins and carbohydrates and is secreted by the major and minor salivary glands, therefore composes part of the saliva (Peppas and Buri, 1985). The key role of the mucus present in the saliva is to aid the bioadhesion of mucoadhesive drug systems in the oral cavity, keeping the oral cavity hydrated and providing adequate lubrication (Rathbone et al., 1994; Tabak et al., 1982).
Drugs can be absorbed in the oral cavity either via the transcellular, intracellular or paracellular pathway (Harris and Robinson, 1992). A limitation with the route of absorption is that salivary secretion within the oral cavity is a constant process, making it difficult for dosage forms to be retained for prolonged periods of time, which can cause the accidental swallowing of dosage forms resulting in gastric absorption (Dixit and Puthli, 2009).
ODFs have many advantages and disadvantages compared to other oral/non-oral dosage forms. The advantages can be categorised into patient focused and therapeutic focused advantages. The disadvantages are focused on the manufacturing process and the therapeutic outcomes.
Advantages
The therapeutic focused advantages are that they are small, equating to a large surface area; increasing disintegration and dissolution (Dixit and Puthli, 2009). The first-pass metabolism mechanism can be avoided as the oral cavity is highly vascularised (Dixit and Puthli, 2009). This route is ideal for drugs that are sensitive to acidic hydrolysis, or extensively metabolised/degraded; allowing a reduction in the dose and possible side effects, yet still achieving the desired therapeutic outcome (Rathbone et al., 1994). ODFs have the ability to provide accurate and precise doses dependent on the production method, which is ideal for Narrow Therapeutic Index drugs (Borges et al., 2015).
The patient focused advantages are that they have a rapid onset of action making them ideal for patients suffering from allergic reactions, and motion sickness (Choudhary et al., 2012). The size of the ODFs also avoids the prevalence of choking and suffocation and they are hardly spat out, so they can be given to bedridden and non-cooperative patients (Karki et al., 2016). A key advantage is that water is not needed for administration, increasing convenience for patients such as those suffering from dysphagia, improving medication adherence, and making them easily portable (Bala et al., 2013; Borges et al., 2015).
Disadvantages
The main therapeutic disadvantages are that high doses of drug cannot be incorporated into ODFs and they are dependent on the flow of saliva (Bala et al., 2013; Rathbone et al., 1994). The manufacturing disadvantages are that the packaging process of ODFs requires special and expensive equipment (Bala et al., 2013). Also, unstable drugs at buccal pH cannot be formulated this way and ODFs can be poorly stable in environments of high humidity and high temperature (Borges et al., 2015).
Many drugs have been formulated as ODFs, some of which are given in Table 1 (see Appendix 1).
ODFs are principally made up of the Active Pharmaceutical Ingredient (API), hydrophilic polymers, plasticisers and surfactants (which will be the focus of this project), with additional agents often added to improve the aesthetics of the dosage form, such as flavour, sweetening agents, saliva stimulating agents and colouring agents.
Hydrophilic polymers are the major component of ODFs (ranging from 45-65% of the total film) and are important for the rapid dissolution of ODFs upon contact with saliva, as well as the mechanical strength of the ODFs (Irfan et al., 2016). Hydrophilic polymers should exhibit sufficient tensile strength, be non-toxic, and possess good spread ability (Irfan et al., 2016). They can be either natural (e.g. starch, pullulan, and gelatine) or synthetic (e.g. HPMC, and polyvinyl alcohol) (Chauhan et al., 2012). Plasticisers, which account for 0-20% of the total film, improve the mechanical properties of ODFs, such as tensile strength and percentage elongation (Dixit and Puthli, 2009; Irfan et al., 2016). Examples of plasticisers are PEG, glycerol, diethyl phthalate, triethyl citrate, and tributyl citrate (Bala et al., 2013). Surfactants act as agents that wet and solubilise ODFs, equating to rapid disintegration and drug release, particularly important when working with poorly soluble drugs (Dixit and Puthli, 2009). Examples of common surfactants used are tweens, sodium lauryl sulfate, and poloxamer 407 (Irfan et al., 2016). A study has shown how surfactants can help poorly soluble drugs dissolute more readily (Vuddanda et al., 2017). In this study the disintegration and dissolution time was improved for poorly soluble drugs in formulations containing surfactants (Vuddanda et al., 2017).
The products on the market (Table 1) use the excipients mentioned above. The current marketed ODFs contain an API that either fits into class 1 or class 2 of the Biopharmaceutical Classification System (BCS) (U.S. Food and Drug Administration, 2016). Class 1 drugs have a high permeability and a high solubility, whereas class 2 drugs have a high permeability but a low solubility; a class 2 drug will be used in this project (U.S. Food and Drug Administration, 2016). Poor solubility can prove to be a challenge and can lead to poor drug uniformity within an ODF, as solvents are used in the production process. Currently excipients such as chitosan are used to overcome this; however, this project will focus on the use of surfactants (i.e Span and Tweens) to overcome this challenge (Vuddanda et al., 2017).
The optimisation of the drug load and dose uniformity in an ODF by the addition of different surfactant strengths, quantities, and grades (namely Spans and Tweens) will be explored. Surfactants are surface active agents and are often added into formulations to overcome the fluctuations between surface tension and surface energy. They also play a key role in controlling the balance of hydrophilic and hydrophobic interactions within a molecule (Pouton, 2006). The addition of surfactants in polymer-based formulations can prevent precipitation/crystallisation of molecules, therefore, avoiding non-uniform drug dispersion and aiding drug loading (Pouton, 2006).
The benefits of surfactants are that they improve the dissolution profile of poorly soluble drugs, they reduce surface tension, and they can also be used to stabilise drug suspensions (Pouton, 2006). Surfactants improve the solubility of poorly soluble drugs by forming micelles (Savjani, et al., 2012). These are formed by exceeding the surfactant concentration above the critical micelle concentration (CMC), which ranges form 0.05-0.10% for most surfactants (Savjani, et al., 2012). Micelles can entrap drugs within their structure hence increasing the solubility of the drug and drug loading (Savjani, et al., 2012). Surfactants also improve the wetting of solids and they can be used to stabilise microemulsions and suspensions in which drugs are suspended (Savjani, et al., 2012).
There are four main classes of surfactant that are explained in Table 2. The non-ionic surfactant group will be used in this project, as their primary function is to act as wetting/emulsifying agents.
| Surfactant type | Uses | Examples |
| Anionic- hydrophilic group is negatively charged | Laundry detergents, dishwashing liquids, and shampoos
Bacteriostatic activity Can be used in medicated shampoo (Lichtarowicz, 2013) |
alkylbenzene sulfonates, alkyl sulfates, alkyl ether sulfates, and soaps (Lichtarowicz, 2013) |
| Cationic- hydrophilic group is positively charged | Fabric softeners
Bactericidal activity pharmaceutically (Lichtarowicz, 2013) |
quaternary ammonium, and pyridinium cationic surfactants
(Attwood and Florence, 2012) |
| Non-ionic- No electrical charge | Laundry detergents (both powders and liquids), hard surface cleaners, polymerisation of emulsions,
wetting agents, and agrochemical formulations (Lichtarowicz, 2013) |
Sorbitan esters (Spans)- have a low HLB value, Polysorbates (Tweens) high HLB value,
Poloxamers (Pluronic), HLB dependent on chain length. (Attwood and Florence, 2012) |
| Amphoteric (Zwitterionic) – head group carries both a positive and negative charge. | Shampoo and cosmetics.
(Lichtarowicz, 2013) |
Betaines (Lichtarowicz, 2013) |
Table 2: Classes of surfactants and examples
Aims & Objectives
Aims:
- To optimise the loading and uniformity of a poorly soluble drug in an ODF, using different surfactant types, strengths, and grades.
Objectives:
- Formulate the ODF with and without the API using the solvent casting method, and optimise the film with the addition of different surfactant strength, quantities, and grades.
- Compare the properties of the formulated films, by analysing the drug load and uniformity with different surfactant strength and grades present, and by assessing the physical properties (i.e. thickness and appearance) and mechanical properties (i.e. tear resistance, percent elongation, and tensile strength).
Methods
In this project different surfactant types and grades will be used (namely Span and Tween, from standard quality to ultra-refined high purity grades), to see how they affect drug content and uniformity of the ODFs. The surfactants used will be acting as wetting agents, e.g. Span (Hirsch, 2015). A poorly water-soluble drug will be used, such as Loratadine, which has a water solubility of 0.000011 mg/ml (PubChem; Loratadine, 2017) and is a class 2 drug according to the BCS.
There are two main production methods:
- Mixing both the API and excipients together, before forming a film (i.e. Hot Metal Extrusion (HME), and Solvent Casting (SC)) (Buanz et al., 2015).
- Producing a film using the excipients (known as a ‘free film’), and then depositing the API into this (i.e. Flexography and Thermal-Ink Jet Printing (TIJP)), as shown in Figure 1 (Buanz et al., 2015).
HME and SC are two very similar processes, they mix the API, and excipients together to form a film. HME does this using heat and SC achieves this using solvents (Figure 2) (Buanz et al., 2015).
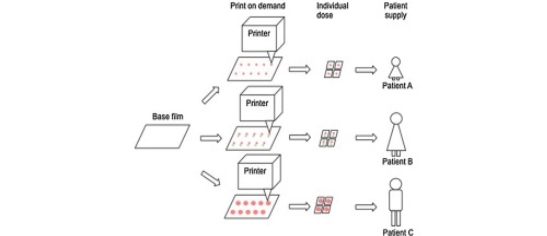
Figure 1: Schematic diagram of the printing methods (Flexography and Thermal-Ink Jet Printing) (Preis et al., 2015)
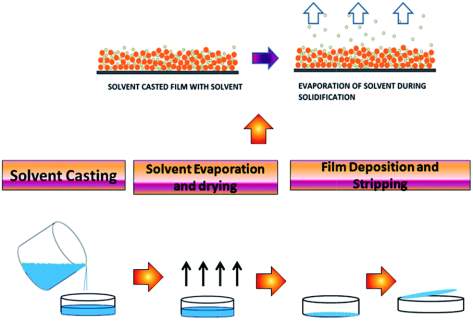
Figure 2: Schematic diagram of the Solvent Casting method (Katiyar and Balasubramanian, 2015)
The solvent used depends on the API within the formulation, hence the API will need to be screened for compatibility with the solvents, to ensure the best one is used (Dixit and Puthli, 2009). Commonly used solvents are class 3 solvents (methyl acetate) as these solvents are regarded as less toxic and of lower risk to human health ICH Topic Q3C (R4) Impurities: Guideline for Residual Solvents, 2009). Heat can be used if required, to ensure complete dissolution once the solvent has been added (Dixit and Puthli, 2009). Air pockets in the ODFs during formulation can lead to reduced drug uniformity within the ODF but this can be avoided by the type of stirring method used (Dixit and Puthli, 2009). After the removal of air pockets, the solution is put into a mould and inert substrates such as glass and a roller can be used at this stage (Morales and McConville, 2011). Upon drying the films are cut into the desired shape and size, and packaged (Dixit and Puthli).
Flexography is uncommon. The process uses printing and rotating rollers to deposit the drug on the film (Buanz et al., 2015). TIJP works similarly, but it can deposit a minute volume of the drug onto the film, which allows it to be used for Narrow Therapeutic Index (NTI) drugs; having high precision and accuracy (Buanz et al., 2015).
Limitations of the methods:
- HME cannot be used on heat liable APIs (Buanz et al., 2015).
- SC increases the possibility of crystallisation of the API out the film; resulting in a brittle film with altered taste (Buanz et al., 2015).
- Flexography poses a risk of contamination, low resolution and is only used on a medium-scale (Buanz et al., 2015).
Benefits of the methods:
- Flexography can be used for NTI drugs, due to the accuracy (Buanz et al., 2015).
- HME produces a drug in the form of solid dispersion or solution, which is ideal to use for poorly soluble drugs as it can improve their solubility (Repka et al., 2005).
- SC method has quite a few benefits, such as better physical properties of the ODF formulated, ease of preparation, cost-effectiveness, and excellent uniformity of thickness (Karki et al., 2016).
Analysis of the drug load and uniformity of the films with different surfactant strength and grades present is key in this project. This will be achieved by assessing the physical properties (i.e. thickness and appearance) and mechanical properties (i.e. tear resistance, percent elongation, and tensile strength). The drug content/uniformity test will be carried out as directed by the British Pharmacopeia. Tensile strength, percent elongation, tear resistance, and young modulus are interlinked; they all should be considered when evaluating the mechanical properties of a film. These tests are represented in Table 3. The equipment used are represented in Figure 3.
| Characterisation | Measurements/Purpose | How the test will be carried out/Equipment required. |
| Thickness | To ensure uniformity of thickness, which in turn ensures dose uniformity. | We can test for thickness using a micrometre screw gauge at a different location, and we can see if the formulation is at its desired thickness by observations when assembling. |
| Organoleptic evaluation/Appearance | Taste and appearance should be accepted among most of the population. | Tasting tests will not be carried out, but by observing the formulation we can ensure that the film has a smooth surface and that they are all the same size and colour. |
| Percent Elongation |  Increase in length of strip x 100 Increase in length of strip x 100
Initial length of strip A measure of strain/plasticity. |
This test will be carried out using a texture analyser. The texture analyser stretches the sample increasing its length, allowing us to measure the stress which the oral film is able to withstand without breaking. This should be carried out on 3 or more doasge forms. |
| Young’s Modulus |  Slope x 100 Slope x 100
Strip thickness x Cross-head speed A measure of stiffness. Indicates a formulations resistance to deformation. |
This test will be carried out using a texture analyser. The measurement of this is dependent on the speed of the cross-head which would be changed. |
| Tear Resistance | Find the force at which the film will tear. Measurement is taken in Newtons (N).
A measure of strength |
The test will be using the texture analyser. The equipment will stretch the film in a comparable way to the percent elongation test and the force at which the film tears will be recorded. This test will be carried out on 3 or more films. |
| Tensile strength |  Load at failure x 100 Load at failure x 100
Strip thickness x Strip width A measure of strength. |
The test will be using the texture analyser. The equipment will stretch the film in a comparable way to the percent elongation test and the force at which the film breaks will be recorded. This test will be carried out on 3 or more films. |
| Folding Endurance | Fold the strip in the same place several times till it breaks.
A measure of endurance. |
In this test the oral film will be folded numerous times at the same place at a 180° angle until it breaks. The number of folds will be recorded in a table and the test will be carried out for at least 10 dosage units. A folding endurance of 300 or more would classify a film as having excellent flexibility. |
| Disintegration time | Should disintegrate within 5-30 seconds | Will be carried out using the basket-rack assembly technique. One dosage unit will be placed into each of the 6 tubes of the basket. The tubes will have a specified medium at 37 ± 2 °C. At the end of 30 seconds observations will be carried out to see what has happened to the dosage unit. All the dosage units should have disintegrated. If 1 or 2 fail, then the test needs to be repeated with an additional 12 dosage units. The test is passed if 16 of the 18 dosage units successfully disintegrated within the specified time. |
| Dissolution time | Using the paddle or basket apparatus as defined by the British Pharmacopeia. | This test will be carried out using the basket apparatus.
You would place the stated volume of the dissolution medium in the apparatus, after which you would equilibrate the temperature to 37 ± 0.5 °C. One dosage unit will be placed into this apparatus and you would take measurements at specific time intervals to establish the dissolution time. When the measurements are taken the apparatus should be topped up with the dissolution medium and the temperature should be maintained throughout the process. You can repeat this test with other dosage units. It would be best to use 6 or more dosage units to carry out this test. |
| Drug Content/Uniformity | Using the content uniformity test as defined by the British Pharmacopeia. Limit is between 85%- 115% | The content of the API will be taken from 10 dosage units at random using a suitable analytical technique. To pass the test each dosage unit should have a content between 85-115% of the average content of the total dosage units. The uniformity of content test will fail if one dosage is out the 75-125% limit. If a dosage unit is outside the 85-115% limit but within the 75-125% limit, then the content of another 20 dosage units should be taken. No more than one dosage unit of the 30 taken can be outside of the 85-115% limit for this test to be passed and not a single dosage unit can be out of the 75-125% limit of the average content. |
Table 3: Characterisation for ODFs (Dixit and Puthli, 2009, British Pharmacopoeia, 2017, and Karki et al., 2016).
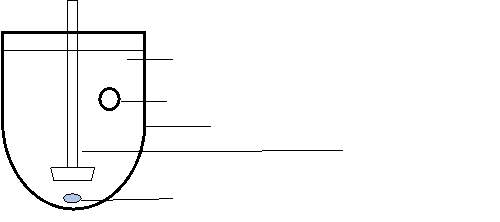
(a) Basket apparatus: Dissolution test
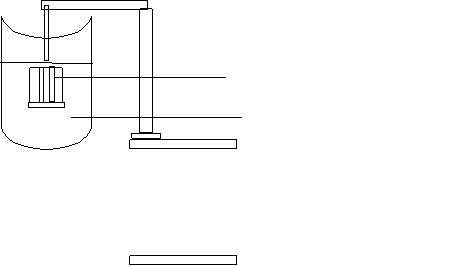
(b) Basket-rack apparatus: Disintegration test
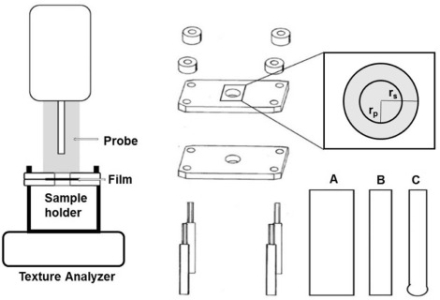
(c) Texture analyser used for testing mechanical properties.
A, B and C represent the different size probes that can fit into the analyser Karki et al., 2016).
Figure 3: Schematic diagrams of the apparatus’ used for the tests: (a) Basket apparatus, (b) Basket-rack apparatus, (c) Texture analyser.
Appendix 1
| Active Pharmaceutical Ingredient (API) |
Indication |
Dose |
Water solubility |
Main Excipients used |
| Ondansetron
(Zuplenz/ Setofilm) (Monosolrx, 2010, and Tesa Labtec, 2011) |
Anti-emetic- used for nausea and vomiting induced by chemotherapy, radiotherapy, and for post-operative nausea and vomiting (PONV) | 4-8mg | Very slightly soluble
0.3551 mg/mL (PubChem; Ondansetron, n.d.) Class 1/2 (Koland et al., 2011) |
Rice Starch
Poly (vinyl alcohol) Macrogol 1000 Acesulfame potassium E950 Glycerol E422 Titanium dioxide E171 Levomenthol Polysorbate 80 E433 (electronic Medicines Compendium (eMC), 2017) |
| Nicotine
(NiQuitin) (electronic Medicines Compendium (eMC); NiQuitin, 2015) |
Smoking cessation | 2.5mg | Freely soluble
1×106 mg/L (PubChem; Nicotine, n.d.) Class 1 |
Methacrylic Acid-Ethyl Acrylate
Triethyl Citrate (E1505) Peppermint Flavour Sucralose (E55) Sodium Hydrogen Carbonate (E500 ii) Ethanol (electronic Medicines Compendium (eMC); NiQuitin, 2015) |
| Diphenhydramine HCl/ Phenylephrine HCl
(Night time Triaminic Thin Strips) (Dixit and Puthli, 2009) |
Anti-histamine/cough suppressant
For cough and cold relief |
12.5mg/
5mg |
Slightly soluble
3060 mg/L (at 37°C) (PubChem; Diphenhydramine, n.d.) Class 1 Freely soluble 1×106 mg/L (at 25°C) (PubChem; Phenylephrine Hydrochloride, n.d.) |
Acetone, Flavours, hypromellose, Maltodextrin, Mannitol, Polyethylene glycol, Polypropylene glycol, Purified water, Sodium polystyrene sulfonate, Sucralose, Titanium dioxide, Colour. (Dixit and Puthli, 2009) |
| Fentanyl
(Breakyl) (electronic Medicines Compendium (eMC); Breakyl, 2015) |
Treatment of breakthrough pain in cancer patients already receiving maintenance opioid treatment | 200 micrograms- 800 micrograms | Very slightly soluble
200 mg/L (at 25 °C) (PubChem; Fentanyl, n.d.) Class 2 (Karki et al., 2016). |
Propylene glycol (E1520), Sodium benzoate (E211), Ferric oxide (red) (E 172), Anhydrous citric acid,
All-rac-alpha-tocopheryl acetate, Monobasic sodium phosphate (anhydrous), Sodium hydroxide, tribasic sodium phosphate (anhydrous), Polycarbophil, Hydroxypropylcellulose, Hydroxyethylcellulose, Carmellose sodium, Titanium dioxide (E 171), Saccharin sodium, Peppermint oil, Colour (electronic Medicines Compendium (eMC); Breakyl, 2015) |
| Benzocaine (Chloraseptic)
(Dixit and Puthli, 2009) |
Sore throat/mouth irritation | 3mg | Slightly soluble
1310 mg/L (at 30 °C) (PubChem; Benzocaine, n.d.) Class 2 (Van Eerdenbrugh et al., 2010) |
Benzocaine 3 mg, BHT, cornstarch, erythritol, FD&C Red 40, hydroxypropyl methylcellulose, malic acid, menthol, monoammonium glycyrrhizinate, cherry flavors, polyethylene oxide, sucralose. (Dixit and Puthli, 2009) |
| Listerine pocket packs (Dixit and Puthli, 2009) | Fresh breath | Varies | n/a | Pullulan
Cool mint, fresh citrus flavours. (Dixit and Puthli, 2009) |
Table 1: Drugs available as orally dissolving films
Annotated Bibliography
Attwood, D. and Florence, A. (2012). Physical Pharmacy. 2nd ed. [ebook] London: Pharmaceutical Press, pp.43-45. Available at: https://www.pharmpress.com/files/docs/FTphyspharm_sample.pdf [Accessed 6 Nov. 2017]
This textbook focuses on the physicochemical properties of pharmaceutics. It has a range of chapters from nanotechnology to drug stability. I looked at chapter 4 of this textbook that focused on the different types of surfactants and their properties; I have mentioned these in Table 3 of my project.
Bala, R., Khanna, S., Pawar, P. and Arora, S. (2013). Orally dissolving strips: A new approach to oral drug delivery system. International Journal of Pharmaceutical Investigation, [online] 3(2), p.67. Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3757902/ [Accessed 1 Nov. 2017].
Fast dissolving films are becoming more popular. This article highlights the advantages/disadvantages of this new drug delivery system as well as the methods of manufacture, excipients used, and the current manufacturing systems that are available for this type of dosage form. The information I used from this article were the advantages/disadvantages, the excipients used for dosage forms, and the target patient groups for these films.
Borges, A., Silva, C., Coelho, J. and Simões, S. (2015). Oral films: Current status and future perspectives. Journal of Controlled Release, [online] 206, pp.1-19. Available at: https://doi.org/10.1016/j.jconrel.2015.03.006 [Accessed 1 Nov. 2017].
This review explores the oral film manufacture and the technology involved. Areas explored vary from the polymeric matrices, advantages/disadvantages and the new possible market applications. From this article, I took some advantages/disadvantages mainly concerning the therapeutic and manufacturing areas.
British Pharmacopoeia. (2017). Appendix XII C. Consistency of Formulated Preparations. [online] Available at: https://www.pharmacopoeia.com/bp-2018/appendices/appendix-12/appendix-xii-c–consistency-of-formulated-preparations.html?published-date=2017-08-01&text=uniformity [Accessed 31 Oct. 2017].
The British Pharmacopoeia (BP) set out the quality standards for UK medicinal products. Both individuals and organisations who are involved in pharmaceutical research, development, manufacturing, and testing can use it. The standards are publicly available, and together with the British National Formulary (BNF), the British Pharmacopeia defines the UK’s pharmaceutical standards. From this source, I obtained information on how to carry out the uniformity of content test for the films that I will be producing.
Buanz, A., Belaunde, C., Soutari, N., Tuleu, C., Gul, M. and Gaisford, S. (2015). Ink-jet printing versus solvent casting to prepare oral films: Effect on mechanical properties and physical stability. International Journal of Pharmaceutics, [online] 494(2), pp.611-618. Available at: https://doi.org/10.1016/j.ijpharm.2014.12.032 [Accessed 31 Oct. 2017].
The purpose of this article was to compare and contrast the mechanical properties and physical stabilities of oral films after formulation by either thermal-ink jet printing (TJP) or solvent casting. The drug used was clonidine as it has a low therapeutic dose. The information I obtained from this article was the advantages/disadvantages of both methods and how to carry out each method.
Chauhan, I., Yasir, M., Nagar, P. (2012). Insights into polymers: film formers in mouth dissolving films. Drug Invent. pp. 56-73
The article highlights various natural and synthetic polymers, their properties and applications in oral film delivery systems. From this article, I obtained information regarding the types of polymers used in oral films.
Choudhary, D., Patel, V., Chhalotiya, U., Patel, H. and Kundawala, A. (2012). Development and Characterization of Pharmacokinetic Parameters of Fast Dissolving Films Containing Levocetirizine. Scientia Pharmaceutica, 80(3), pp.779-787.
This journal article looked at the physical stability and mechanical properties of the oral films produced at the start of the project. It has also looked at the pharmacokinetic properties of the films as well as the advantages/disadvantages and the purpose of the films. I used a few advantages/disadvantages for my research proposal from this article.
Dixit, R. and Puthli, S. (2009). Oral strip technology: Overview and future potential. Journal of Controlled Release, [online] 139(2), pp.94-107. Available at: https://www.sciencedirect.com/science/article/pii/S016836590900426X#bib102 [Accessed 31 Oct. 2017].
Many research groups have been focusing their research on oral films, as it is an area, which is currently expanding in the pharmaceutical world. This review article focuses on the materials used in oral films, and the purpose of oral films. Other aspects looked at are the manufacturing process, commercial technologies, and future business prospects of this technology. The information I used from this article were the advantages/disadvantages, the methods of formulation, marketed oral films, and the process of characterising the oral films.
electronic Medicines Compendium (eMC). (2015). Breakyl 200mcg buccal film. [online] Available at: https://www.medicines.org.uk/emc/medicine/28361 [Accessed 1 Nov. 2017].
The electronic Medicines Compendium (eMC) contains up to date, easily accessible information about medicines licensed for use in the UK. The eMC contains two types of documents: summaries of product characteristics (SPCs) and patient information leaflets. The SPCs contain the pharmacokinetic and pharmacodynamics properties of the drug in question. I used the SPC on this website for the Breakyl buccal film to find out the excipients present in the formulation, which I used in Table 1 of this document.
electronic Medicines Compendium (eMC). (2015). NiQuitin Strips 2.5mg Oral Film. [online] Available at: https://www.medicines.org.uk/emc/medicine/27614 [Accessed 1 Nov. 2017].
The electronic Medicines Compendium (eMC) contains up to date, easily accessible information about medicines licensed for use in the UK. The eMC contains two types of documents: summaries of product characteristics (SPCs) and patient information leaflets. The SPCs contain the pharmacokinetic and pharmacodynamics properties of the drug in question. I used the SPC on this website for the NiQuitin oral film to find out the excipients present in the formulation, which I used in Table 1 of this document.
electronic Medicines Compendium (eMC). (2017). Setofilm 4 mg Orodispersible Films. [online] Available at: https://www.medicines.org.uk/emc/medicine/28020 [Accessed 1 Nov. 2017].
The electronic Medicines Compendium (eMC) contains up to date, easily accessible information about medicines licensed for use in the UK. The eMC contains two types of documents: summaries of product characteristics (SPCs) and patient information leaflets. The SPCs contain the pharmacokinetic and pharmacodynamics properties of the drug in question. I used the SPC on this website for the Setofilm oral film to find out the excipients present in the formulation, which I used in table 1 of this document.
Galey, W., Lonsdale, H. and Nacht, S. (1976). The In Vitro Permeability Of Skin And Buccal Mucosa To Selected Drugs And Tritiated Water. Journal of Investigative Dermatology, [online] 67(6), pp.713-717. Available at: https://doi.org/10.1111/1523-1747.ep12598596 [Accessed 1 Nov. 2017].
This article looks at the permeability of the skin and buccal mucosa for several drugs in different environments. I used this article to explain the composition of the buccal mucosa, which allowed me to describe the process of the absorption of the oral films in the oral cavity.
Harris, D. and Robinson, J. (1992). Drug Delivery via the Mucous Membranes of the Oral Cavity. Journal of Pharmaceutical Sciences, 81(1), pp.1-10.
This article focuses on the routes of drug delivery in the oral mucosa. It highlights what is required for the absorption of the drugs in the oral mucosa and the pathways by which the drug from an oral film is delivered into systemic circulation. From this article, I was able to obtain information on ways that a drug can be absorbed/delivered in the oral cavity.
Hirsch, M. (2015). Surface Active Agents (Surfactants): Types and Applications. [online] Prospector. Available at: https://knowledge.ulprospector.com/3106/pc-surface-active-agents-surfactants/ [Accessed 31 Oct. 2017].
The Prospector is a search engine that offers information for many products, ranging from household/industrial cleaners to pharmaceuticals. This website offers accurate and reliable information in these areas. I used this website to obtain information on the different types of surfactants used in pharmaceuticals and their roles.
ICH Topic Q3C (R4) Impurities: Guideline for Residual Solvents. (2009). [ebook] London: European Medicines Agency, pp.8-9. Available at: https://www.tga.gov.au/sites/default/files/ich28395enrev4.pdf [Accessed 31 Oct. 2017].
The purpose of this guideline is to recommend acceptable volume of residual solvents that can be used in pharmaceutical products without endangering the patient. The guideline describes the different classes of solvents and provides evidence based toxicology reports for these classes. It also gives information on how you can appropriately select a solvent for the drug that you will be formulating. From this article I looked at the class 3 solvent section as these were the solvents that were known to be the least toxic therefore, were recommended for use in pharmaceutical products.
Irfan, M., Rabel, S., Bukhtar, Q., Qadir, M., Jabeen, F. and Khan, A. (2016). Orally disintegrating films: A modern expansion in drug delivery system. Saudi Pharmaceutical Journal, [online] 24(5), pp.537-546. Available at: https://doi.org/10.1016/j.jsps.2015.02.024 [Accessed 1 Nov. 2017].
This review focuses on the advantages/disadvantages of oral films and it also touches upon the manufacturing process of oral films highlighting the excipients used and their role as well as giving examples of commonly used excipients. From this article I obtained information on the function of each excipient in an oral film and some examples within these excipient groups.
Karki, S., Kim, H., Na, S., Shin, D., Jo, K. and Lee, J. (2016). Thin films as an emerging platform for drug delivery. Asian Journal of Pharmaceutical Sciences, [online] 11(5), pp.559-574. Available at: https://doi.org/10.1016/j.ajps.2016.05.004 [Accessed 1 Nov. 2017].
The purpose of this review is to discuss the key factors regarding the formulation of oral films, which includes the physico-chemical properties, advantages/disadvantages of oral films, methods of characterisation, and current trends/future perspectives of these oral films. From this article I used information regarding the purpose of oral films, their advantages/disadvantages, and tests that need to be carried out on the oral films (Table 4).
Katiyar, N. and Balasubramanian, K. (2015). Nano-heat-sink thin film composite of PC/three-dimensional networked nano-fumed silica with exquisite hydrophobicity. RSC Advances, 5(6), pp.4376-4384.
From this article I used a schematic diagram of the solvent casting method which is presented in figure 1.
Koland, M., Vijayanarayana, K., Charyulu, R. and Prabhu, P. (2011). In vitro and in vivo evaluation of chitosan buccal films of ondansetron hydrochloride. International Journal of Pharmaceutical Investigation, 1(3), p.164.
This article focuses on the evaluation of developed oral films containing ondansetron and it gives the properties of the drug used and from this I was able to find the BCS class of ondansetron.
Lesch, C., Squier, C., Cruchley, A., Williams, D. and Speight, P. (1989). The Permeability of Human Oral Mucosa and Skin to Water. Journal of Dental Research, 68(9), pp.1345-1349.
This article was based on a study carried out on specimens of the oral mucosa taken form 58 individuals. The permeability of the specimens was studied. The information that I was able to take from this article was how permeable the oral mucosa was and how this would affect the absorption of drug in the oral mucosa.
Lichtarowicz, M. (2013). Surfactants. [online] The Essential Chemical Industry. Available at: http://www.essentialchemicalindustry.org/materials-and-applications/surfactants.html [Accessed 6 Nov. 2017].
This website highlights the key topics regarding industry chemistry ranging from industrial processes to materials and application. I focused on the surfactant area of this website and it is where I found most the information on the different surfactant groups with examples of their uses.
Mattes, R. (1997). Physiologic Responses to Sensory Stimulation by Food. Journal of the American Dietetic Association, 97(4), pp.406-413.
This article highlights how we physiologically respond to food and it focuses on how saliva plays a role in this sensory stimulation. From this article I obtained important information on the flow rate of saliva and how it can have both advantages and disadvantages in the delivery of oral films.
Monosolrx. (2010). MonoSol Rx Announces FDA Approval of Zuplenz® Oral Soluble Film for Treatment of Nausea and Vomiting. [online] Available at: http://www.monosolrx.com/news_10/news_070710.html [Accessed 1 Nov. 2017].
This website had information regarding Zuplenz which is an oral film containing ondansetron. This information was used in Table 1 where I spoke about the current marketed formulation of oral films.
Morales, J. and McConville, J. (2011). Manufacture and characterization of mucoadhesive buccal films. European Journal of Pharmaceutics and Biopharmaceutics, [online] 77(2), pp.187-199. Available at: https://doi.org/10.1016/j.ejpb.2010.11.023 [Accessed 31 Oct. 2017].
This is a review that looked at past literature on the manufacturing process of oral films and the role of mucoadhesives in this process. From this article I was able to extract key information regarding the manufacturing process of oral films.
Peppas, N. and Buri, P. (1985). Surface, interfacial and molecular aspects of polymer bioadhesion on soft tissues. Journal of Controlled Release, 2, pp.257-275.
This article focuses on the oral mucosa and how using certain polymers can improve the bioadhesion to the oral mucosa’. I used this article when explaining the structure of the mucosa and drug delivery within it.
Ponchel, G. (1994). Formulation of oral mucosal drug delivery systems for the systemic delivery of bioactive materials. Advanced Drug Delivery Reviews, 13(1-2), pp.75-87.
This article focuses on drug delivery in the oral mucosa region. Areas highlighted are factors that can optimise drug delivery and how drug delivery takes place in this region. I used this article to talk about the target site for drug delivery in the oral mucosa and how this can be potentially optimised.
Pouton, C. (2006). Formulation of poorly water-soluble drugs for oral administration: Physicochemical and physiological issues and the lipid formulation classification system. European Journal of Pharmaceutical Sciences, 29(3-4), pp.278-287.
Article mentions the limitations of poorly soluble drugs and discusses ways to overcome this, one of which is by the use of surfactants which I have discussed in the proposal above.
Preis, M., Breitkreutz, J. and Sandler, N. (2015). Perspective: Concepts of printing technologies for oral film formulations. International Journal of Pharmaceutics, 494(2), pp.578-584.
This article contains information regarding the differences and methods of the orienting technologies used to formulate oral films. From this article I used the diagram shown in Figure 2 which is a representative diagram of printing technologies in general.
PubChem. (n.d.). Benzocaine [online] Available at: https://pubchem.ncbi.nlm.nih.gov/compound/2337#section=Solubility [Accessed 1 Nov. 2017].
PubChem is an electronic database containing information on the structure of drugs as well as their physico-chemical and pharmacokinetic data. I used this website to find out the water solubility of the benzocaine, so I could find out the range of solubility that the marketed products had as I was choosing to focus my project on poorly soluble drugs.
PubChem. (n.d.). Diphenhydramine [online] Available at: https://pubchem.ncbi.nlm.nih.gov/compound/3100#section=Solubility [Accessed 1 Nov. 2017].
PubChem is an electronic database containing information on the structure of drugs as well as their physico-chemical and pharmacokinetic data. I used this website to find out the water solubility of the diphenhydramine, so I could find out the range of solubility that the marketed products had as I was choosing to focus my project on poorly soluble drugs.
PubChem. (n.d.). Fentanyl [online] Available at: https://pubchem.ncbi.nlm.nih.gov/compound/3345#section=Solubility [Accessed 1 Nov. 2017].
PubChem is an electronic database containing information on the structure of drugs as well as their physico-chemical and pharmacokinetic data. I used this website to find out the water solubility of the fentanyl, so I could find out the range of solubility that the marketed products had as I was choosing to focus my project on poorly soluble drugs.
Pubchem. (2017). Loratadine. [online] Available at: https://pubchem.ncbi.nlm.nih.gov/compound/loratadine#section=Melting-Point [Accessed 31 Oct. 2017].
PubChem is an electronic database containing information on the structure of drugs as well as their physico-chemical and pharmacokinetic data. I used this website to find out the water solubility of the loratadine, so I could find out the range of solubility that the marketed products had as I was choosing to focus my project on poorly soluble drugs.
PubChem. (n.d.). Nicotine. [online] Available at: https://pubchem.ncbi.nlm.nih.gov/compound/89594#section=Solubility [Accessed 1 Nov. 2017].
PubChem is an electronic database containing information on the structure of drugs as well as their physico-chemical and pharmacokinetic data. I used this website to find out the water solubility of the nicotine, so I could find out the range of solubility that the marketed products had as I was choosing to focus my project on poorly soluble drugs.
PubChem. (n.d.). Ondansetron. [online] Available at: https://pubchem.ncbi.nlm.nih.gov/compound/4595#section=Solubility [Accessed 1 Nov. 2017].
PubChem is an electronic database containing information on the structure of drugs as well as their physico-chemical and pharmacokinetic data. I used this website to find out the water solubility of the ondansetron, so I could find out the range of solubility that the marketed products had as I was choosing to focus my project on poorly soluble drugs.
PubChem. (n.d.). Phenylephrine Hydrochloride [online] Available at: https://pubchem.ncbi.nlm.nih.gov/compound/5284443#section=Solubility [Accessed 1 Nov. 2017].
PubChem is an electronic database containing information on the structure of drugs as well as their physico-chemical and pharmacokinetic data. I used this website to find out the water solubility of the phenylephrine hydrochloride, so I could find out the range of solubility that the marketed products had as I was choosing to focus my project on poorly soluble drugs.
Rathbone, M., Drummond, B., and Tucker, I. (1994). The oral cavity as a site for systemic drug delivery. Advanced Drug Delivery Reviews, 13(1-2), pp.1-22.
This review discusses the advantages of delivering drugs via the oral mucosa, the composition of the membrane and environmental issues that may limit the use of the oral mucosa as a site for drug delivery. I used this article for the discussion of the structure of the oral mucosa and how this enables drug delivery, as well as for certain advantages/disadvantages regarding oral films.
Repka, M., Gutta, K., Prodduturi, S., Munjal, M. and Stodghill, S. (2005). Characterization of cellulosic hot-melt extruded films containing lidocaine. European Journal of Pharmaceutics and Biopharmaceutics, 59(1), pp.189-196.
This article focuses on the hot-metal extrusion process, which is one of the methods of formulating oral films. Advantages and disadvantages are also discussed in this article which is what I used for my research proposal.
Savjani, K., Gajjar, A. and Savjani, J. (2012). Drug Solubility: Importance and Enhancement Techniques. ISRN Pharmaceutics, 2012, pp.1-10.
This article mentions how poorly solubility can be overcome. I used this to discuss the role of surfactants in formulations and how they can overcome poor solubility.
Squier, C.A., Johnson, N.W. and Hopps, R.M., The organisation of oral mucosa, ln: Human Oral Mucosa Development, Structure and Function. Blackwell, Oxford, 1976a, pp. 7-16.
This is a book containing information regarding the oral mucosa and various regions have been focused on specifically, such as the oral epithelium, the connective tissue and barriers within the oral mucosa. I focused on chapter 1 of this book looking at the structure of the oral mucosa and the function of these structures.
Tabak, L., Levine, M., Mandel, I. and Ellison, S. (1982). Role of salivary mucins in the protection of the oral cavity. Journal of Oral Pathology and Medicine, 11(1), pp.1-17.
This article focuses on the role that saliva and mucus play in the protection of the oral cavity. It also touches on the role of saliva and mucus in the drug delivering process of oral drugs, this is the part which I used in this proposal.
Tesa Labtec. (2011). APR Applied Pharma Research s.a. and Labtec GmbH announce the European Approval of Ondansetron Rapidfilm®, co-developed in partnership with MonoSol Rx and licensed to BioAlliance Pharma SA for EU countries after completion of a decentralized procedure in the major 16 EU countries. [online] Available at: http://www.tesa-labtec.com/eng/company/press/european-approval-of-ondansetron-rapidfilm,1441613,1.html [Accessed 1 Nov. 2017].
This article gave information regarding a marketed oral film formulation containing ondansetron. I have used this information in Table 1 of this research proposal.
U.S. Food and Drug Administration. (2016). The Biopharmaceutics Classification System (BCS) Guidance. [online] Available at: https://www.fda.gov/AboutFDA/CentersOffices/OfficeofMedicalProductsandTobacco/CDER/ucm128219.htm [Accessed 17 Nov. 2017].
The FDA website contains information regarding the safety of drugs and information about classification systems. I used this website to talk about the BCS guidance, my project will be looking at class 2 drugs within this system.
Van Eerdenbrugh, B., Baird, J. and Taylor, L. (2010). Crystallization Tendency of Active Pharmaceutical Ingredients Following Rapid Solvent Evaporation—Classification and Comparison with Crystallization Tendency from Under cooled Melts. Journal of Pharmaceutical Sciences, 99(9), pp.3826-3838.
In this study a variety of compounds were studied for their crystallisation process and they were initially screened before being chosen and a variety of factors were taken into account, one of them being the class of the drug based on the BCS. I used this study to find out the class of some of the marketed oral films based on the BCS.
Vuddanda, P., Montenegro-Nicolini, M., Morales, J. and Velaga, S. (2017). Effect of surfactants and drug load on physico-mechanical and dissolution properties of nanocrystalline tadalafil-loaded oral films. European Journal of Pharmaceutical Sciences, [online] 109, pp.372-380. Available at: https://doi.org/10.1016/j.ejps.2017.08.019 [Accessed 1 Nov. 2017].
This article focused on how hydrophilic surfactants and drug load can affect the physico-mechanical and dissolution properties of oral films. The active ingredient used in this research was tadalafil which is poorly soluble, so I used this article to support the area of research that I will be considering.
GANTT chart
This dissertation proposal has been written by a student and is published as an example. See our guide on How to Write a Dissertation Proposal for guidance on writing your own proposal.
Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allRelated Content
All TagsContent relating to: "Medical"
The word Medical refers to preventing or treating injuries or illnesses, relating to the study or practice of medicine. Medical care involves caring for a patient and helping them through their journey to recovery.
Related Articles
DMCA / Removal Request
If you are the original writer of this dissertation proposal and no longer wish to have your work published on the UKDiss.com website then please:




