ACDF Outcomes with Perioperative Neurological Complications
Info: 7259 words (29 pages) Dissertation
Published: 21st Feb 2022
Tagged: Neurology
Perioperative Neurological Complications Following Anterior Spinal Discectomy and Fusion: Clinical Impact on 317,789 Patients from the National Inpatient Sample
ABSTRACT
Introduction: Perioperative neurological complication may occur after an anterior cervical discectomy and fusion (ACDF) with devastating consequences. We aim to estimate the incidence of Perioperative neurological complications, identify their risk factors, and to evaluate their impact on morbidity and mortality after ACDF.
Methods: ACDF cases (n=317,789 patients) were extracted from the National Inpatient Sample (NIS) between 1999 and 2011. Based on their Elixhauser-van Walraven score (VWR) score, patients were classified as low (VWR14) for surgery. The primary outcome was perioperative neurological complications, which were defined as events with potential central nervous system injury including spinal cord injury, cerebral hypoxia, cerebrospinal fluid leak, postoperative stroke, or any other unspecified nervous system complication. Secondary outcomes included in-house morbidity (hospital length of stay >14 days or discharge disposition to a location other than home) and in-hospital mortality.
Results: The rate of perioperative neurological complications, morbidity, and mortality after ACDF was 0.4%, 8.4%, and 0.1%, respectively. Perioperative neurological complications were highly associated with in-house morbidity (OR, 3.7 [3.1-4.4]) and mortality (OR, 8.0 [4.1-15.5]). The strongest predictors for perioperative neurological complications were moderate (OR, 3.1 [2.6-3.7]) and high-risk VWR (OR, 5.4 [3.3-8.9]), hematoma/seroma formation (OR, 5.4 [3.9-7.4]), and obesity (OR, 1.9 [1.6-2.3]). The rate of perioperative neurological complications increased from 0.2% to 0.7% from 1999 to 2011, which was temporally associated with the rise in moderate (R2=0.57, p=0.002) and high-risk patients (R2=0.61, p=0.001) undergoing ACDF.
Conclusion: Perioperative neurological complications are independent predictors of in-hospital morbidity and mortality after ACDF. Both morbidity and perioperative neurological complications have increased between 1999 and 2011, which may be due, in part, to increasing numbers of moderate and high-risk patients undergoing ACDF.
Key Words: Anterior Cervical Discectomy and Fusion; In-hospital Outcomes; Mortality; Perioperative Neurological Complications; Intraoperative Neuromonitoring
INTRODUCTION
Anterior cervical discectomy and fusion (ACDF) is an effective and commonly utilized approach for the treatment of cervical radiculopathy and myelopathy.1,27 Between 1990-1999 approximately 540,000 ACDFs were performed in the United States,1 and this figure appears to be increasing in recent years.27 Clinical and radiographic outcomes are generally favorable in most patients,2,20,24 with an estimated mortality and morbidity of 0.1% and 19%, respectively.10 Intraoperative and immediate postoperative complications most commonly include dural tear (3.7%),5,8 airway compromise due to edema or hematoma (1.7-6.0%),5,8,9,22 and epidural hematomas (0.2-1.9%).5,8
Perioperative neurological complications, including myelopathic (0.2-0.9%)5,8,13,26 and non-myelopathic injury (0.2-3.2%),3,5,14,26 are devastating and can result from vascular insult, distraction, and direct mechanical compression of the spinal cord and nerve roots.15,27 These injuries are particularly unfortunate because they can often be monitored and prevented with the use of intraoperative neurophysiologic monitoring techniques, such as transcranial motor and somatosensory evoked potentials.17,28 Patients who experience iatrogenic neurological injuries may sustain adverse effects that range from transient numbness or paresthesias to irreversible paralysis.23
Previous studies have described the incidence of perioperative neurological complications following ACDF procedures; however these data are largely limited to single institutional reviews.5,8,14,26 In a previous study, we demonstrated that spine surgery-associated perioperative neurological complications between 1999-2011 were associated with increased morbidity, mortality, as well as healthcare expenditures, and that the national incidence of Perioperative neurological complications is increasing.27 Among the three most commonly performed spine surgeries (ACDFs, posterior cervical fusions, and thoracolumbar fusions), ACDFs represented the largest proportion of procedures (82%, 1,486,083 of 1,828,156 total cases), and thus the largest complication burden. In that study, the average incidence of perioperative neurological complications following ACDFs was 0.61% on initial procedures and 0.67% on revision procedures.
Our previous analyses provided information regarding the national incidence and trends of perioperative neurological complications over time following spinal fusion surgery, however several questions remain unanswered.6 For instance, given the high volume of ACDFs and the increasing incidence of perioperative neurological deficits, it seems pertinent to understand why certain patients sustain complications after ACDFs, while others do not. Prior literature has suggested that patients with previous myelopathy, cervical kyphosis, spinal cord atrophy, spinal instability, and fractures through long, fused spinal segments may be particularly susceptible to spinal cord injury.5 Strategies such as maintaining intraoperative systolic blood pressure greater than 80 mmHg and avoidance of excessive extension and distraction of the cervical spine during positioning may further protect against neural injury.5 Data assessment utilizing a large nationwide population-based cohort may allow for broader and more sophisticated multivariate analyses to understand the relative contribution of risk factors such as demographics, patient comorbidity load, and other perioperative events on the incidence of neurological deficits.
Thus, the primary aim of this study was to describe and quantify factors associated with perioperative neurological complications after ACDF. Secondarily, we aim to measure the clinical impact, namely morbidity and mortality, of patients sustaining these deficits after ACDF surgery. Understanding patient-specific risk factors for and clinical sequelae of neurological complications may help to further facilitate the safety of ACDF surgery through improved preoperative screening, intraoperative monitoring, and postoperative management protocols.
METHODS
Database
The National Inpatient Sample (NIS) Database, which is indexed by ICD-9 (International Classification of Diseases) billing codes for patient diagnoses and procedures, was obtained from the Healthcare Cost and Utilization Project. The database samples from 95% of all hospitals nationwide and contains about 7 million total hospital admissions from across 44 states. Furthermore, the NIS database contains patients of all payer types, including Medicare, Medicaid, private insurance, and uninsured.
Data Extraction
Cases were extracted from the NIS between 1999 and 2011 using the ICDF-9 code for ACDF (81.02). We collected baseline patient characteristics including age, gender, and admission type. Patient comorbidity load was evaluated using the Elixhauser-van Walraven score (VWR), which is calculated from common diagnoses that are associated with in-house mortality.29 Patients were classified as low- (VWR14) based on their VWR score. Of note, patients with trauma (800-897.7, 733.1-733.19) were excluded to eliminate cases with preoperative neurological injuries.
Outcomes
The primary outcome was perioperative neurological complications, which were defined as events with potential central nervous system injury including spinal cord injury without spinal bone injury, cerebral hypoxia, cerebrospinal fluid leak, postoperative stroke, or any other unspecified nervous system complication (ICD-9 952.0-952.09, 952.1-952.19, 997.0-997.09). Secondary outcomes included in-house morbidity and in-hospital mortality. Morbidity was defined as hospital length of stay greater than 14 days or discharge disposition to a location other than home.
Statistical Methods
Initially, a univariate analysis was performed to evaluate for factors significantly associated with perioperative neurological complications after ACDF. Results that were deemed statistically significant (p
RESULTS
Our study demonstrates that perioperative neurological complications are independent predictors of both in-hospital morbidity and mortality after ACDF. Risk factors for perioperative neurological complications include moderate and high-risk VWR, hematoma formation, and obesity. Both morbidity and perioperative neurological complications have increased between 1999 and 2011, which may be due, in part, to increasing numbers of moderate- and high-risk patients undergoing ACDF.
Patient Characteristics and Postoperative Complications
A total of 317,789 patients were included in this study. Mean age was 51.7 (95% CI, 51.5-51.9) years. The majority of patients were between 65-74 years old (61%), were stratified as low-risk VWR (93%), and presented on an elective basis (90%). The most common comorbidities were hypertension (35%), chronic pulmonary disease (13%), and uncomplicated diabetes (11%). The rate of in-hospital morbidity and mortality after ACDF was 8.4% and 0.1%, respectively. The total rate of perioperative neurological complications was 0.4%. Other notable postoperative complications included myocardial infarction (2%), dysphagia (1%), bleeding (1%), intraspinal abscess (0.2%), and hoarseness (0.1%). Detailed patient characteristics and postoperative complications are presented in Table 1.
Trends in baseline patient characteristics and postoperative complications over time are displayed in Figure 1. From the years 1999-2011, the comorbidity load of patients undergoing ACDF increased with time. Over the 12-year period, the rate of low-risk VWR patients decreased from 95.6% to 91.0%; moderate-risk VWR patients increased from 4.1% to 8.3%; and high-risk VWR patients increased from 0.3% to 0.8%. In-house morbidity also increased from 6.8% to 11.3% and the rate of perioperative neurological complications increased from 0.2% to 0.7% from 1999 to 2011. In-house mortality was stable between 0.11 and 0.18%. Notably, the increasing annual rate of perioperative neurological complications was temporally associated with the rise in moderate- (R2=0.57, p=0.002) and high-risk patients (R2=0.61, p=0.001) undergoing ACDF (Figures 2A and 2B). Furthermore, the rise in morbidity from 1999-2011 was strongly associated with the rise in moderate- (R2=0.88, p2=0.97, p
Impact and risk factors of Perioperative neurological complications
After performing an initial univariate regression, all significant variables were selected for inclusion into a multivariable logistic regression analysis to identify independent predictors of in-hospital morbidity and mortality. Almost all baseline patient characteristics and postoperative complications were significantly associated with morbidity and mortality and were thus included in the multivariate analysis. The rate of perioperative neurological complications in patients with in-house morbidity and mortality was 2% and 7%, respectively (Table 1). Conversely, the rate of morbidity and mortality in patients with perioperative neurological complications was 37% and 3%, respectively. Figures 3 and 4 display the results of our multivariate logistic regression to identify independent risk factors for morbidity and mortality. Here, perioperative neurological complications were highly associated with in-house morbidity (OR, 3.7 [3.1-4.4]) and mortality (OR, 8.0 [4.1-15.5]). Other predictors of morbidity after ACDF were intraspinal abscess (OR, 38.0 [26.7-53.9]), pneumonia (OR, 12.1 [3.6-40.3]), infection (OR, 10.9 [7.0-16.8]), and paralysis (OR, 8.9 [7.9-10.0]). Risk factors for in-hospital mortality included moderate- (OR, 5.3 [3.7-7.6]) and high-risk VWR (OR, 8.0 [4.1-15.5]), perioperative neurological complications (OR, 6.4 [3.8-10.5]), and DVT/PE (deep venous thrombosis/pulmonary embolus) (OR, 4.7 [2.9-7.6]).
Figure 5 displays all independent predictors of perioperative neurological complications. The strongest predictors for perioperative neurological complications were moderate (OR, 3.1 [2.6-3.7]) and high-risk VWR (OR, 5.4 [3.3-8.9]), hematoma/seroma formation (OR, 5.4 [3.9-7.4]), and obesity (OR, 1.9 [1.6-2.3]). Variables such as dysphagia, hoarseness, and urinary/renal complications were strongly associated with perioperative neurological complications, although these are likely to be the result of rather than actual risk factors of perioperative neurological complications.
DISCUSSION
Through the use of a large, multi-center national database, this study approximates the frequency of perioperative neurological complications after ACDF surgery and analyzes their impact and risk factors for development. Common perioperative neurological complications following ACDF include vocal cord paresis or dysphagia due to injury of the recurrent laryngeal or vagus nerve (4-11%),19 nerve root injury (0.2-3.2%),3,5,14,26 and spinal cord injury (0.2-0.9%).5,8,13,26 The incidence observed in our study (0.4%) appears to be lower than that reported in the literature, suggesting an underreporting of perioperative neurological complications after ACDF.
We observed that perioperative neurological complications, although rare, are significantly associated with increased in-hospital morbidity and mortality. We were unable to discern the direct cause of morbidity or mortality following a new postoperative deficit; however, it is reasonable to assume that perioperative neurological complications propagate downstream complications that prolong length of stay and increase the risk of mortality. For instance, patients with intraoperative recurrent laryngeal nerve injury are at increased risk for aspiration pneumonia, which could precipitate an extended hospital course and possibly death. Patients that harbor spinal cord or nerve root injuries are increasingly immobile and can develop DVTs and/or PE as a result.
Given the impact of perioperative neurological complications on patient outcomes, patients should be carefully risk stratified and medically optimized preoperatively to decrease the rate of neural injury. Our study demonstrated that moderate and high-risk VWR as well as obesity were independent risk factors for the development of perioperative neurological complications. Furthermore, our data suggest that over the past 12 years, surgeons have been accepting an increasing number of high-risk patients for ACDF surgery. These patients may benefit from prehabilitation exercise regimens aimed at managing modifiable risk factors and improving cardiovascular and respiratory conditioning.4 Granted, many high-risk patients must undergo surgery on an emergent basis, and in these cases, careful preoperative risk stratification and optimization may be more difficult.
Intraoperative neuromonitoring (IONM) techniques such as SSEPs (somatosensory evoked potentials) and MEPs (motor evoked potentials) can be instrumental in detecting neurological insults in the operating theatre, which can prompt surgical corrective action. Studies on the diagnostic accuracy of intraoperative neuromonitoring during cervical spine surgery have shown that SSEPs are highly specific (96-100%), while MEPs are both sensitive (100%) and specific (96-98%) for new postoperative neurological deficits.16,30 Furthermore, the use of SSEPs has been shown to minimize neurological injuries during cervical spine surgery. In a single-surgeon series of 809 patients, Roh et al. estimates that SSEP monitoring prevented new postoperative neurological deficits in 1.9% of patients.21
However, IONM for cervical spine surgery is still not universally accepted among surgeons. Some feel that it is not worth utilizing due to the low incidence of perioperative neurological deficits.25 Other authors report suboptimal sensitivity when using SSEPs as a standalone method, which subsequently fails to inform surgical decision-making.11,18,25 We feel that although the incidence of perioperative neurological deficits is low, given the risk for devastating consequences such as quadriplegia, the benefits of neuromonitoring outweigh its shortcomings. Furthermore, the accuracy of IONM is only fully realized when using multimodal monitoring including EMGs (electromyography), SSEPs, and MEPs to provide a global assessment of cord and nerve root function.16,30
Our study is limited by several methodological factors. Primarily we were limited by the use of an administrative database, which is based on insurance claims data. Although large and offers a national perspective, this data can often be inaccurate, is subject to reporting bias, and does not provide detailed information regarding a patient’s hospital stay. For instance, information such as the use of IONM, indications for the procedure (radiculopathy, myelopathy, etc.), or radiographic findings, which may have influenced postoperative outcomes, are not available through the NIS database. Additionally, our study only contains short-term hospital outcomes and thus, does not delineate the transiency of neurological deficits or provide information on postoperative functional status.
Future studies should focus on further developing techniques for monitoring and preventing perioperative neurological deficits. From a pathophysiological standpoint, animal studies are needed in order to accurately model iatrogenic neural injuries so that potential treatments can be developed. We recently developed a model of iatrogenic spinal cord injury in rats and validated the use of SSEPs and MEPs in predicting neurological injury.27 Such a model could be useful in further optimizing IONM parameters and alarm criteria for predicting intraoperative neural injury. Furthermore, real-time simulation in the laboratory of iatrogenic perioperative neurological complications under IONM offers the ability to develop and trial potential therapies under experimental conditions. Neuroprotective techniques such as hypothermia and steroid use have been described in the traumatic spinal cord injury literature,7,12 but similar paradigms have not been employed intraoperatively for iatrogenic injury. Further study on IONM techniques as well as development of pharmacologic agents for intraoperative use will help to minimize the risk of perioperative neurological deficits, thus improving the safety and efficacy of spine surgery.
CONCLUSION
The incidence of perioperative neurological complications is low, but this rate has been increasing, and may be due to the increasing frequency of high-risk patients undergoing ACDF. Risk factors for perioperative neurological complications include obesity, hematoma formation, and moderate- and high-risk VWR scores. Patients who develop perioperative neurological complications after ACDF are at increased risk for morbidity and in-hospital mortality. Given these findings, efforts should be focused on screening high-risk patients for participation in prehabilitation programs in preparation for surgery. Continued study of IONM techniques and basic science models for iatrogenic neural injury can help to identify, prevent, and treat perioperative neurological deficits intraoperatively and facilitate improved patient outcomes.
REFERENCES
1. Angevine PD, Arons RR, McCormick PC: National and regional rates and variation of cervical discectomy with and without anterior fusion, 1990-1999. Spine (Phila Pa 1976) 28:931-939; discussion 940, 2003
2. Barnes B, Haid RW, Rodts GE, Subach BR, Kaiser M: Early results using the Atlantis anterior cervical plate system. Neurosurg Focus 12:E13, 2002
3. Bertalanffy H, Eggert HR: Complications of anterior cervical discectomy without fusion in 450 consecutive patients. Acta Neurochir (Wien) 99:41-50, 1989
4. Cabilan CJ, Hines S, Munday J: The effectiveness of prehabilitation or preoperative exercise for surgical patients: a systematic review. JBI Database System Rev Implement Rep 13:146-187, 2015
5. Daniels AH, Riew KD, Yoo JU, Ching A, Birchard KR, Kranenburg AJ, et al: Adverse events associated with anterior cervical spine surgery. J Am Acad Orthop Surg 16:729-738, 2008
6. Dayananda S, Mehta A, Agarwal N, Nwachuku EL, Hamilton DK, Thirumala PD: Impact of Perioperative Neurological Deficits on Clinical Outcomes Following Posterior Cervical Fusion. World Neurosurg, 2018
7. Dietrich WD, Levi AD, Wang M, Green BA: Hypothermic treatment for acute spinal cord injury. Neurotherapeutics 8:229-239, 2011
8. Emery SE, Bohlman HH, Bolesta MJ, Jones PK: Anterior cervical decompression and arthrodesis for the treatment of cervical spondylotic myelopathy. Two to seventeen-year follow-up. J Bone Joint Surg Am 80:941-951, 1998
9. Epstein NE, Hollingsworth R, Nardi D, Singer J: Can airway complications following multilevel anterior cervical surgery be avoided? J Neurosurg 94:185-188, 2001
10. Fountas KN, Kapsalaki EZ, Nikolakakos LG, Smisson HF, Johnston KW, Grigorian AA, et al: Anterior cervical discectomy and fusion associated complications. Spine (Phila Pa 1976) 32:2310-2317, 2007
11. Ginsburg HH, Shetter AG, Raudzens PA: Postoperative paraplegia with preserved intraoperative somatosensory evoked potentials. Case report. J Neurosurg 63:296-300, 1985
12. Hall ED, Springer JE: Neuroprotection and acute spinal cord injury: a reappraisal. NeuroRx 1:80-100, 2004
13. Hilibrand AS, Schwartz DM, Sethuraman V, Vaccaro AR, Albert TJ: Comparison of transcranial electric motor and somatosensory evoked potential monitoring during cervical spine surgery. J Bone Joint Surg Am 86-A:1248-1253, 2004
14. Ikenaga M, Shikata J, Tanaka C: Radiculopathy of C-5 after anterior decompression for cervical myelopathy. J Neurosurg Spine 3:210-217, 2005
15. Kamerlink JR, Errico T, Xavier S, Patel A, Patel A, Cohen A, et al: Major intraoperative neurologic monitoring deficits in consecutive pediatric and adult spinal deformity patients at one institution. Spine (Phila Pa 1976) 35:240-245, 2010
16. Kelleher MO, Tan G, Sarjeant R, Fehlings MG: Predictive value of intraoperative neurophysiological monitoring during cervical spine surgery: a prospective analysis of 1055 consecutive patients. J Neurosurg Spine 8:215-221, 2008
17. Lee JY, Hilibrand AS, Lim MR, Zavatsky J, Zeiller S, Schwartz DM, et al: Characterization of neurophysiologic alerts during anterior cervical spine surgery. Spine (Phila Pa 1976) 31:1916-1922, 2006
18. Lesser RP, Raudzens P, Luders H, Nuwer MR, Goldie WD, Morris HH, 3rd, et al: Postoperative neurological deficits may occur despite unchanged intraoperative somatosensory evoked potentials. Ann Neurol 19:22-25, 1986
19. Netterville JL, Koriwchak MJ, Winkle M, Courey MS, Ossoff RH: Vocal fold paralysis following the anterior approach to the cervical spine. Ann Otol Rhinol Laryngol 105:85-91, 1996
20. Papadopoulos EC, Huang RC, Girardi FP, Synnott K, Cammisa FP, Jr.: Three-level anterior cervical discectomy and fusion with plate fixation: radiographic and clinical results. Spine (Phila Pa 1976) 31:897-902, 2006
21. Roh MS, Wilson-Holden TJ, Padberg AM, Park JB, Daniel Riew K: The utility of somatosensory evoked potential monitoring during cervical spine surgery: how often does it prompt intervention and affect outcome? Asian Spine J 1:43-47, 2007
22. Sagi HC, Beutler W, Carroll E, Connolly PJ: Airway complications associated with surgery on the anterior cervical spine. Spine (Phila Pa 1976) 27:949-953, 2002
23. Schwartz DM, Auerbach JD, Dormans JP, Flynn J, Drummond DS, Bowe JA, et al: Neurophysiological detection of impending spinal cord injury during scoliosis surgery. J Bone Joint Surg Am 89:2440-2449, 2007
24. Shen FH, Samartzis D, Khanna N, Goldberg EJ, An HS: Comparison of clinical and radiographic outcome in instrumented anterior cervical discectomy and fusion with or without direct uncovertebral joint decompression. Spine J 4:629-635, 2004
25. Taunt CJ, Jr., Sidhu KS, Andrew SA: Somatosensory evoked potential monitoring during anterior cervical discectomy and fusion. Spine (Phila Pa 1976) 30:1970-1972, 2005
26. Tew JM, Jr., Mayfield FH: Complications of surgery of the anterior cervical spine. Clin Neurosurg 23:424-434, 1976
27. Thirumala P, Zhou J, Natarajan P, Balzer J, Dixon E, Okonkwo D, et al: Perioperative neurologic complications during spinal fusion surgery: incidence and trends. Spine J 17:1611-1624, 2017
28. Thirumala PD, Muralidharan A, Loke YK, Habeych M, Crammond D, Balzer J: Value of intraoperative neurophysiological monitoring to reduce neurological complications in patients undergoing anterior cervical spine procedures for cervical spondylotic myelopathy. J Clin Neurosci 25:27-35, 2016
29. van Walraven C, Austin PC, Jennings A, Quan H, Forster AJ: A modification of the Elixhauser comorbidity measures into a point system for hospital death using administrative data. Med Care 47:626-633, 2009
30. Xu R, Ritzl EK, Sait M, Sciubba DM, Wolinsky JP, Witham TF, et al: A role for motor and somatosensory evoked potentials during anterior cervical discectomy and fusion for patients without myelopathy: Analysis of 57 consecutive cases. Surg Neurol Int 2:133, 2011
FIGURES
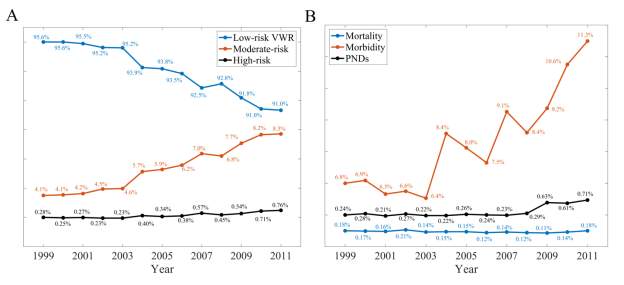
Figure 1. (A) Proportions of low, medium, and high-risk patients undergoing ACDF and (B) prevalence of mortality, morbidity, and Perioperative neurological complications after ACDF from 1999-2011.
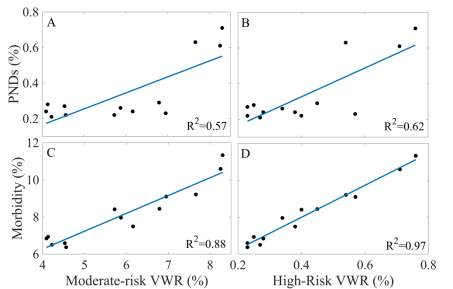
Figure 2. Scatterplots and corresponding correlation (R2) between annual rates of (A) Perioperative neurological complications vs. moderate risk VWR, (B) Perioperative neurological complications vs. high-risk VWR, (C) morbidity vs. moderate-risk VWR, and (D) morbidity vs. high-risk VWR.
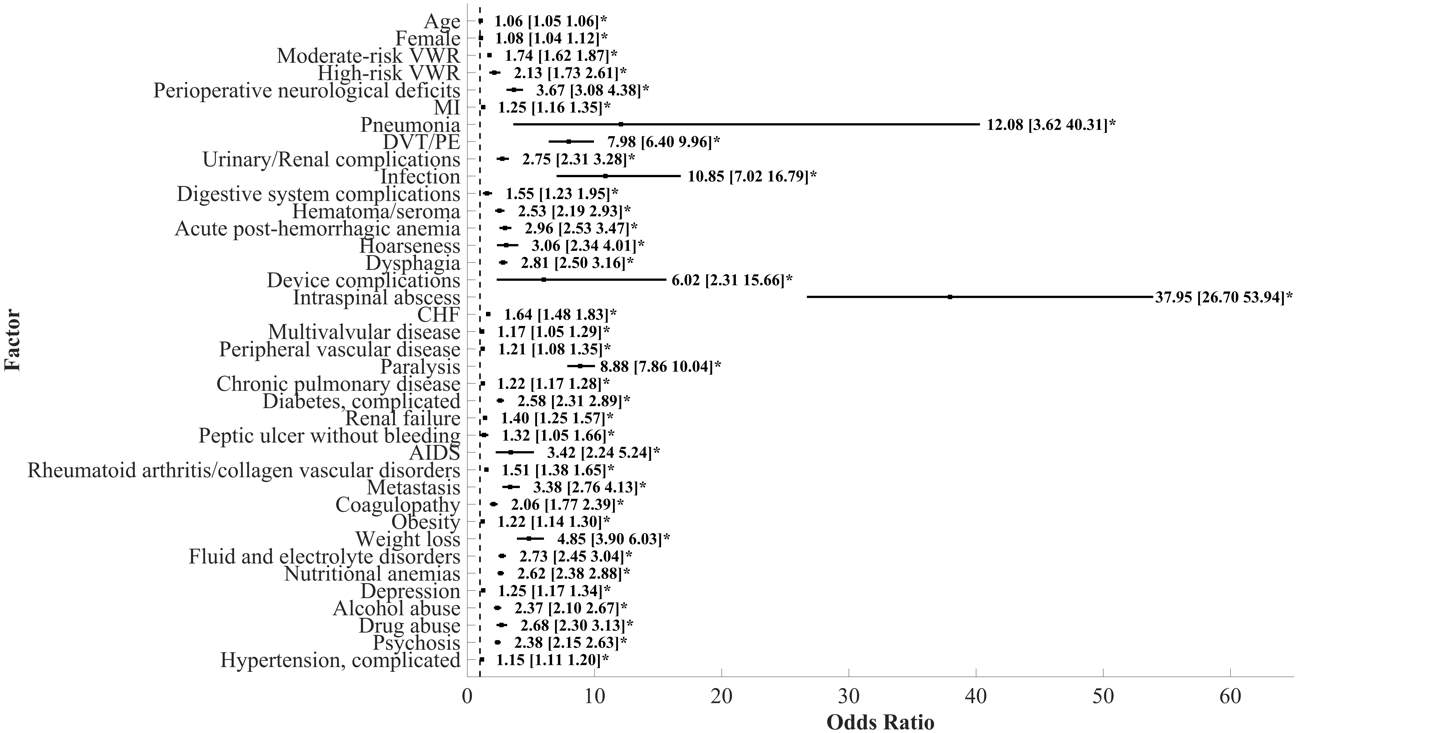
Figure 3. Predictors of morbidity after ACDF with OR and 95% CI. Asterisk (*) indicates OR that does not cross dashed line (OR=1) and is statistically significant.
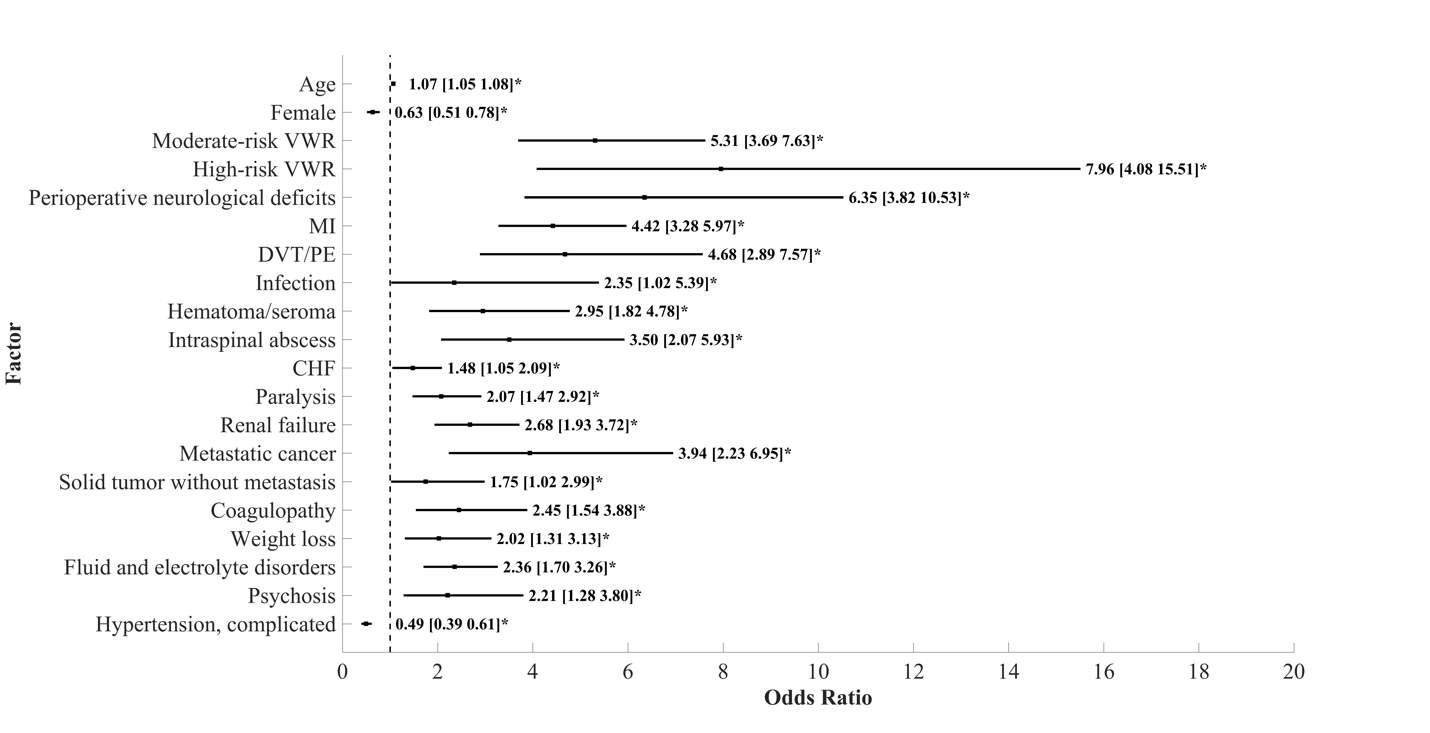
Figure 4. Predictors of mortality after ACDF with OR and 95% CI. Asterisk (*) indicates OR that does not cross dashed line (OR=1) and is statistically significant.
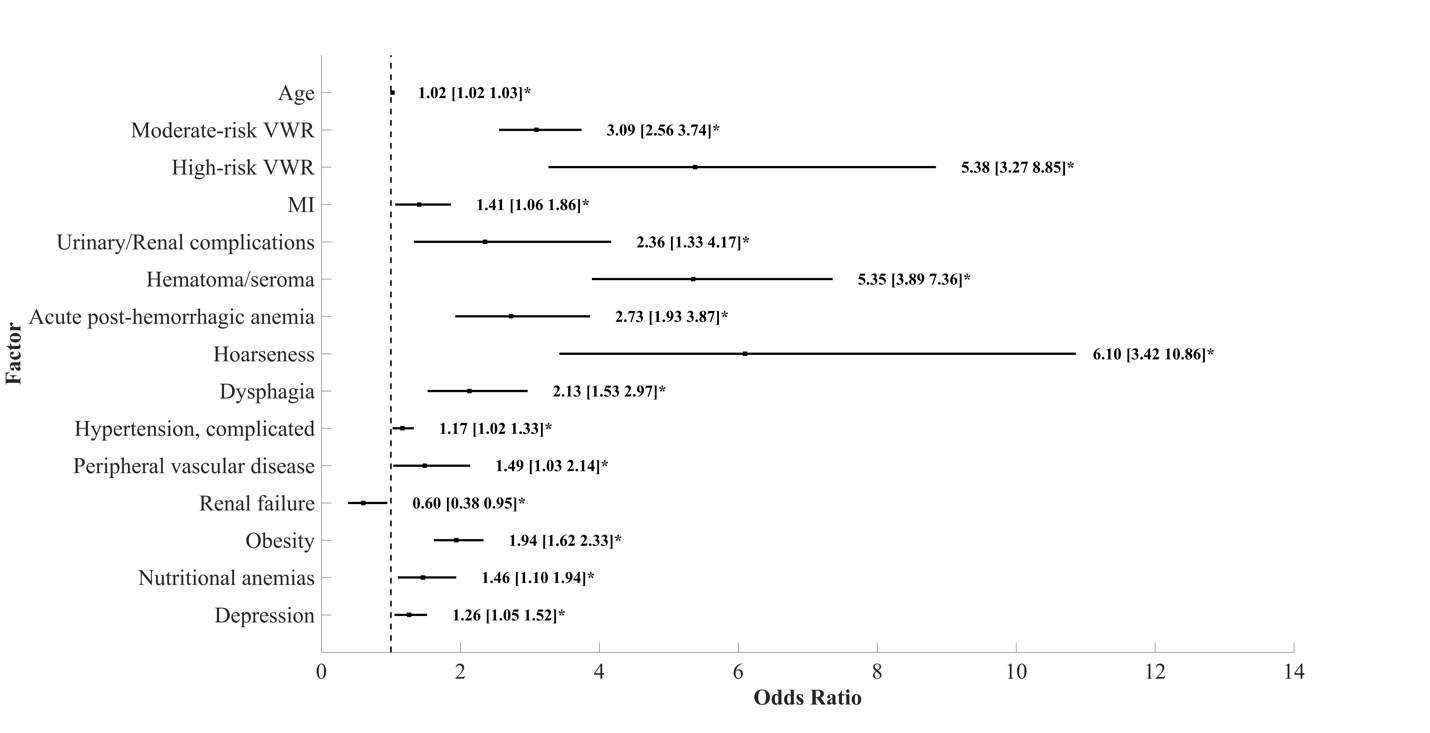
Figure 5. Predictors of Perioperative neurological complications after ACDF with OR and 95% CI. Asterisk (*) indicates OR that does not cross dashed line (OR=1) and is statistically significant.
TABLES
| Table 1. Univariate analysis for in-hospital morbidity and mortality. | ||||||
| Variable | Total value | Morbidity (no, yes) | p-value | Mortality (no, yes) | p-value | |
| Mean age (95% CI) | 51.7 (51.5-51.9) | 50.9, 60.5 | 51.7, 67.0 | |||
| 16% | 17%, 7% | 16%, 2% | ||||
| 65-74 | 61% | 63%, 42% | 61%, 30% | |||
| 75-84 | 21% | 19%, 44% | 21%, 51% | |||
| >84 | 1% | 1%, 6% | 1%, 17% | |||
| Gender (female) | 52% | 52.3%, 51.5% | 0.076 | 52%, 32% | ||
| Race | ||||||
| White | 83% | 84%, 73% | 83%, 71% | |||
| African American | 9% | 8%, 16% | 9%, 18% | |||
| Hispanic | 5% | 5%, 7% | 5%, 7% | |||
| Asian | 1% | 1%, 2% | 1%, 2% | |||
| Native American | 0.3% | 0.3%, 0.3% | 0.3%, 0% | |||
| Admission type | ||||||
| Emergent | 4% | 3%, 17% | 4%, 30% | |||
| Urgent | 7% | 6%, 11% | 7%, 18% | |||
| Elective | 90% | 91%, 72% | 90%, 51% | |||
| Mean VWR score (95% CI) | 0.39 (0.36-0.41) | 0.2, 2.6 | 0.4, 8.9 | |||
| Low-risk (VWR | 93% | 95%, 73% | 93%, 29% | |||
| Moderate-risk (VWR 5-14) | 6% | 5%, 24% | 6%, 47% | |||
| High-risk (VWR>14) | 0.4% | 0.1%, 4% | 0.4%, 24% | |||
| Elixhauser comorbidity index | ||||||
| Congestive heart failure | 1% | 1%, 5% | 1%, 19% | |||
| Multivalvular disease | 2% | 2%, 4% | 2%, 4% | 0.016 | ||
| Pulmonary hypertension | 0.2% | 0.1%, 0.8% | 0.2%, 4% | |||
| Peripheral vascular disease | 1% | 1%, 3% | 1%, 6% | |||
| Paralysis | 1% | 1%, 10% | 1%, 22% | |||
| Other Neurological disorders | 2% | 2%, 6% | 2%, 15% | |||
| Chronic pulmonary disease | 13% | 12%, 20% | 13%, 25% | |||
| Diabetes, uncomplicated | 11% | 11%, 20% | 12%, 22% | |||
| Diabetes, complicated | 1% | 1%, 4% | 1%, 6% | |||
| Hypothyroidsm | 7% | 6%, 10% | 7%, 5% | 0.069 | ||
| Renal Failure | 1% | 1%, 5% | 1%, 19% | |||
| Liver disease | 1% | 1%, 2% | 1%, 3% | 0.008 | ||
| Peptic ulcer disease with bleeding | 0.3% | 0.3%, 0.5% | 0.3%, 0.2% | 0.669 | ||
| AIDS | 0.1% | 0.1%, 0.2% | 0.1%, 0.2% | 0.562 | ||
| Lymphoma | 0.2% | 0.1%, 0.4% | 0.2%, 1% | 0.095 | ||
| Metastatic cancer | 0.2% | 0.1%, 1.5% | 0.2%, 7% | |||
| Solid tumor without metastasis | 0.9% | 1%, 2% | 1%, 4% | |||
| Rheumatoid arthritis/collagen vascular disorders | 2% | 2%, 4% | 0.2%, 3% | 0.160 | ||
| Coagulopathy | 1% | 0.3%, 2% | 1%, 10% | |||
| Obesity | 7% | 7%, 9% | 7%, 7% | 0.967 | ||
| Weight loss | 0.3% | 0.1%, 2% | 0.2%, 11% | |||
| Fluid and electrolyte disorders | 2% | 1%, 11% | 2%, 34% | |||
| Chronic blood loss anemia | 0.1% | 0.1%, 0.5% | 0.1%, 0.7% | 0.143 | ||
| Nutritional anemias | 2% | 2%, 8% | 2%, 12% | |||
| Alcohol abuse | 1% | 1%, 3% | 1%, 4% | 0.004 | ||
| Drug abuse | 1% | 1%, 2% | 1%, 2% | 0.131 | ||
| Psychoses | 2% | 1%, 4% | 2%, 4% | 0.005 | ||
| Depression | 9% | 9%, 12% | 10%, 5% | |||
| Hypertension | 35% | 34%, 53% | 35%, 44% | |||
| Perioperative complications | ||||||
| Perioperative neurological deficits | 0.4% | 0.2%, 2% | 0.3%, 7% | |||
| Myocardial infarction | 2% | 2%, 5% | 2%, 23% | |||
| DVT/PE | 0.2% | 0.1%, 2% | 0.2%, 10% | |||
| Infection | 0.1% | 0%, 0.6% | 0.1%, 2% | 0.002 | ||
| Acute post-hemorrhagic anemia | 1% | 0.5%, 4% | 1%, 6% | |||
| Hoarseness | 0.1% | 0.1%, 0.5% | 0.1%, 1% | 0.023 | ||
| Dysphagia | 1% | 1%, 4% | 1%, 4% | 0.006 | ||
| Intraspinal abscess | 0.2% | 0%, 2% | 0.2, 6% | |||
Cite This Work
To export a reference to this article please select a referencing stye below:
Related Services
View allRelated Content
All TagsContent relating to: "Neurology"
Neurology is the specialist branch of medicine that deals with the treatment of disorders of the nervous system. This means that neurologists concern themselves with issues affecting the brain, the nerves, and the spinal cord.
Related Articles
DMCA / Removal Request
If you are the original writer of this dissertation and no longer wish to have your work published on the UKDiss.com website then please:




